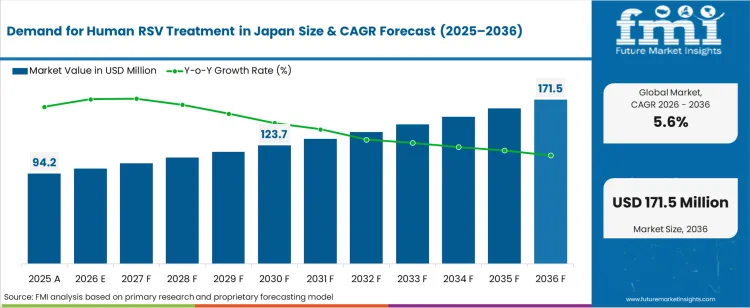
The demand for human rsv treatment in japan was valued at USD 94.20 million in 2025, projected to reach USD 99.48 million in 2026, and is forecast to expand to USD 171.54 million by 2036 at a 5.6% CAGR. Japan's aging population and the seasonal burden of respiratory syncytial virus infections across paediatric and elderly demographics are sustaining institutional demand for RSV treatment services. The approval of new RSV immunisation products by the Pharmaceuticals and Medical Devices Agency is reshaping the treatment landscape, with prophylactic monoclonal antibody therapies reducing hospitalisation rates while shifting cost centres from inpatient supportive care toward outpatient immunisation programmes. Hospital-based supportive care remains the dominant treatment modality at 50.0% share.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 99.48 million |
| Industry Value (2036) | USD 171.54 million |
| CAGR (2026 to 2036) | 5.6% |
Source: Future Market Insights, 2026
The incremental opportunity between 2026 and 2036 amounts to USD 72.06 million. As per FMI, the growth trajectory reflects the dual effect of expanding immunisation coverage reducing severe hospitalisations while increasing per-patient treatment expenditure through high-cost biologic prophylactics. National health insurance reimbursement for RSV monoclonal antibody administration in high-risk infants and elderly patients provides an institutional procurement floor. The emergence of maternal RSV vaccination programmes is expected to generate incremental demand for prenatal immunisation services within the obstetric care pathway.
Sub-national growth patterns reflect paediatric and geriatric population density and hospital infrastructure. Kyushu & Okinawa at 7.0%, Kanto at 6.5%, Kansai at 5.7%, Chubu at 5.0%, Tohoku at 4.4%, Rest of Japan at 4.2%. Kyushu and Okinawa lead due to high birth rates relative to the national average and active prefectural paediatric immunisation programmes. Kanto follows, anchored by the Tokyo metropolitan area's concentration of tertiary paediatric hospitals and neonatal intensive care units. Kansai benefits from Osaka's large elderly population requiring seasonal RSV management, while Chubu and Tohoku reflect incremental growth from rural community hospital capacity expansion.
Human RSV treatment encompasses the clinical management of respiratory syncytial virus infections through supportive care interventions and hospital-based treatment protocols. The Japan market covers supportive care (oxygen supplementation, fluid management, respiratory monitoring) and hospital care (intensive care admission, mechanical ventilation support) delivered across hospitals of varying bed capacities, ambulatory surgical centres, and clinics. Treatment is administered to paediatric patients with acute bronchiolitis and elderly patients with RSV-associated lower respiratory tract infections.
Market scope includes oxygen therapy administration, intravenous fluid management, bronchodilator therapy, respiratory monitoring services, and intensive care unit admissions for RSV-related respiratory failure. Prophylactic monoclonal antibody administration (palivizumab, nirsevimab) and maternal RSV vaccination services are included.
RSV diagnostic testing equipment, vaccine manufacturing, and pharmaceutical drug development costs are excluded. Outpatient over-the-counter symptom management products and home nebuliser equipment are outside the analytical scope.
The demand for human RSV treatment in Japan is closely tied to pediatric care patterns, rapid diagnostics in outpatient clinics, and the growing clinical focus on respiratory disease risk in infants and older adults. Historically, RSV infections were managed largely through supportive care in hospitals, especially for infants with breathing difficulty. As pediatric emergency services expanded and diagnostic testing became routine during seasonal respiratory surges, RSV detection increased across both urban and regional clinics. Japans dense childcare attendance, early school enrollment, and strong surveillance of pediatric infections reinforced steady clinical exposure to RSV each year. In recent years, rising awareness of RSV impact on premature infants, immunocompromised patients, and frail elderly adults has further expanded the treated population beyond early childhood.
Future demand for human RSV treatment in Japan will be shaped by population aging, expansion of preventive strategies, and stronger risk stratification in respiratory medicine. As older adults increasingly present with severe viral pneumonia, RSV is gaining attention alongside influenza and other seasonal infections. Growth will also be supported by wider use of targeted therapies and long acting preventive agents in high risk infants and elderly patients. Barriers include the absence of widespread curative therapies, reliance on hospital based supportive care, and variability in regional access to advanced antiviral options. Long term demand will depend on how effectively Japan integrates early diagnosis, preventive immunization strategies, and outpatient respiratory management into routine care for both pediatric and aging populations.
Demand for human RSV treatment in Japan is shaped by seasonal infection cycles, pediatric hospitalization patterns, and structured respiratory care pathways. Supportive care leads by treatment because most RSV cases rely on oxygen therapy, hydration, and monitoring rather than antiviral intervention. Hospitals dominate by treatment provider due to the need for inpatient respiratory support in high risk infants and elderly patients. Public hospitals, pediatric specialty centers, and regional medical networks drive procurement. Import reliance persists for select respiratory devices and pharmaceuticals. Substitution pressure between outpatient monitoring and inpatient admission remains limited by disease severity thresholds. Predictable winter outbreaks and aging population risk exposure reinforce demand stability.
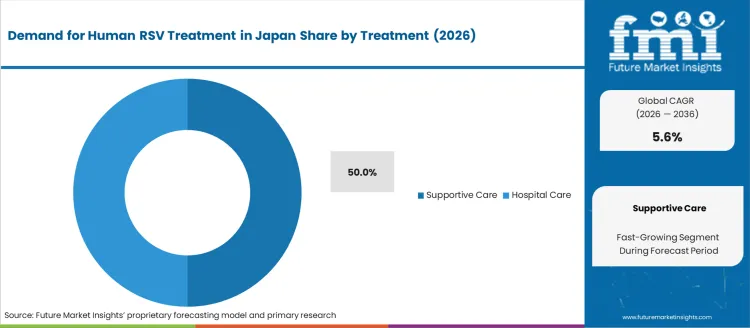
Supportive care accounts for 50% of the demand for human RSV treatment in Japan by treatment, reflecting the absence of widely deployed curative antiviral therapy. Consumption intensity is driven by the need for oxygen support, intravenous fluids, fever management, and continuous respiratory monitoring in infants and older adults. Usage remains stable because clinical guidelines emphasize symptom control and complication prevention. Procurement is led by hospital respiratory wards and pediatric intensive care units. Price sensitivity remains moderate because supportive care relies on standard inpatient resources rather than high cost specialty drugs. Specification control emphasizes oxygen delivery stability, fluid balance accuracy, and continuous pulse oximetry performance.
Supportive care also generates consistent repeat demand during annual RSV peaks that lead to predictable hospitalization surges. Repeat utilization remains high because reinfection risk persists across early childhood and older adult populations. Providers favor standardized care protocols to reduce length of stay and complication rates. Margin structure remains controlled under national health insurance inpatient reimbursement systems. Regulatory exposure centers on infection control compliance and pediatric safety reporting. Import reliance persists for respiratory consumables and monitoring sensors. Substitution pressure from early discharge exists in mild cases, though inpatient monitoring remains essential for oxygen dependent patients nationwide.
Hospitals represent 50.0% of the demand for human RSV treatment in Japan by treatment provider, reflecting the high rate of inpatient care for severe pediatric and geriatric cases. Consumption intensity is driven by oxygen therapy, bronchodilator support, and continuous observation that cannot be delivered in most outpatient settings. Usage remains stable because RSV complications escalate quickly in high risk groups. Procurement is centered on hospitals with pediatric wards, respiratory units, and emergency departments. Price sensitivity remains moderate because hospitalization is reimbursed under standardized national insurance schedules. Specification control emphasizes bed turnover efficiency, isolation room availability, and respiratory support equipment readiness.
Hospitals also generate strong seasonal repeat demand as RSV case numbers rise consistently during winter months. Repeat utilization remains predictable because outbreak timing follows established seasonal patterns. Providers favor centralized admission protocols to manage surge capacity and prevent cross infection. Margin structure remains controlled under fixed inpatient reimbursement frameworks. Regulatory exposure remains elevated due to pediatric care standards and hospital infection prevention mandates. Import reliance persists for ventilatory support components and oxygen delivery devices. Substitution pressure from clinics and ambulatory centers remains limited because advanced respiratory support and overnight monitoring are not routinely available outside hospital environments.
Demand for human RSV treatment in Japan is shaped by strict pediatric hospitalization practices, rising elderly respiratory vulnerability, and strong seasonal infection surveillance. RSV remains a leading cause of infant bronchiolitis and pneumonia admissions, particularly among premature infants and children with cardiac or pulmonary conditions. Japan expanding elderly population also shows rising hospitalization from RSV-related lower respiratory infections. Universal health coverage supports early hospital presentation, which increases clinical detection and treatment activity. Demand is therefore anchored in inpatient respiratory care, seasonal outbreak management, and protection of high-risk age groups rather than outpatient viral self-care trends.
Japan maintains cautious admission standards for infants with respiratory distress, even in moderate RSV cases. Pediatric wards prioritize oxygen support, antiviral intervention where indicated, and close monitoring to prevent rapid progression. Neonatal intensive care units remain key drivers due to vulnerability among low-birth-weight infants. Parents also show low tolerance for home-based monitoring during viral respiratory illness. This conservative admission culture structurally sustains consistent RSV treatment demand across each seasonal wave.
RSV is increasingly recognized in Japan as a serious respiratory threat to elderly patients with chronic lung disease, cardiac disorders, and weakened immunity. Nursing homes and long-term care hospitals report recurrent seasonal clusters. Even mild cases can progress rapidly to pneumonia in advanced age groups. This shifts RSV treatment demand beyond pediatrics into geriatric inpatient medicine, broadening utilization across internal medicine departments and respiratory wards.
Demand for RSV treatment in Japan remains restrained by the limited availability of broadly effective direct antiviral therapies. Most clinical care relies on supportive treatment, oxygen therapy, and infection control rather than targeted viral suppression. Seasonal bed shortages during winter respiratory peaks also restrict admission flexibility. In addition, outpatient confirmation remains limited, concentrating treatment demand inside hospital settings rather than community clinics. These clinical boundaries cap rapid therapy expansion despite recurring seasonal disease burden.
.webp)
| Region | CAGR (%) |
|---|---|
| Kyushu & Okinawa | 7.0% |
| Kanto | 6.5% |
| Kansai | 5.7% |
| Chubu | 5.0% |
| Tohoku | 4.4% |
| Rest of Japan | 4.2% |
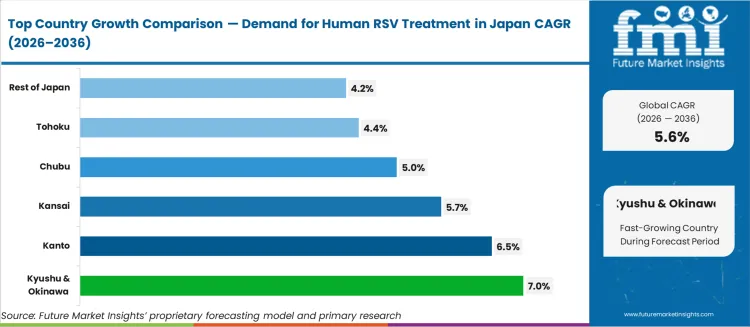
The demand for human RSV treatment in Japan is rising steadily across pediatric and respiratory care settings, led by Kyushu and Okinawa at a 7.0% CAGR. Growth in this region is supported by higher seasonal RSV incidence, strong pediatric hospital coverage, and improving access to antiviral and supportive therapies. Kanto follows at 6.5%, driven by dense urban populations, large tertiary children hospitals, and high diagnostic testing volumes. Kansai records 5.7% growth, reflecting stable demand from university hospitals and regional pediatric care centers. Chubu at 5.0% shows moderate uptake linked to routine respiratory infection management and expanding outpatient pediatric services. Tohoku and the Rest of Japan, at 4.4% and 4.2%, reflect slower growth shaped by lower population density, fewer high volume pediatric centers, and longer care access intervals during peak respiratory infection seasons.
Demand for human RSV treatment in Kyushu and Okinawa is advancing at a CAGR of 7.0% through 2036, supported by rising pediatric respiratory admissions, seasonal infection surges, and strong public hospital dependency for infant care. This region records high treatment volumes during winter outbreaks across neonatal and early childhood wards. Kyushu and Okinawa differ from Kanto through greater reliance on inpatient RSV management rather than outpatient antibody prophylaxis. Demand is sustained by birth rates, regional childcare density, and steady government funded respiratory infection control programs.
Demand for human RSV treatment in Kanto is expanding at a CAGR of 6.5% through 2036, supported by dense pediatric hospital networks, high neonatal intensive care capacity, and wider use of early intervention therapies in Tokyo and surrounding cities. Kanto leads in early diagnosis, antibody administration, and protocol based inpatient RSV care. This region contrasts with Kyushu and Okinawa through higher outpatient treatment penetration and advanced monitoring practices. Demand reflects high urban birth volumes, childcare facility density, and strong seasonal surveillance across metropolitan medical centers.
Demand for human RSV treatment in Kansai is advancing at a CAGR of 5.7% through 2036, supported by stable pediatric population levels, strong university hospital presence, and structured respiratory infection management protocols in Osaka and Kyoto. Kansai shows balanced inpatient and outpatient RSV treatment utilization across teaching hospitals and regional pediatric clinics. This region differs from Kanto through steadier patient flow and lower extreme seasonal surges. Demand is shaped by routine infant respiratory screening, integrated care pathways, and stable regional hospital admissions for viral respiratory conditions.
Demand for human RSV treatment in Chubu is progressing at a CAGR of 5.0% through 2036, supported by steady regional birth rates, workplace linked healthcare access, and consistent pediatric hospital coverage in Aichi. Chubu differs from Kansai through stronger reliance on general hospitals rather than specialist pediatric centers. RSV treatment demand remains concentrated on inpatient care for moderate infections, with selective outpatient management. Growth is stable and shaped by family household density in industrial cities and employer supported healthcare coverage for dependent children.
Demand for human RSV treatment in Tohoku is advancing at a CAGR of 4.4% through 2036, supported by aging caregiver populations, lower birth rates, and stable but limited pediatric hospital expansion. Miyagi serves as the primary treatment hub for RSV across the region. Tohoku contrasts with Kanto through reduced pediatric case turnover and fewer high capacity neonatal units. Treatment demand is centered on essential inpatient care during seasonal peaks, with limited use of newer outpatient antibody therapies.
Demand for human RSV treatment in Rest of Japan is advancing at a CAGR of 4.2% through 2036, supported by small city hospitals, basic pediatric respiratory care access, and steady seasonal infection incidence. These areas differ from Kanto through limited neonatal intensive care availability and delayed outpatient diagnosis in early infection stages. RSV treatment remains focused on inpatient management for moderate to severe cases in regional public hospitals. Growth remains gradual and is guided by demographic decline, local hospital catchment sizes, and public health surveillance reach.
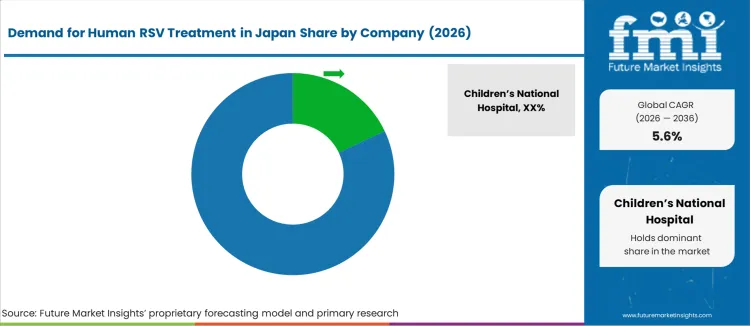
The demand for human RSV treatment in Japan is shaped by pediatric hospitalization rates, rising RSV incidence among older adults, and expanding use of preventive antibodies in high risk infants. Meiji Seika Pharma holds a central domestic role through antiviral and pediatric infectious disease portfolios supplied to children hospitals and regional medical centers. Shionogi supports demand through respiratory focused anti-infective development and surveillance programs linked to public health monitoring. Chugai Pharmaceutical anchors biologic adoption through hospital distribution networks tied to pediatric and adult respiratory care. AstraZeneca Japan plays a key role through long acting antibody based prevention used in neonatal and elderly risk groups. These firms shape treatment access through PMDA approvals, hospital formulary positioning, and national immunization coordination.
Children’s National Hospital, Cleveland Clinic, Cedars Sinai Medical Center, Boston Children’s Hospital, and Texas Children’s Hospital influence RSV care at a clinical research level rather than direct treatment supply in Japan. Their role is limited to published clinical protocols, trial data, and guideline contributions referenced by Japanese academic societies. Prescribing in Japan is governed by Japanese Pediatric Society guidance, inpatient reimbursement rules, and strict cold chain handling for antibody based prevention. Buyer preference favors suppliers with stable domestic manufacturing, pediatric safety data, and Japanese language medical support. Demand visibility tracks seasonal RSV surges, growth in elderly respiratory admissions, expansion of antibody based prevention, and national policy focus on reducing pediatric hospitalization burden across prefectural health systems.
| Metric | Value |
|---|---|
| Quantitative Units | USD 99.48 to USD 171.54 million, at a CAGR of 5.6% |
| Market Definition | Human RSV treatment encompasses the clinical management of respiratory syncytial virus infections through supportive care interventions and hospital-based treatment protocols. The Japan market covers supportive care (oxygen supplementation, fluid management, respiratory monitoring) and hospital care (intensive care admission, mechanical ventilation support) delivered across hospitals of varying bed capacities, ambulatory surgical centres, and clinics. |
| Segmentation | Treatment (Supportive Care, Hospital Care), Treatment Provider (Hospitals, Ambulatory Surgical Centres, Clinics) |
| Regions Covered | Kyushu & Okinawa, Kanto, Kansai, Chubu, Tohoku, Rest of Japan |
| Key Companies Profiled | Children's National Hospital, Cleveland Clinic, Cedars-Sinai Medical Center, Boston Children's Hospital, Texas Children's Hospital |
| Forecast Period | 2026 to 2036 |
| Approach | Forecasting models apply a bottom-up methodology starting with unit shipment and average selling price data, cross-validated against manufacturer financial disclosures and distributor records. |
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with primary research documentation.
How large is the demand for human rsv treatment in japan in 2026?
The demand for human rsv treatment in japan is estimated to be valued at USD 99.48 million in 2026.
What will be the market size by 2036?
Market size is projected to reach USD 171.54 million by 2036.
What is the expected growth rate between 2026 and 2036?
Growth is expected at a CAGR of 5.6% between 2026 and 2036.
Which treatment is poised to lead by 2026?
Supportive Care accounts for 50.0% share in 2026.
How is hospitals positioned in 2026?
Hospitals holds 50.0% share of the treatment provider segment in 2026.
What is the growth outlook for Kyushu & Okinawa?
Kyushu & Okinawa is projected to grow at a CAGR of 7.0% during 2026 to 2036.
What is included in the scope of this report?
Market scope includes oxygen therapy administration, intravenous fluid management, bronchodilator therapy, respiratory monitoring services, and intensive care unit admissions for RSV-related respiratory failure. Prophylactic monoclonal antibody administration (palivizumab, nirsevimab) and maternal RSV vaccination services are included.
How does FMI build and validate this forecast?
Forecasting models apply a bottom-up methodology starting with unit shipment and average selling price data, cross-validated against manufacturer financial disclosures and distributor records.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.