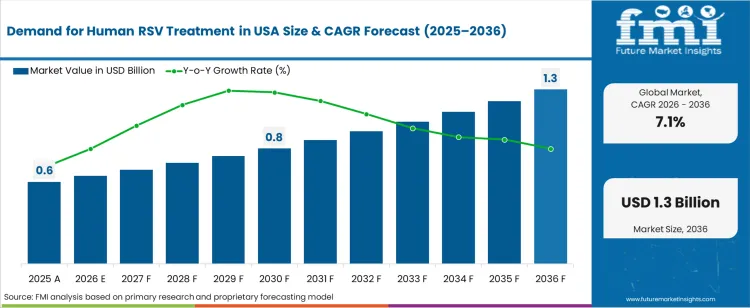
The demand for human rsv treatment in usa was valued at USD 0.60 billion in 2025, projected to reach USD 0.64 billion in 2026, and is forecast to expand to USD 1.28 billion by 2036 at a 7.1% CAGR. The USA's large paediatric and elderly population base, combined with expanding insurance coverage for RSV prophylactic biologics, is driving sustained growth in RSV treatment expenditure. FDA approvals for nirsevimab (infant prophylaxis) and RSV maternal vaccines have restructured the treatment pathway, shifting a portion of disease management costs from inpatient supportive care toward outpatient immunisation and prophylactic antibody administration. The Advisory Committee on Immunization Practices (ACIP) recommendations for universal infant RSV immunisation are expected to generate sustained institutional procurement volumes.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 0.64 billion |
| Industry Value (2036) | USD 1.28 billion |
| CAGR (2026 to 2036) | 7.1% |
Source: Future Market Insights, 2026
The incremental opportunity between 2026 and 2036 amounts to USD 0.64 billion. FMI is of the opinion that the growth trajectory reflects the high per-patient cost of biologic prophylactics combined with expanding eligibility criteria across paediatric and elderly populations. Commercial and Medicaid payer coverage for RSV monoclonal antibodies is broadening access, while the Vaccines for Children programme is extending immunisation coverage to uninsured and underinsured paediatric populations. Hospital-based supportive care remains a major cost component for breakthrough infections and for patients not reached by prophylactic programmes.
Sub-national growth trajectories reflect paediatric population density and payer coverage expansion. West at 8.2%, South at 7.3%, Northeast at 6.6%, Midwest at 5.7%. The West leads, driven by California's large paediatric population and early commercial payer adoption of RSV prophylaxis coverage. The South follows, where high RSV hospitalisation rates and expanding Medicaid coverage across sunbelt states generate concentrated treatment demand. The Northeast benefits from dense academic medical centre networks with established RSV clinical protocols, while the Midwest reflects steady growth from community hospital immunisation programme rollouts.
Human RSV treatment encompasses the clinical management of respiratory syncytial virus infections through supportive care interventions, hospital-based treatment protocols, and prophylactic biologic administration. The USA market covers supportive care (oxygen supplementation, fluid management, respiratory support) and hospital care (ICU admission, mechanical ventilation) delivered across hospitals, ambulatory surgical centres, and clinics. The scope includes prophylactic monoclonal antibody administration and maternal RSV vaccination services.
Market scope includes oxygen therapy, intravenous fluid management, bronchodilator administration, respiratory monitoring, ICU admissions, prophylactic monoclonal antibody administration (nirsevimab, palivizumab), and maternal RSV vaccination services. Emergency department visits and observation unit stays for RSV-related presentations are included.
RSV diagnostic testing equipment, vaccine manufacturing costs, pharmaceutical R&D expenditure, and over-the-counter symptom relief products are excluded. Home nebuliser equipment and telehealth consultation fees for RSV symptom management are outside the scope.
Demand for treatment and preventive interventions against Respiratory Syncytial Virus (RSV) in the USA is rising because RSV continues to cause a high burden of serious respiratory illness - particularly among infants, young children, older adults, and individuals with weakened immune systems or chronic conditions. Every year tens of thousands of children under five are hospitalized due to RSV-related complications. Severe RSV infections also impact older adults: before recent vaccine rollouts, an estimated 123,000 to 193,000 adult hospitalizations occurred annually, with especially high rates in those aged 75 and older. These figures reflect a substantial clinical and public-health need for effective RSV therapies and prevention options.
At the same time, advances in medical science and expanded options for prevention and treatment are fueling greater adoption. The introduction of new vaccines and monoclonal-antibody prophylaxis has broadened the tools available to prevent and manage RSV - this increases demand among pediatricians, geriatric physicians, and caregivers alike. Moreover, growing awareness of RSV’s risks, improved diagnostic capacity, and more comprehensive guidelines for RSV prevention and care are encouraging healthcare providers and families to rely more on RSV-targeted treatment and prevention strategies. As population segments at high risk (infants, older adults, immunocompromised) remain substantial, demand for RSV treatment and prophylaxis in the USA is expected to grow steadily in coming years.
The human respiratory syncytial virus (RSV) treatment market in the USA is segmented by treatment type and treatment provider. Supportive care is the leading treatment type, accounting for 50% of the market share. In terms of treatment provider, hospitals dominate, also holding 50% of the market. The growing incidence of RSV infections, especially among vulnerable populations such as infants, elderly adults, and immunocompromised individuals, is driving the demand for effective treatments and healthcare services.
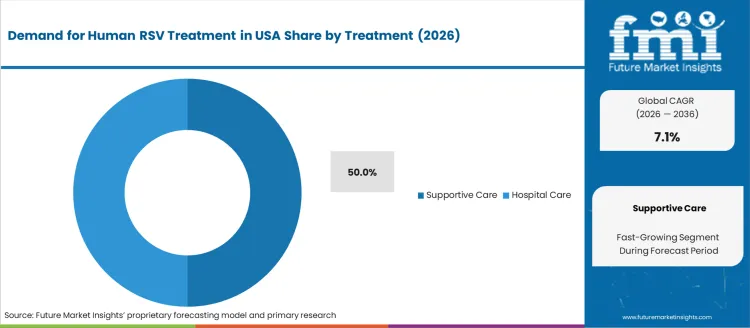
Supportive care is the most common treatment for human RSV, holding 50% of the market share. This approach primarily focuses on relieving symptoms and managing the respiratory distress caused by RSV infections. Supportive care includes oxygen therapy, hydration, and mechanical ventilation when necessary, along with fever management and ensuring proper nutrition. While there is no specific antiviral treatment for RSV, supportive care is crucial for alleviating symptoms and preventing complications, especially in high-risk patients such as young children and the elderly.
The demand for supportive care is expected to remain strong as it remains the cornerstone of managing RSV, particularly for outpatients and those who do not require hospitalization. Hospital care is also important, particularly in severe cases of RSV infection, but supportive care remains the primary treatment for most patients.
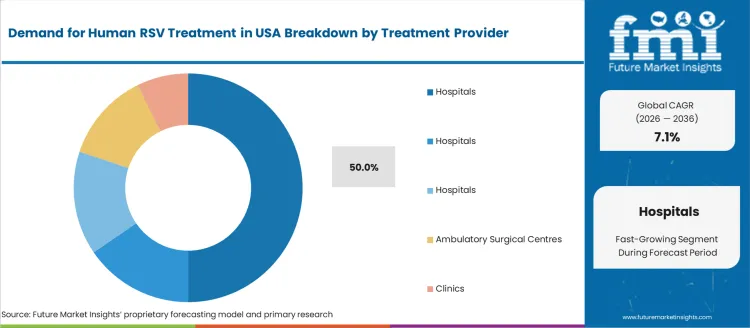
Hospitals are the primary treatment providers for human RSV in the USA, accounting for 50% of the market share. Hospitals, particularly those with more than 200 beds, are equipped to manage severe RSV cases that require intensive care, such as those involving respiratory failure or complications in high-risk populations. The availability of specialized staff, including pediatric and respiratory therapists, along with advanced medical equipment such as ventilators, makes hospitals the primary setting for managing serious RSV infections.
In addition to large hospitals, smaller facilities like those with 200-400 beds and less than 200 beds also contribute significantly to the care of RSV patients. Ambulatory surgical centers and clinics are increasingly providing outpatient care for less severe cases of RSV, focusing on supportive therapies, but hospitals remain the dominant provider due to their capacity to handle more critical situations. The demand for RSV treatments is expected to continue to be high in hospital settings, particularly during RSV seasons, when hospitalizations peak.
The demand for human respiratory syncytial virus (RSV) treatment in the USA is increasing due to the growing awareness of RSV's impact on vulnerable populations, particularly infants, young children, and the elderly. RSV is a leading cause of lower respiratory tract infections and hospitalization in these high-risk groups. As the incidence of RSV infections rises during the colder months, the need for effective treatments and preventive measures continues to grow. Advances in antiviral therapies and monoclonal antibodies for RSV are contributing to better management and treatment outcomes, fueling the demand for such treatments in the USA.
Several factors are driving the demand for human RSV treatment in the USA. First, the increasing incidence of RSV infections, particularly in vulnerable populations such as infants, the elderly, and individuals with compromised immune systems, is driving the need for effective treatments. Second, the growing awareness of RSV's impact on public health and its ability to cause severe respiratory illnesses is increasing the demand for early diagnosis and intervention. Third, advancements in antiviral treatments, vaccines, and monoclonal antibodies are improving the options available for managing and preventing RSV infections, further driving market demand. Additionally, the rise in healthcare access and investments in pediatric and geriatric care are contributing to the increased use of RSV treatments.
Despite growing demand, several factors could limit the widespread adoption of human RSV treatments in the USA. One major restraint is the high cost of newer treatments, particularly monoclonal antibodies and antiviral medications, which can be a financial burden for some patients and healthcare systems. Another challenge is the limited availability of RSV vaccines and treatments for all age groups, particularly in adults who are at higher risk. The seasonal nature of RSV infections may also lead to fluctuating demand, with some healthcare providers not prioritizing RSV treatment outside of peak seasons. Finally, the lack of widespread awareness and screening for RSV, especially in adults, may hinder early detection and timely treatment, limiting the effectiveness of available therapies.
Several key trends are shaping the demand for human RSV treatment in the USA. One significant trend is the increasing focus on preventive treatments, such as monoclonal antibodies and vaccines, which are expected to reduce the severity and incidence of RSV infections. Another trend is the rise in the development of more effective antiviral treatments and therapies tailored to specific patient populations, including high-risk infants and elderly adults.
Additionally, there is growing interest in early diagnosis and rapid intervention, which is driving the demand for diagnostic tools that can quickly identify RSV infections. The expansion of healthcare coverage and access to RSV treatments is also influencing the market, particularly as healthcare providers work to manage RSV-related hospitalizations and reduce associated healthcare costs. Finally, the continued research and investment in RSV therapies and vaccines are likely to lead to more advanced and effective treatment options in the future, further increasing market demand.
The Human Respiratory Syncytial Virus (RSV) Treatment market in the USA is expected to grow steadily, with the West USA leading the way at a projected CAGR of 8.2%. South USA follows closely with a growth rate of 7.3%, while the Northeast USA is expected to grow at 6.6%. The Midwest USA shows moderate growth with a CAGR of 5.7%. RSV, a major cause of respiratory illness in infants, young children, and the elderly, continues to drive demand for effective treatments. As awareness of RSV-related complications grows, the market for treatments is expected to expand across the United States.
.webp)
| Region | CAGR (2026-2036) |
|---|---|
| West USA | 8.2% |
| South USA | 7.3% |
| Northeast USA | 6.6% |
| Midwest USA | 5.7% |
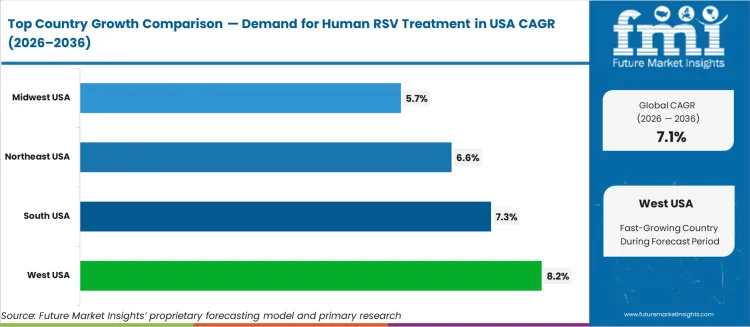
The West USA is projected to experience the highest growth in the Human RSV Treatment market, with a projected CAGR of 8.2%. The region’s large healthcare infrastructure, combined with a significant population of vulnerable individuals, is a key driver of this growth. States like California, Washington, and Oregon have a high rate of RSV infections, particularly among infants, the elderly, and those with weakened immune systems.
The West’s healthcare systems are increasingly focused on providing effective treatments to combat RSV, including antivirals and monoclonal antibody therapies. Additionally, the region’s strong research and development sector, especially in biotechnology and pharmaceutical innovation, is contributing to the availability of new RSV treatments. As the region’s healthcare providers continue to adopt the latest treatment options and educate patients about RSV prevention and treatment, the market for RSV treatments is expected to expand.
In South USA, the demand for Human RSV Treatment is projected to grow at a CAGR of 7.3%. The region’s large and diverse population, combined with the rising incidence of RSV infections, is driving the adoption of effective treatments. States like Texas, Florida, and Georgia are experiencing higher rates of RSV infection, particularly during the peak seasons of respiratory illnesses. As healthcare providers in the South continue to focus on improving patient outcomes and reducing RSV-related complications, there is a growing need for effective antiviral treatments and prevention strategies.
The region’s expanding healthcare infrastructure, coupled with increasing public awareness about RSV risks and the availability of new treatments, is contributing to market growth. The South’s warmer climate, which may influence RSV transmission, further supports the demand for timely and accessible treatment options for those affected by the virus.
In Northeast USA, the Human RSV Treatment market is projected to grow at a CAGR of 6.6%. The region’s high population density and well-established healthcare systems make it a key market for RSV treatments. Cities like New York, Boston, and Philadelphia have large numbers of at-risk populations, including infants, the elderly, and individuals with underlying health conditions, who are more susceptible to severe RSV infections. Healthcare providers in the Northeast are increasingly adopting advanced treatment options, such as monoclonal antibodies and antivirals, to manage RSV infections.
The Northeast also places a strong emphasis on research and public health initiatives focused on respiratory infections, which is contributing to increased awareness and treatment adoption. As RSV continues to pose a significant health threat, the market for effective treatments in the Northeast is expected to grow steadily, driven by both demand for new therapies and ongoing education on prevention.
In Midwest USA, the Human RSV Treatment market is expected to grow at a more moderate rate of 5.7%. The region’s focus on improving healthcare access and outcomes, particularly in rural and underserved areas, is contributing to the demand for RSV treatments. States like Illinois, Michigan, and Ohio are seeing an increasing number of RSV infections, particularly among vulnerable populations, which is driving the need for effective treatments.
The Midwest is also witnessing an expansion in healthcare facilities and infrastructure, which helps to improve access to RSV treatment options. However, the growth rate in the Midwest is slightly slower compared to other regions, reflecting a more gradual shift toward adopting new therapies. As healthcare providers continue to focus on improving diagnosis and treatment of respiratory illnesses like RSV, the market for RSV treatments in the Midwest is expected to continue growing, driven by both improved access to care and heightened awareness of the virus.
Demand for treatment of Respiratory Syncytial Virus (RSV) in the USA remains substantial as RSV continues to cause significant hospitalizations and healthcare burden among infants, older adults, and vulnerable populations. Each year tens of thousands of children, primarily under five years old, are hospitalized due to severe RSV infection. Among older adults and immunocompromised patients, RSV also contributes to respiratory illness, hospital admissions, and increased medical costs. The expanding disease burden, combined with growing awareness and demand for effective therapeutics, fuels the growing need for RSV treatment and prevention options.
On the supply side, leading hospitals and medical centers play a central role in delivering RSV treatment and supportive care. According to your list, major USA institutions such as Children's National Hospital, Cleveland Clinic, Cedars-Sinai Medical Center, Boston Children’s Hospital, and Texas Children’s Hospital are among those involved in RSV care. These centers offer diagnostics, inpatient care, supportive therapies, and increasingly act as hubs for administering prophylactic or therapeutic agents as they become available.
Competition among care providers and institutions is shaped by capacity to manage RSV cases, quality of pediatric and geriatric care, access to advanced treatments or prophylactics, and ability to follow evolving clinical guidelines. Hospitals with strong pediatric care services, ICU facilities, and ability to coordinate early diagnosis and intervention are best positioned to meet demand and provide effective RSV treatment as the overall RSV-related disease burden persists and evolves in the USA.
| Metric | Value |
|---|---|
| Quantitative Units | USD 0.64 to USD 1.28 billion, at a CAGR of 7.1% |
| Market Definition | Human RSV treatment encompasses the clinical management of respiratory syncytial virus infections through supportive care interventions, hospital-based treatment protocols, and prophylactic biologic administration. The USA market covers supportive care (oxygen supplementation, fluid management, respiratory support) and hospital care (ICU admission, mechanical ventilation) delivered across hospitals, ambulatory surgical centres, and clinics. |
| Segmentation | Treatment (Supportive Care, Hospital Care), Treatment Provider (Hospitals, Ambulatory Surgical Centres, Clinics) |
| Regions Covered | West, South, Northeast, Midwest |
| Key Companies Profiled | Children's National Hospital, Cleveland Clinic, Cedars-Sinai Medical Center, Boston Children's Hospital, Texas Children's Hospital |
| Forecast Period | 2026 to 2036 |
| Approach | Forecasting models apply a bottom-up methodology starting with unit shipment and average selling price data, cross-validated against manufacturer financial disclosures and distributor records. |
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with primary research documentation.
How large is the demand for human rsv treatment in usa in 2026?
The demand for human rsv treatment in usa is estimated to be valued at USD 0.64 billion in 2026.
What will be the market size by 2036?
Market size is projected to reach USD 1.28 billion by 2036.
What is the expected growth rate between 2026 and 2036?
Growth is expected at a CAGR of 7.1% between 2026 and 2036.
Which treatment is poised to lead by 2026?
Supportive Care accounts for 50.0% share in 2026.
How is hospitals positioned in 2026?
Hospitals holds 50.0% share of the treatment provider segment in 2026.
What is the growth outlook for West?
West is projected to grow at a CAGR of 8.2% during 2026 to 2036.
What is included in the scope of this report?
Market scope includes oxygen therapy, intravenous fluid management, bronchodilator administration, respiratory monitoring, ICU admissions, prophylactic monoclonal antibody administration (nirsevimab, palivizumab), and maternal RSV vaccination services. Emergency department visits and observation unit stays for RSV-related presentations are included.
How does FMI build and validate this forecast?
Forecasting models apply a bottom-up methodology starting with unit shipment and average selling price data, cross-validated against manufacturer financial disclosures and distributor records.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.