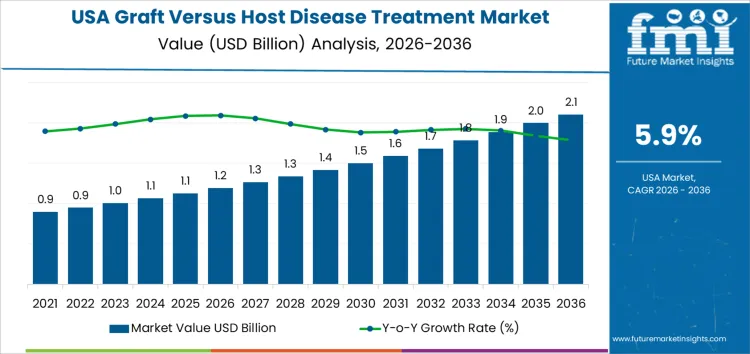
Demand for graft versus host disease treatment in USA is valued at USD 1.2 billion in 2026 and is forecasted to reach USD 2.1 billion by 2036, registering a CAGR of 5.9%. Demand growth is supported by rising hematopoietic stem cell transplant volumes, improved survival rates, and expanded clinical recognition of chronic GVHD management requirements.
Monoclonal antibodies represent the leading product category due to targeted immunomodulatory action, improved efficacy in steroid-refractory cases, and growing adoption in standardized treatment protocols. Increased clinical trial activity and regulatory approvals for biologic therapies strengthen long-term treatment uptake across transplant centers.
West USA, South USA, Northeast USA, and Midwest USA constitute key growth regions, driven by concentration of advanced transplant hospitals, oncology networks, and academic research institutions. Pfizer, Incyte, Novartis, Bristol-Myers Squibb, and Mallinckrodt Pharmaceuticals maintain competitive positioning through diversified GVHD portfolios, ongoing pipeline development, and established distribution across USA specialty care settings.
| Metric | Value |
|---|---|
| USA Graft Versus Host Disease Treatment Sales Value (2026) | USD 1.2 billion |
| USA Graft Versus Host Disease Treatment Forecast Value (2036) | USD 2.1 billion |
| USA Graft Versus Host Disease Treatment Forecast CAGR (2026-2036) | 5.9% |
Demand for graft versus host disease treatment in the United States grows due to increasing volumes of allogeneic stem cell and bone marrow transplants. Advances in hematologic cancer therapy expand transplant eligibility for older patients and individuals with complex disease profiles. Higher transplant activity raises incidence of immune mediated complications requiring structured prevention and treatment. Improved survival after transplantation extends patient follow up periods, which increases long term management of acute and chronic graft versus host disease. Clinical protocols emphasize early diagnosis and sustained therapy to preserve organ function and quality of life.
Expanded use of unrelated and haploidentical donors increases immunologic mismatch risk, driving demand for targeted immunomodulatory treatments. Growth in specialized transplant centers improves access to advanced therapies and multidisciplinary care models. Ongoing clinical research strengthens understanding of inflammatory pathways involved in graft versus host disease, supporting development of novel treatment options. Broader insurance coverage for transplant related complications supports consistent treatment utilization. Outpatient management programs increase reliance on long term pharmacologic control. Patient monitoring through registries and digital health tools improves detection rates and treatment continuity. Expansion of biologic and small molecule therapies increases available options, reinforcing sustained demand across transplant care settings nationwide.
Demand for graft versus host disease treatment in USA is shaped by hematopoietic stem cell transplant volumes, post-transplant complication management, and advances in targeted immunotherapies. Clinicians evaluate treatment efficacy, immunosuppression balance, long-term safety, and compatibility with transplant conditioning regimens. Adoption patterns reflect structured care within transplant centers, supported by protocol-driven therapy selection and continuous monitoring of acute and chronic disease progression.
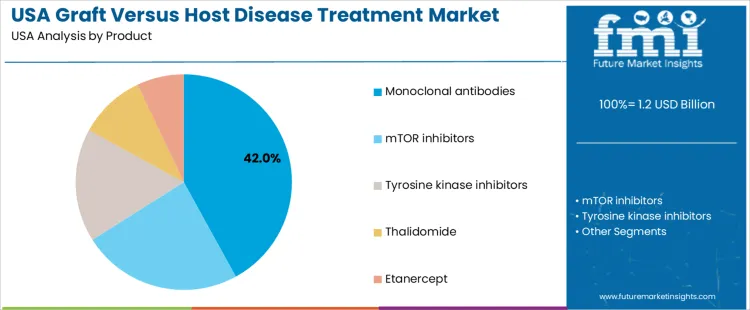
Monoclonal antibodies hold 42.0%, representing the largest product category in USA demand. These therapies target specific immune pathways involved in donor T-cell activation and inflammatory signaling. Their targeted mechanism supports disease control while limiting broad immunosuppression. mTOR inhibitors hold 24.0%, supporting immune regulation and graft tolerance maintenance. Tyrosine kinase inhibitors account for 17.0%, applied in steroid-refractory cases requiring alternative pathway inhibition. Thalidomide holds 10.0%, used in select chronic management scenarios. Etanercept accounts for 7.0%, addressing cytokine-mediated inflammation. Product distribution reflects reliance on targeted biologics within modern treatment protocols.
Key Points
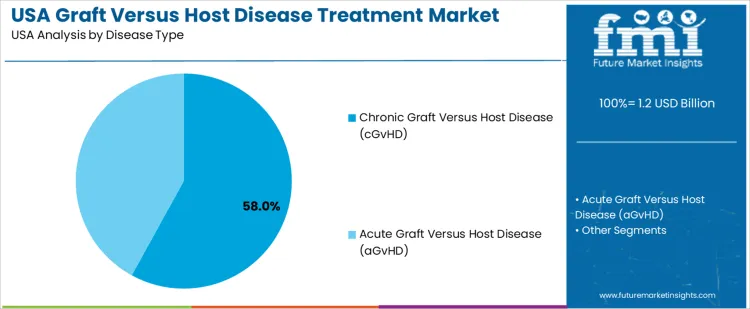
Chronic graft versus host disease holds 58.0%, driving the majority of treatment demand in USA. Chronic forms require prolonged therapy, ongoing symptom management, and long-term immunosuppressive strategies. Treatment focuses on controlling multi-organ involvement and preserving quality of life. Acute graft versus host disease holds 42.0%, requiring intensive short-term intervention following transplantation. Disease-type distribution reflects higher treatment duration and resource utilization associated with chronic disease management.
Key Points
Demand increases as allogeneic stem cell and bone marrow transplant procedures expand across hematology and oncology centers in USA. Graft versus host disease (GVHD) emerges as a key post-transplant complication requiring systemic immunosuppressive therapy and targeted agents. Adoption aligns with advanced transplant programs, enhanced patient survival expectations, and structured long-term follow-up protocols. Usage concentrates in specialized hospitals, transplant centers, and outpatient infusion clinics with multidisciplinary care teams.
USA performs a high volume of allogeneic hematopoietic stem cell transplants for leukemia, lymphoma, and other hematologic disorders. Clinicians employ prophylactic immunosuppressive regimens to reduce acute GVHD risk and initiate treatment promptly upon diagnosis to preserve organ function. Novel targeted therapies, monoclonal antibodies, and small molecules complement corticosteroids and calcineurin inhibitors for chronic and steroid-refractory GVHD. Transplant programs integrate early detection through biomarker monitoring and adopt evidence-based management pathways to improve outcomes. Multidisciplinary coordination among hematologists, immunologists, and supportive care teams reinforces structured therapy adoption and continuity of care.
GVHD therapies often involve high cost and prolonged administration, prompting evaluation under Medicare, Medicaid, and private payer coverage policies. Prior authorization, formulary placement, and cost-sharing influence therapy selection and patient access. Clinical complexity and comorbidity profiles require individualized regimens and close monitoring, increasing outpatient visit frequency and supportive care needs. Side-effect management necessitates additional medical resources, affecting overall care planning. Competition from emerging agents and evolving consensus guidelines shape prescribing patterns. Skilled specialist availability and long-term follow-up infrastructure influence treatment continuity. Demand remains robust due to medical necessity, while growth depends on payer alignment, evidence supporting comparative effectiveness, and integration of advanced GVHD therapies within USA transplant care practice.
Demand for graft versus host disease treatment in the USA is increasing due to rising hematopoietic stem cell transplants, improved survival rates, and long-term complication management. West USA leads with a 6.8% CAGR, supported by transplant center density and early adoption of novel therapies. South USA follows at 6.1%, driven by expanding transplant programs and growing patient volumes. Northeast USA records a 5.4% CAGR, shaped by academic medicine leadership and protocol-driven care. Midwest USA posts 4.7%, reflecting integrated health systems and conservative therapy escalation. Regional variation reflects transplant activity, specialist access, reimbursement discipline, and post-transplant care infrastructure across the USA.
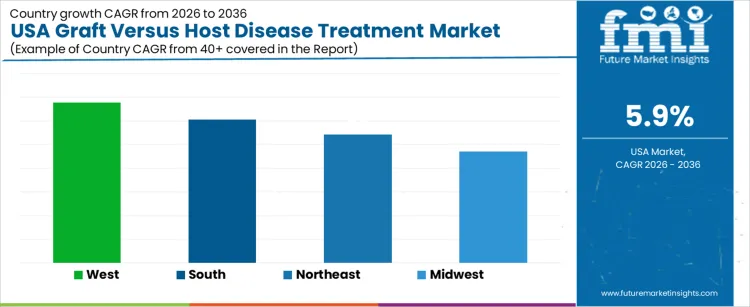
| Region | CAGR (2026 to 2036) |
|---|---|
| West USA | 6.8% |
| South USA | 6.1% |
| Northeast USA | 5.4% |
| Midwest USA | 4.7% |
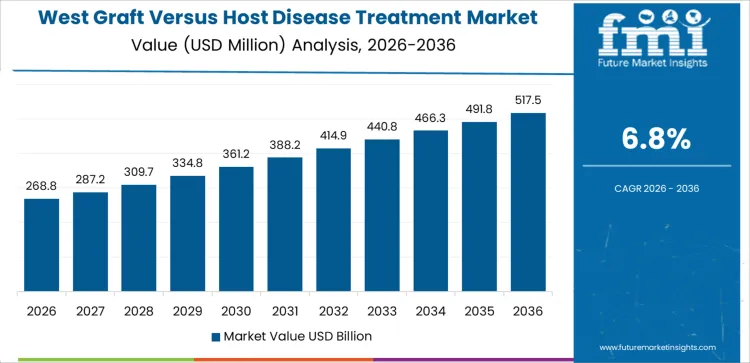
West USA drives demand through concentration of high-volume transplant centers, hematology specialists, and advanced supportive care services. Region’s CAGR of 6.8% reflects frequent management of acute and chronic GVHD following allogeneic stem cell transplantation. Centers emphasize early diagnosis, biomarker monitoring, and timely therapy initiation. Access to clinical trials supports adoption of targeted immunomodulatory treatments. Multidisciplinary teams coordinate long-term outpatient care, addressing skin, gastrointestinal, and hepatic complications. Demand favors therapies with steroid-sparing profiles and strong safety data. Growth remains care-intensity driven, aligned with complex case management and sustained follow-up requirements.
South USA demand is shaped by expanding transplant capacity, demographic growth, and improving access to specialty hematology care. Region’s CAGR of 6.1% reflects increasing numbers of transplant recipients requiring GVHD prevention and treatment. Health systems invest in standardized post-transplant pathways to manage complications efficiently. Community-based oncology centers strengthen referral links with transplant hubs. Clinicians prioritize therapies that reduce hospitalization and improve outpatient management. Demand growth remains volume-driven, supported by rising transplant procedures and broader availability of follow-up care across regional networks.
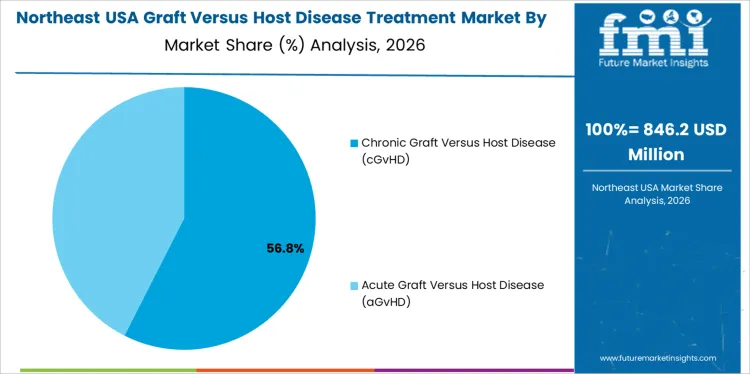
Northeast USA demand reflects leadership of academic medical centers and evidence-based transplant care. Region’s CAGR of 5.4% is supported by structured GVHD management protocols informed by clinical research. Physicians emphasize risk stratification, organ-specific assessment, and stepwise therapy escalation. Access to specialized diagnostics supports precise treatment decisions. Formularies favor therapies with robust clinical validation and reimbursement clarity. Care delivery remains coordinated across inpatient and outpatient settings. Growth aligns with protocol refinement and dissemination rather than rapid expansion of transplant volumes.
Midwest USA demand is anchored in integrated health systems, centralized transplant services, and cost-managed care delivery. Region’s CAGR of 4.7% reflects steady use of GVHD treatments within standardized pathways. Providers emphasize consistency, formulary control, and long-term patient monitoring. Referral patterns concentrate complex cases at regional centers. Therapy adoption progresses cautiously, aligned with clinical necessity and budget considerations. Demand growth remains disciplined and system-driven, reflecting stable transplant activity and focus on sustained graft outcomes rather than aggressive therapy expansion.
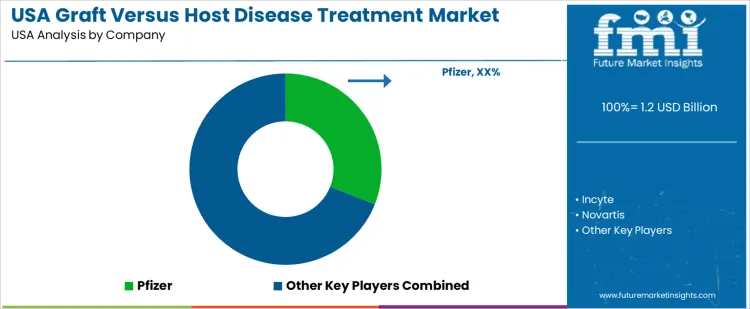
Demand for graft-versus-host disease (GVHD) treatment in the USA is driven by allogeneic hematopoietic stem cell transplantation procedures and increasing survival rates that elevate chronic and acute GVHD incidence. Clinical use spans first-line therapies, steroid-refractory interventions, and symptom management across skin, liver, and gastrointestinal involvements. Buyers evaluate efficacy evidence, safety profiles, formulation stability, and compatibility with existing immunosuppressive regimens. Procurement teams prioritize FDA-approved agents, robust clinical data, and suppliers with comprehensive support for specialty care centers and transplant programs. Trend in the USA market reflects growth in transplant volumes, personalized therapy approaches, and development of targeted treatments that improve quality-of-life and long-term outcomes.
Pfizer holds a leading position in the USA with its JAK inhibitor therapy approved for chronic GVHD, supported by clinical studies demonstrating symptomatic control and steroid-sparing potential. Incyte participates with additional JAK pathway inhibitors offering therapeutic options for patients with refractory or chronic forms of the disease. Novartis supports demand through targeted small molecules and kinase inhibitors employed in clinical practice to manage immune dysregulation in transplant recipients. Bristol-Myers Squibb contributes with immunomodulatory agents and biologics used off-label or in combination regimens addressing GVHD manifestations. Mallinckrodt Pharmaceuticals maintains visibility with therapeutic products used in inflammation control and supportive care for GVHD patients. Competitive positioning in the USA reflects regulatory alignment, clinical evidence strength, patient support infrastructure, and integration with transplant center protocols optimizing treatment sequencing and outcomes.
| Items | Details |
|---|---|
| Quantitative Units | USD billion |
| Product | Monoclonal antibodies; mTOR inhibitors; Tyrosine kinase inhibitors; Thalidomide; Etanercept |
| Disease Type | Chronic Graft Versus Host Disease (cGvHD); Acute Graft Versus Host Disease (aGvHD) |
| Regions Covered | West USA; South USA; Northeast USA; Midwest USA |
| Key Companies Profiled | Pfizer; Incyte; Novartis; Bristol-Myers Squibb; Mallinckrodt Pharmaceuticals |
| Additional Attributes | Demand is driven by rising hematopoietic stem cell transplant volumes, higher survival rates, and expanded use of targeted immunomodulatory therapies. Chronic GvHD accounts for the larger treatment share, while biologics and kinase inhibitors dominate prescribing patterns across major US transplant centers. |
How big is the demand for graft versus host disease treatment in USA in 2026?
The demand for graft versus host disease treatment in USA is estimated to be valued at USD 1.2 billion in 2026.
What will be the size of graft versus host disease treatment in USA in 2036?
The market size for the graft versus host disease treatment in USA is projected to reach USD 2.1 billion by 2036.
How much will be the demand for graft versus host disease treatment in USA growth between 2026 and 2036?
The demand for graft versus host disease treatment in USA is expected to grow at a 5.9% CAGR between 2026 and 2036.
What are the key product types in the graft versus host disease treatment in USA?
The key product types in graft versus host disease treatment in USA are monoclonal antibodies, mtor inhibitors, tyrosine kinase inhibitors, thalidomide and etanercept.
Which disease type segment is expected to contribute significant share in the graft versus host disease treatment in USA in 2026?
In terms of disease type, chronic graft versus host disease (cgvhd) segment is expected to command 58.0% share in the graft versus host disease treatment in USA in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.