Rare Inflammatory Disease Treatment Market : Global Industry Analysis 2015 - 2024 and Opportunity Assessment 2025 - 2035
Rare Inflammatory Disease Treatment Market Size and Share Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 - 2024 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2025 - 2035
Rare Inflammatory Disease Treatment Market Size and Share Forecast Outlook 2025 to 2035
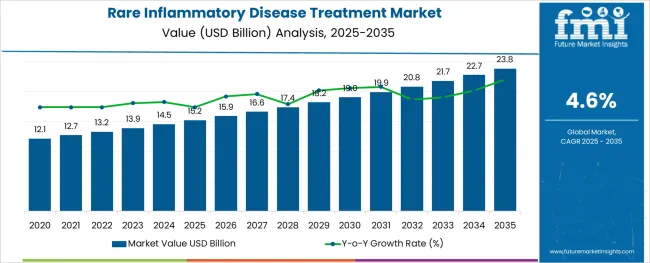
The Rare Inflammatory Disease Treatment Market is estimated to be valued at USD 15.2 billion in 2025 and is projected to reach USD 23.8 billion by 2035, registering a compound annual growth rate (CAGR) of 4.6% over the forecast period.
Quick Stats for Rare Inflammatory Disease Treatment Market
- Industry Value (2025): USD 15.2 billion
- Forecast Value (2035): USD 23.8 billion
- Forecast CAGR: 4.6%
- Leading Segment in 2025: RID Treatment for Ulcerative Colitis (38.2%)
- Key Growth Region: North America, Asia-Pacific, Europe
- Top Key Players: Pfizer, Inc., Novartis AG, Allergan Plc, Abbvie, Inc., Valeant Pharmaceuticals International, Inc., Abbott Laboratories, Johnson & Johnson, Regeneron Pharmaceuticals, Inc., Mallinckrodt Plc, Swedish Orphan Biovitrum AB
Rationale for Segmental Growth in the Rare Inflammatory Disease Treatment Market
Market Overview
The rare inflammatory disease treatment market is growing steadily, driven by increasing recognition of rare inflammatory conditions and advances in therapeutic options. The rising prevalence of complex diseases such as ulcerative colitis has led to greater demand for specialized treatments that address underlying inflammation. Healthcare providers have prioritized improving patient outcomes through innovative therapies that target specific disease pathways.
Biologics have emerged as a critical treatment class due to their precision and efficacy in managing rare inflammatory diseases. The introduction of injectable formulations has enhanced patient compliance and treatment delivery.
Furthermore, increased investment in research and development, along with enhanced diagnostic capabilities, has expanded treatment accessibility. The market outlook remains positive as new therapies continue to enter clinical practice and healthcare systems emphasize personalized medicine approaches.
Segmental Analysis
The market is segmented by Indication, Drug Class, Mode of Administration, and Distribution Channel and region. By Indication, the market is divided into RID Treatment for Ulcerative Colitis, RID Treatment Juvenile Rheumatoid Arthritis, RID Treatment Psoriatic Arthritis, and RID Treatment Other Indications.
In terms of Drug Class, the market is classified into Biologics-based RID Treatment and Organic Compounds-based RID Treatment. Based on Mode of Administration, the market is segmented into RID Injectables, Oral RID Treatment, and Other RID Treatment Administration Modes.
By Distribution Channel, the market is divided into RID Treatment through Hospital Pharmacies, RID Treatment through Retail Pharmacies, and RID Treatment through Online Pharmacies. Regionally, the market is classified into North America, Latin America, Western Europe, Eastern Europe, Balkan & Baltic Countries, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific, and the Middle East & Africa.
Insights into the Indication Segment: RID Treatment for Ulcerative Colitis
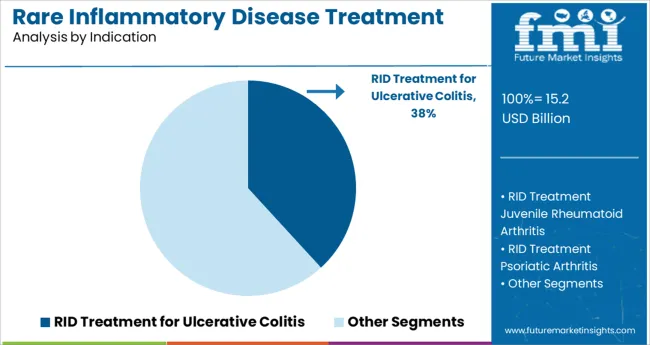
The RID treatment for ulcerative colitis segment is projected to hold 38.2% of the market revenue in 2025, making it the leading indication. Growth in this segment has been driven by the chronic nature and increasing diagnosis rates of ulcerative colitis, a condition that requires sustained management to reduce flare-ups and complications. The complexity of symptoms and the need for long-term control have positioned ulcerative colitis treatments as a key focus in rare inflammatory disease management.
Healthcare providers have adopted a combination of therapeutic strategies to manage inflammation and maintain remission. This has led to increased use of advanced therapies tailored specifically for this indication.
The segment’s prominence is expected to continue as awareness and screening efforts improve.
Insights into the Drug Class Segment: Biologics-based RID Treatment
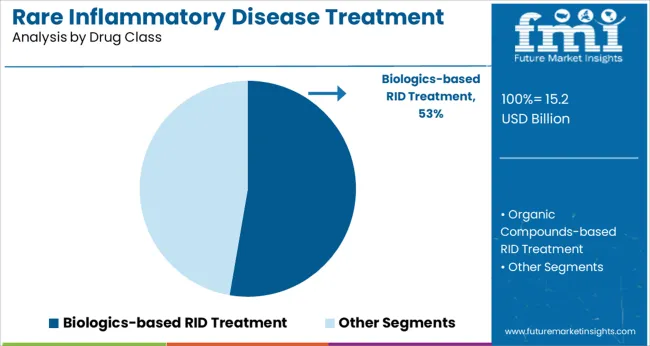
The biologics-based RID treatment segment is forecasted to account for 52.7% of the market revenue in 2025, maintaining its leadership among drug classes. This segment’s expansion has been driven by the high specificity and effectiveness of biologics in targeting inflammatory pathways. Biologics offer advantages in reducing disease activity and improving patient quality of life, especially in refractory cases where conventional therapies fail.
Treatment regimens have increasingly incorporated biologics due to their ability to modulate immune responses with fewer systemic side effects. The development of biosimilars and novel biologic agents has further broadened patient access.
As personalized medicine continues to gain traction, biologics are expected to remain central to rare inflammatory disease treatment strategies.
Insights into the Mode of Administration Segment: RID Injectables
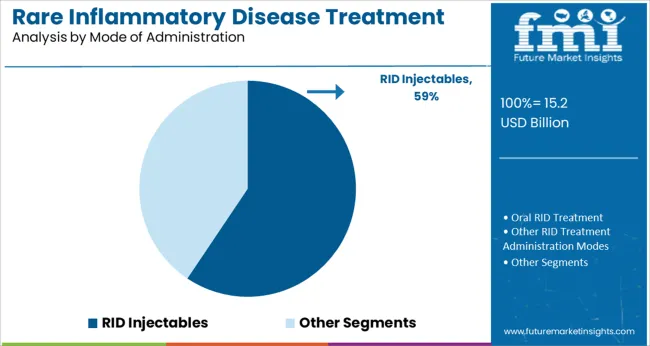
The RID injectables segment is projected to hold 59.4% of the market revenue in 2025, establishing it as the dominant mode of administration. Growth in this segment has been driven by the convenience and efficacy of injectable therapies in delivering targeted treatment with controlled dosing. Injectables enable rapid onset of action and have been preferred in clinical settings for their ability to maintain consistent therapeutic levels.
Patient adherence has benefited from advances in injection device technology and formulations that reduce dosing frequency.
The ability to administer treatment in outpatient or home settings has also contributed to the segment’s expansion. As more injectable biologics enter the market, the RID injectables segment is expected to maintain its strong position.
How The Market Progressed Till June 2025?
| Particulars | Details |
|---|---|
| H1, 2024 | 8.35% |
| H1, 2025 Projected | 8.82% |
| H1, 2025 Outlook | 8.52% |
| BPS Change - H1, 2025 (O) - H1, 2025 (P) | (-) 30 ↓ |
| BPS Change - H1, 2025 (O) - H1, 2024 | (+) 17 ↑ |
A comparative analysis related to the market growth rates and development prospects in the global rare inflammatory disease treatment market is presented by Future Market Insights. The market is highly influenced by the onerous regulatory environment and innovation factors for orphan drugs under the impact of macro and industry factors.
According to Future Market Insights analysis, the global rare inflammatory disease treatment market expects to surge by 17 Basis Point Share (BPS) in H1-2025 as compared to H1- 2024. Whereas in comparison to H1-2025 projected value, the market during H1-2025 outlook period will show a dip of 30 BPS.
The market change in the BPS is associated with the extensive process of orphan drug approval by regulatory bodies for licensure and distribution. Further, revision in molecular portfolio for existing products also face a backlash in terms of patent renewal, owed to the emergence of several clinically viable alternatives to existing solutions.
This has caused a reduction in the BPS value. Additionally, the growth of the biopharmaceutical industry upon the advent of the COVID-19 pandemic has provided a boost in the BPS value change for the market.
The market is subject to activities such as supporting the treatment of rare diseases through collaborative activities, revenue fluidity which arises from product Medicaid rebates, chargebacks due to other government programs, and existing patented product portfolio.
Industry Analysis of Rare Inflammatory Disease Treatment (2020 to 2024) Vs Market Estimates (2025 to 2035)
From 2013 to 2024, treatment for rare inflammatory diseases experienced an increase of 3.8% in value CAGR terms. Various distribution channels such as hospital pharmacies, retail pharmacies, and online pharmacies have observed high demand for treating several rare inflammatory diseases. Recent outbreaks of infectious diseases, such as COVID-19, have increased market demand to a major extent.
During the height of the pandemic crisis, clinicians, doctors and other healthcare stakeholders extensively relied on anti-inflammatory drugs to ease complications experienced by severely infected and immunocompromised patients. Drugs such as ibuprofen, for instance, was cleared by the National Health Service in the UK to be used as a nonsteroidal anti-inflammatory drug (NSAID) to inhibit receptors responsible for triggering an inflammatory response to the coronavirus.
Companies have been considering in their production processes ways to improve the treatment of rare inflammatory diseases, such as eosinophilic granulomatosis with polyangiitis (EGPA), a rare disorder that causes vasculitis. One such treatment is Nucala, a medication for patients aged 6 years or older with relapsing-remitting or refractory EGPA. Likewise, several orphan drugs are coming into the limelight for numerous rare inflammatory diseases. Overall, demand for rare inflammatory disease treatment is anticipated to rise at a CAGR of around 4.6% through 2035.
How is the Rising Incidence of Inflammatory & Autoimmune Diseases Generating Opportunities for New Therapies?
Healthcare experts have associated cytokine IL-23 with treatment of inflammatory and autoimmune diseases. Researchers have identified a fundamental molecular mechanism essential to inflammatory and autoimmune diseases such as multiple sclerosis, rheumatoid arthritis and psoriasis. The action of pro-inflammatory cytokine IL-23, depends on the organizational stimulation of its receptor, namely IL-23R.
Moreover, the occurrence of inflammatory and autoimmune diseases is increasing at a rapid pace. According to the World Health Organization, psoriasis affects approximately 100 million people globally, while between 1.3% and 34.7% develop chronic and inflammatory arthritis. Likewise, inflammatory bowel infections such as ulcerative colitis and Crohn’s disease are increasing at an alarming rate in formerly unaffected fragments of the world.
Hence, medical practitioners are introducing several drug candidates to address some of the most debilitating rare inflammatory diseases. In 2020, for instance, the European Medicines Agency's Committee for Medicinal Products for Human Use (CHMP) granted an extension of indication to Kineret (anakinra) for treating Still's disease. Kineret is an immunosuppressive medicine which inhibits chemical messenger activity of interleukin-1, an agent which is produced in excess amongst patients suffering from rheumatoid arthritis and juvenile idiopathic arthritis.
Region-wise Analysis
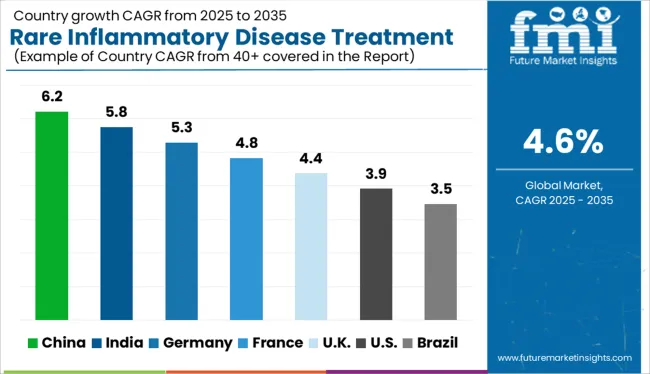
How is Discovery of New Add-On Treatments Facilitating Expansion in Europe?
D of new treatments and advancements in medical technology have driven market demand for rare inflammatory disease treatment throughout Europe. With increasing investments in open-source drug delivery systems and evaluations of their effectiveness in treating rare diseases, more treatment options are becoming available for rare inflammatory diseases.
Recently, in September 20245.2, the European Medicines Agency has recommended granting an extension of indication to Nucala (mepolizumab) as an add-on treatment for patients aged 6 years and older with relapsing-remitting or refractory eosinophilic granulomatosis with polyangiitis (EGPA), responsible for inflaming vital organs such as the lungs, heart, skin, nervous system and gastrointestinal tract.
It is estimated that between 0.15.28 and 4 new cases per 15.2 million are diagnosed each year with EGPA. The drug is already approved for treating patients of the same age group for eosinophilic asthma. According to FMI, rare inflammatory disease treatment in Europe is like to accumulate a market share of 24% in 2025.
High Prevalence Rate amongst Adults & Children Broadening Prospects in North America?
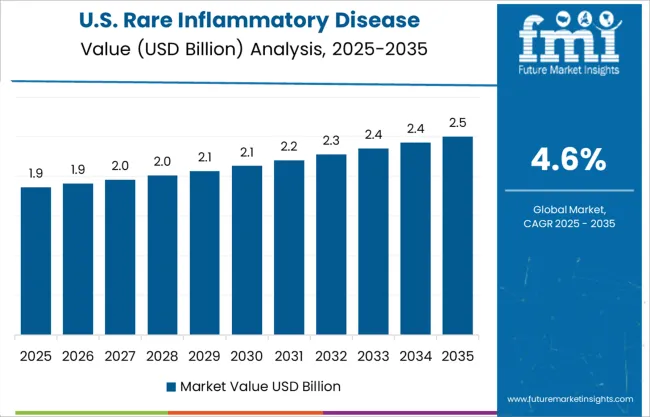
FMI forecasts the rare inflammatory disease treatment industry in North America to accumulate a market share of 39.9% in 2025. The presence of a well-developed health care system and several reimbursement programs aimed at facilitating access to treatment for those with rare illnesses are among the chief growth drivers.
More than 30 million Americans are afflicted with rare diseases. Every year, approximately 6,200 Americans are affected diseases, including Polycythemia Vera, while juvenile rheumatoid arthritis is one of the most common types of arthritic diseases that children are diagnosed with in the United States.
Demand for rare inflammatory disease treatment has risen through various distribution channels including hospital pharmacies, retail pharmacies, and online pharmacies. As a result, the USA is expected to account for the largest market share in the market for rare inflammatory diseases treatment.
Rare Inflammatory Disease Treatment Market - Category-wise Analysis
Will Ulcerative Colitis Spearhead Rare Inflammatory Disease Treatment Demand?
Due to the significant cost savings associated with ulcerative colitis treatments, the producers will be influenced to increase their manufacturing capacities for advanced drugs and therapies in the future.
Technological advancements and the increasing number of patients suffering from ulcerative colitis have contributed to the development of advanced treatments for ulcerative colitis. The influx of new therapeutics methods, demand for enhancing drugs, and advances in treating ulcerative colitis have propelled market demand for rare inflammatory diseases treatments.
Why are Retail Pharmacies Emerging as the Primary POC for Availing Rare Inflammatory Disease Treatment Drugs?
The growing prevalence of diseases and demand for 24/7 medication sales have catalyzed the global market demand for drugs at retail pharmacies. Future growth in artificial intelligence, advanced analytics, and other technologies will help drive the retail pharmacy market in the global market.
Health and wellness services are the key drivers of customer satisfaction at brick-and-mortar pharmacies. These services score 66 points higher than other types of services. Pharmacies with these services are present in 86% of chain pharmacies, 83% of supermarkets, and 75% of mass merchandisers.
Rare Inflammatory Disease Treatment Market - Competitive Landscape
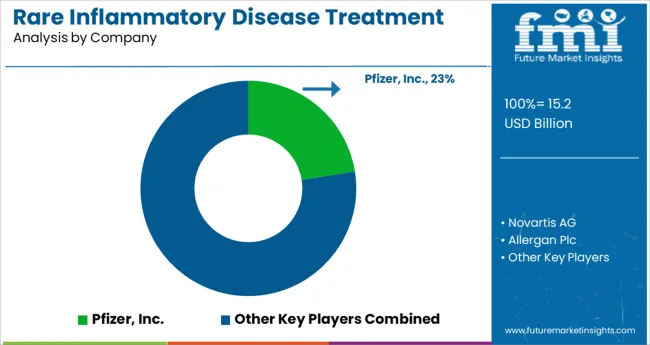
Prominent treatment providers are emphasizing on advancing drug and treatment offerings by initiating a series of clinical studies as well as forging collaborative and acquisition agreements with existing pharmaceutical companies.
- In December 2024, Pfizer Inc. announced that it had reached a definitive agreement to acquire Arena Pharmaceuticals Inc., which is developing innovative possible treatments for a variety of immuno-inflammatory diseases. The proposed acquisition offers best-in-class treatment to address unmet clinical needs for more patients with immuno-inflammatory diseases
- Likewise, in November 2024, Regeneron Pharmaceuticals Inc. announced additional positive results from a Phase 3 trial run jointly with NIAID on the use of single dose of investigational REGEN-COV® 1,200 mg sub-cutaneous injections. The findings revealed that the drug successfully protects healthy individuals from contracting COVID-19 with a success rate of 81.6%
Rare Inflammatory Disease Treatment Report Scope
| Attribute | Details |
|---|---|
| Forecast Period | 2025 to 2035 |
| Historical Data Available for | 2020 to 2024 |
| Market Analysis | million for Value |
| Key Regions Covered | North America; Latin America; Europe; Asia Pacific; Middle East & Africa (MEA) |
| Key Countries Covered | USA, Canada, Brazil, Mexico, Germany, UK, France, Spain, Italy, China, Japan, India, Malaysia, Singapore, Australia, New Zealand, Israel, South Africa, GCC |
| Key Market Segments Covered | Indication, Drug Class, Mode of Administration, Distribution Channel |
| Key Companies Profiled | Novartis AG; Abbvie Inc.; Abbott Laboratories; Regeneron Pharmaceuticals Inc.; Swedish Orphan Biovitrum AB; Pfizer, Inc.; Valeant Pharmaceutical International Inc. (Bausch Health); Mallinckrodt Plc.; Johnson & Johnson |
| Pricing | Available upon Request |
Key Segments Covered in Rare Inflammatory Disease Treatment Industry Survey
Rare Inflammatory Disease Treatment by Indication
- RID Treatment for Ulcerative Colitis
- RID Treatment Juvenile Rheumatoid Arthritis
- RID Treatment Psoriatic Arthritis
- RID Treatment Other Indications
Rare Inflammatory Disease Treatment by Drug Class
- Biologics-based RID Treatment
- Organic Compounds-based RID Treatment
Rare Inflammatory Disease Treatment by Mode of Administration
- RID Injectables
- Oral RID Treatment
- Other RID Treatment Administration Modes
Rare Inflammatory Disease Treatment by Distribution Channel
- RID Treatment through Hospital Pharmacies
- RID Treatment through Retail Pharmacies
- RID Treatment through Online Pharmacies
Rare Inflammatory Disease Treatment Market by Region
- North America RID Treatment Market
- Latin America RID Treatment Market
- Europe RID Treatment Market
- Asia Pacific RID Treatment Market
- Middle East & Africa (MEA)
Frequently Asked Questions
How big is the rare inflammatory disease treatment market in 2025?
The global rare inflammatory disease treatment market is estimated to be valued at USD 15.2 billion in 2025.
What will be the market size for the rare inflammatory disease treatment market in 2035?
It is projected to reach USD 23.8 billion by 2035.
How much will be the CAGR of the rare inflammatory disease treatment market between 2025 and 2035?
The market is expected to grow at a 4.6% CAGR between 2025 and 2035.
What are the key product types in the rare inflammatory disease treatment market?
The key product types are rid treatment for ulcerative colitis, rid treatment juvenile rheumatoid arthritis, rid treatment psoriatic arthritis and rid treatment other indications.
Which biologics-based rid treatment segment is expected to dominate the rare inflammatory disease treatment market in 2025?
biologics-based rid treatment segment is expected to dominate with a 52.7% industry share in 2025.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size Value (USD Million) Analysis, 2020 to 2024
- Current and Future Market Size Value (USD Million) Projections, 2025 to 2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2020 to 2024 and Forecast 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Indication
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Indication , 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Indication , 2025 to 2035
- RID Treatment for Ulcerative Colitis
- RID Treatment Juvenile Rheumatoid Arthritis
- RID Treatment Psoriatic Arthritis
- RID Treatment Other Indications
- Y-o-Y Growth Trend Analysis By Indication , 2020 to 2024
- Absolute $ Opportunity Analysis By Indication , 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Drug Class
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Drug Class, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Drug Class, 2025 to 2035
- Biologics-based RID Treatment
- Organic Compounds-based RID Treatment
- Y-o-Y Growth Trend Analysis By Drug Class, 2020 to 2024
- Absolute $ Opportunity Analysis By Drug Class, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Mode of Administration
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Mode of Administration, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Mode of Administration, 2025 to 2035
- RID Injectables
- Oral RID Treatment
- Other RID Treatment Administration Modes
- Y-o-Y Growth Trend Analysis By Mode of Administration, 2020 to 2024
- Absolute $ Opportunity Analysis By Mode of Administration, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2025 to 2035
- RID Treatment through Hospital Pharmacies
- RID Treatment through Retail Pharmacies
- RID Treatment through Online Pharmacies
- Y-o-Y Growth Trend Analysis By Distribution Channel, 2020 to 2024
- Absolute $ Opportunity Analysis By Distribution Channel, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2020 to 2024
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2025 to 2035
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- USA
- Canada
- Mexico
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- Key Takeaways
- Latin America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- Key Takeaways
- Western Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- Key Takeaways
- Eastern Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- Key Takeaways
- East Asia Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- China
- Japan
- South Korea
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- France
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- India
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- China
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Indication
- By Drug Class
- By Mode of Administration
- By Distribution Channel
- Competition Analysis
- Competition Deep Dive
- Pfizer, Inc.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Novartis AG
- Allergan Plc
- Abbvie, Inc.
- Valeant Pharmaceuticals International, Inc.
- Abbott Laboratories
- Johnson & Johnson
- Regeneron Pharmaceuticals, Inc.
- Mallinckrodt Plc
- Swedish Orphan Biovitrum AB
- Pfizer, Inc.
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2020-2035
- Table 2: North America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 3: Latin America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 4: Western Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 5: Eastern Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 6: East Asia Market Value (USD Million) Forecast by Country, 2020-2035
- Table 7: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2020-2035
- Table 8: Middle East & Africa Market Value (USD Million) Forecast by Country, 2020-2035
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2020-2035
- Figure 3: Global Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 4: Global Market Y-o-Y Growth Comparison by Region, 2025-2035
- Figure 5: Global Market Attractiveness Analysis by Region
- Figure 6: North America Market Incremental $ Opportunity, 2025-2035
- Figure 7: Latin America Market Incremental $ Opportunity, 2025-2035
- Figure 8: Western Europe Market Incremental $ Opportunity, 2025-2035
- Figure 9: Eastern Europe Market Incremental $ Opportunity, 2025-2035
- Figure 10: East Asia Market Incremental $ Opportunity, 2025-2035
- Figure 11: South Asia and Pacific Market Incremental $ Opportunity, 2025-2035
- Figure 12: Middle East & Africa Market Incremental $ Opportunity, 2025-2035
- Figure 13: North America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 14: Latin America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 15: Western Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 16: Eastern Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 17: East Asia Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 18: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 19: Middle East & Africa Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 20: Global Market - Tier Structure Analysis
- Figure 21: Global Market - Company Share Analysis

