Rare NRG1 Fusion Market : Global Industry Analysis 2015 - 2024 and Opportunity Assessment 2025 - 2035
Rare NRG1 Fusion Market Size and Share Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 - 2024 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2025 - 2035
Rare NRG1 Fusion Market Size and Share Forecast Outlook 2025 to 2035
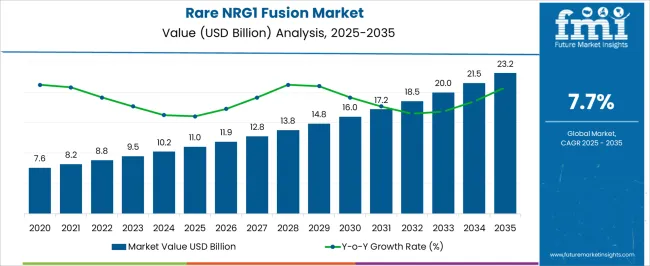
The Rare NRG1 Fusion Market is estimated to be valued at USD 11.0 billion in 2025 and is projected to reach USD 23.2 billion by 2035, registering a compound annual growth rate (CAGR) of 7.7% over the forecast period.
Quick Stats for Rare NRG1 Fusion Market
- Rare NRG1 Fusion Market Industry Value (2025): USD 11.0 billion
- Rare NRG1 Fusion Market Forecast Value (2035): USD 23.2 billion
- Rare NRG1 Fusion Market Forecast CAGR: 7.7%
- Leading Segment in Rare NRG1 Fusion Market in 2025: Immunotherapy (47.5%)
- Key Growth Region in Rare NRG1 Fusion Market: North America, Asia-Pacific, Europe
- Top Key Players in Rare NRG1 Fusion Market: Merus, Other Companies:, Elevation Oncology, Hummingbird Bioscience, Boehringer Ingelheim, CANbridge Pharmaceuticals, Salubris Biotherapeutics, AVEO Oncology, Rain Therapeutics, Eli Lilly and Company, Jiangsu Hengrui Medicine
| Metric | Value |
|---|---|
| Rare NRG1 Fusion Market Estimated Value in (2025 E) | USD 11.0 billion |
| Rare NRG1 Fusion Market Forecast Value in (2035 F) | USD 23.2 billion |
| Forecast CAGR (2025 to 2035) | 7.7% |
Rationale for Segmental Growth in the Rare NRG1 Fusion Market
The rare NRG1 fusion market is undergoing notable evolution as advancements in molecular diagnostics and targeted therapies reshape treatment paradigms. Growing awareness of gene fusions in oncogenic pathways and the introduction of precision medicine have significantly expanded the potential patient pool for novel treatments.
Immunotherapy has emerged as a transformative option, supported by clinical evidence indicating superior outcomes in specific cohorts. Continued investments in genomiC testing infrastructure and heightened clinical trial activity are expected to accelerate innovation and broaden accessibility.
Regulatory support for orphan drug development and increasing collaborations between biopharma and diagnostic companies are paving the way for more personalized treatment options. Future growth opportunities are anticipated to arise from enhanced biomarker-driven patient selection, improved combination regimens, and further integration of next generation sequencing in routine clinical practice.
Segmental Analysis
The market is segmented by Treatment and region. By Treatment, the market is divided into Immunotherapy, Chemotherapy, and Radiation Therapy. Regionally, the market is classified into North America, Latin America, Western Europe, Eastern Europe, Balkan & Baltic Countries, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific, and the Middle East & Africa.
Insights into the Immunotherapy Treatment Segment
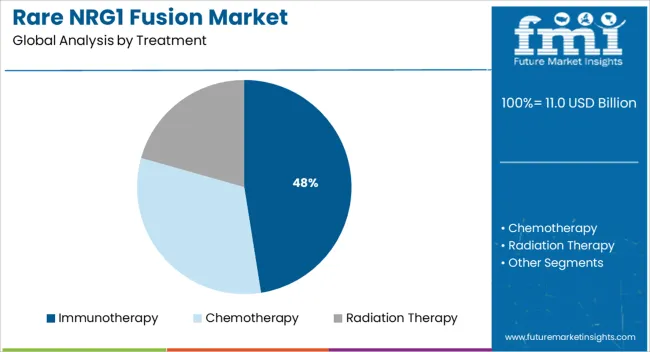
When segmented by treatment, immunotherapy is expected to account for 47.5 % of the rare NRG1 fusion market revenue in 2025, establishing itself as the leading treatment segment. This leadership is attributed to its ability to elicit durable responses by leveraging the body’s immune system to target tumors harboring NRG1 fusions.
Advances in immune checkpoint inhibitors and personalized neoantigen vaccines have improved clinical outcomes in patients who were previously refractory to conventional therapies. The segment’s growth has been reinforced by robust clinical pipelines, expanded regulatory approvals, and growing physician confidence in managing immune related adverse events.
Moreover, ongoing trials exploring synergistic effects of immunotherapy with targeted agents and chemotherapies have strengthened its adoption in both first line and subsequent lines of treatment. The convergence of scientific innovation, favorable safety profiles, and increasing inclusion in treatment guidelines has cemented immunotherapy as the preferred modality within this market.
2020 to 2024 Rare NRG1 Fusion Demand Analysis vs. Forecast 2025 to 2035
The Rare NRG1 fusion market was valued at USD 7.6 Billion in 23.218 while growing at a CAGR of 7.6.7.61%. Technological advancements in the diagnosis and treatment of various cancers have opened up new prospects for the Rare NRG1 Fusion market which are augmenting market growth.
Further, Standard diagnostic procedures are aiding in market expansion. Considering these new advancements in Oncology, the market for Rare NRG1 Fusion is set to achieve a valuation of USD 23.2 billion with a CAGR of 7.73% by the end of 23.233.
Which are Some Prominent Drivers of the Rare NRG1 Fusion Market?
Standard diagnostic protocols to fuel the market expansion
The dynamics of the Rare NRG1 Fusion Market are anticipated to revolutionize in the coming decade owing to the occurrence of such fusions in approximately 0.2% of solid tumors and other invasive tumors. Rare NRG1 Fusion is nothing short of a potential agent for a tumor-agnostic development approach.
Further, the Rare NRG1 Fusion Market is still in its innate state with only a handful of pharmaceutical companies operating in the domain, laying forward immense opportunities for other pharma companies to exploit which will augment the market growth.
Also, standard diagnostic protocols are anticipated to act as a major driver in the steady, smooth growth of the Rare NRG1 Fusion Market size. Additionally, a Rise in the number of research projects for the development of innovative tools and solutions that encourage early detection of tumour-causing mutations is expected to influence substantial growth of the Rare NRG1 Fusion market over the forecast period.
What are the Challenges Projected to be Faced by the Market?
Difficulty in conducting clinical trials is holding back growth
The rarity of fusions undoubtedly in itself is a challenge that makes it difficult for researchers to study and conduct clinical trials, and the diversity of NRG1 fusions makes it difficult to diagnose or detect. These concerns are creating challenges for the market expansion of Rare NRG1 Fusion.
The surge in the use of rebuilt diagnostic imaging restricts the market growth
Due to high costs and budgetary constraints, many hospitals in developing nations are not able to invest in diagnostic imaging procedures. However, due to the high demand for diagnostic imaging procedures in these countries, the hospitals that cannot afford to purchase new and advanced imaging systems, prefer to use rebuilt imaging systems. Thus, this factor is a huge challenge for the Rare NRG1 Fusion market growth during the forecast period.
Region-Wise Insights
The advent of novel cancer diagnosis methods to fuel the market in Europe
Europe is the fastest-growing region in the Rare NRG1 Fusion market. The increasing incidence of cancer cases in Europe clearly demands early detection and diagnosis of cancer. Researchers are consistently working to discover novel cancer diagnosis methods with minimal invasion. There are approximately 375000 cases of cancers detected each year, with a mortality rate of over 44.1%.
On the other hand, the increasing current health expenditure per capita in the European Union, which according to the World Bank, reached USD 3476.43 (in current USD) in the year 2020, up from USD 2636.35 in the year 2006, is projected to aid in the growth of Europe RARE NRG1 FUSION market. Besides this, the growing GDP per capita in the European Union is also anticipated to display significant growth opportunities for the Europe RARE NRG1 FUSION market during the forecast period.
Increasing geriatric population opening up new avenues in Japan
The Japan Rare NRG1 Fusion Market is predicted to witness substantial expansion during the forecast period, i.e., 2025 to 2035. The growth of the market can majorly be attributed to the growing geriatric population (aged 65 years or over) in the nation. This growing geriatric population would be in need of advanced healthcare services, which is anticipated to drive the growth of the market in the coming years.
On the other hand, the Japan Rare NRG1 Fusion Market is also poised to grow on account of the numerous opportunities generated by the rising healthcare expenditure by the government of the nation.
Category wise Insights
By treatment type, Radiation therapy dominates the segment
Radiation therapy is the most widely used intervention in the Rare NRG1 Fusion market with a segment share of 45%. This segment is expected to grow at a high pace compared to immunotherapy and chemotherapy owing to its high effectiveness and use in primary stages of cancer in killing cancer cells.
Start-ups in Rare NRG1 Fusion Market
- Founded in 2014, ‘Freenome’ is a San Francisco, US-based provider of deep learning platforms for detecting circulating cancer-related markers. It is developing a platform for detecting circulating cancer-related markers in patient blood samples. Its proprietary technology uses deep learning to detect the entire range of cell-free DNA, immune cell fragments, and metabolic markers in the blood. It allows physicians to classify tumor types and develop treatment therapies. The company claims that its test performs better than the standard-of-care for four types of cancer: prostate, breast, colorectal, and lung cancer. It received funding of USD 1.1 Billion from Roche, Perceptive Advisors, RA Capital Management, and 50 other investors.
- Founded in 2012, ‘Lightpoint Medical’ is a UK-based developing an intraoperative imaging technology called Cerenkov Luminescence Imaging (CLI). The device has the potential to detect cancer in real-time during surgery, and thereby reduce the likelihood of cancer recurrence and the need for reoperation. It enables optical detection of Positron Emission Tomography (PET) radiopharmaceuticals, thereby combining the advantages of optical imaging (namely, very low cost and small size) with the strengths of PET imaging. It received funding of USD 10.2 million from Oxford technology, Gov. uk Horizon 2024, and 76 other investors.
Market Competition
Key players in the Rare NRG1 Fusion Market are Elevation Oncology, Merus, Hummingbird Bioscience, Boehringer Ingelheim, CANbridge Pharmaceuticals, Salubris Biotherapeutics, AVEO Oncology, Rain Therapeutics, Eli Lilly and Company, Jiangsu Hengrui Medicine.
Some recent developments in the market are:
- In November 2024 - GE Healthcare collaborated with GenesisCare to develop patient outcomes for the two major health burdens worldwide, heart and cancer disease. GE Healthcare would offer MRI, CT, SPECT, PET/CT, digital mammography, and ultrasound equipment to GenesisCare’s 440+ cardiovascular and cancer disease treatment centers across Australia, the USA, Spain, and the UK.
- In January 2024- Roche Diagnostics collaborated with Illumina to offer broad access to clinical oncology next-generation sequencing
Report Scope
| Report Attribute | Details |
|---|---|
| Market Value in 2025 | USD 11.0 billion |
| Market Value in 2035 | USD 23.2 billion |
| Growth Rate | CAGR of 7.73% from 2025 to 2035 |
| Base Year for Estimation | 2025 |
| Historical Data | 2020 to 2025 |
| Forecast Period | 2025 to 2035 |
| Quantitative Units | Revenue in USD Billion and CAGR from 2025 to 2035 |
| Report Coverage | Revenue Forecast, Volume Forecast, Company Ranking, Competitive Landscape, Growth Factors, Trends, and Pricing Analysis |
| Segments Covered | Treatment, Region |
| Regions Covered | North America; Latin America; Europe; Asia Pacific; Middle East & Africa |
| Key Countries Profiled | USA, Canada, Brazil, Mexico, Mexico, Germany, UK, France, Spain, Italy, Russia, China, Japan, South Korea, India, Australia, South Africa, Saudi Arabia, UAE, Israel |
| Key Companies Profiled | Elevation Oncology; Merus; Hummingbird Bioscience; Boehringer Ingelheim; CANbridge Pharmaceuticals; Salubris Biotherapeutics; AVEO Oncology; Rain Therapeutics; Eli Lilly and Company; Jiangsu Hengrui Medicine |
| Customization | Available Upon Request |
Key Segments Profiled in the Rare NRG1 Fusion Market Industry Survey
By Treatment:
- Immunotherapy
- Chemotherapy
- Radiation Therapy
By Region:
- North America
- Latin America
- Europe
- Asia Pacific
- Middle East & Africa
Frequently Asked Questions
How big is the rare nrg1 fusion market in 2025?
The global rare nrg1 fusion market is estimated to be valued at USD 11.0 billion in 2025.
What will be the size of rare nrg1 fusion market in 2035?
The market size for the rare nrg1 fusion market is projected to reach USD 23.2 billion by 2035.
How much will be the rare nrg1 fusion market growth between 2025 and 2035?
The rare nrg1 fusion market is expected to grow at a 7.7% CAGR between 2025 and 2035.
What are the key product types in the rare nrg1 fusion market?
The key product types in rare nrg1 fusion market are immunotherapy, chemotherapy and radiation therapy.
Which segment to contribute significant share in the rare nrg1 fusion market in 2025?
In terms of , segment to command 0.0% share in the rare nrg1 fusion market in 2025.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size Value (USD Million) Analysis, 2020 to 2024
- Current and Future Market Size Value (USD Million) Projections, 2025 to 2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2020 to 2024 and Forecast 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Treatment
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Treatment , 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Treatment , 2025 to 2035
- Immunotherapy
- Chemotherapy
- Radiation Therapy
- Y-o-Y Growth Trend Analysis By Treatment , 2020 to 2024
- Absolute $ Opportunity Analysis By Treatment , 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2020 to 2024
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2025 to 2035
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- USA
- Canada
- Mexico
- By Treatment
- By Country
- Market Attractiveness Analysis
- By Country
- By Treatment
- Key Takeaways
- Latin America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Treatment
- By Country
- Market Attractiveness Analysis
- By Country
- By Treatment
- Key Takeaways
- Western Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Treatment
- By Country
- Market Attractiveness Analysis
- By Country
- By Treatment
- Key Takeaways
- Eastern Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Treatment
- By Country
- Market Attractiveness Analysis
- By Country
- By Treatment
- Key Takeaways
- East Asia Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- China
- Japan
- South Korea
- By Treatment
- By Country
- Market Attractiveness Analysis
- By Country
- By Treatment
- Key Takeaways
- South Asia and Pacific Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Treatment
- By Country
- Market Attractiveness Analysis
- By Country
- By Treatment
- Key Takeaways
- Middle East & Africa Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Treatment
- By Country
- Market Attractiveness Analysis
- By Country
- By Treatment
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2024
- By Treatment
- Canada
- Pricing Analysis
- Market Share Analysis, 2024
- By Treatment
- Mexico
- Pricing Analysis
- Market Share Analysis, 2024
- By Treatment
- Brazil
- Pricing Analysis
- Market Share Analysis, 2024
- By Treatment
- Chile
- Pricing Analysis
- Market Share Analysis, 2024
- By Treatment
- Germany
- Pricing Analysis
- Market Share Analysis, 2024
- By Treatment
- UK
- Pricing Analysis
- Market Share Analysis, 2024
- By Treatment
- Italy
- Pricing Analysis
- Market Share Analysis, 2024
- By Treatment
- Spain
- Pricing Analysis
- Market Share Analysis, 2024
- By Treatment
- France
- Pricing Analysis
- Market Share Analysis, 2024
- By Treatment
- India
- Pricing Analysis
- Market Share Analysis, 2024
- By Treatment
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2024
- By Treatment
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2024
- By Treatment
- China
- Pricing Analysis
- Market Share Analysis, 2024
- By Treatment
- Japan
- Pricing Analysis
- Market Share Analysis, 2024
- By Treatment
- South Korea
- Pricing Analysis
- Market Share Analysis, 2024
- By Treatment
- Russia
- Pricing Analysis
- Market Share Analysis, 2024
- By Treatment
- Poland
- Pricing Analysis
- Market Share Analysis, 2024
- By Treatment
- Hungary
- Pricing Analysis
- Market Share Analysis, 2024
- By Treatment
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2024
- By Treatment
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2024
- By Treatment
- South Africa
- Pricing Analysis
- Market Share Analysis, 2024
- By Treatment
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Treatment
- Competition Analysis
- Competition Deep Dive
- Merus
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Other Companies:
- Elevation Oncology
- Hummingbird Bioscience
- Boehringer Ingelheim
- CANbridge Pharmaceuticals
- Salubris Biotherapeutics
- AVEO Oncology
- Rain Therapeutics
- Eli Lilly and Company
- Jiangsu Hengrui Medicine
- Merus
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2020-2035
- Table 2: Global Market Value (USD Million) Forecast by Treatment , 2020-2035
- Table 3: North America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 4: North America Market Value (USD Million) Forecast by Treatment , 2020-2035
- Table 5: Latin America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 6: Latin America Market Value (USD Million) Forecast by Treatment , 2020-2035
- Table 7: Western Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 8: Western Europe Market Value (USD Million) Forecast by Treatment , 2020-2035
- Table 9: Eastern Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 10: Eastern Europe Market Value (USD Million) Forecast by Treatment , 2020-2035
- Table 11: East Asia Market Value (USD Million) Forecast by Country, 2020-2035
- Table 12: East Asia Market Value (USD Million) Forecast by Treatment , 2020-2035
- Table 13: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2020-2035
- Table 14: South Asia and Pacific Market Value (USD Million) Forecast by Treatment , 2020-2035
- Table 15: Middle East & Africa Market Value (USD Million) Forecast by Country, 2020-2035
- Table 16: Middle East & Africa Market Value (USD Million) Forecast by Treatment , 2020-2035
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2020-2035
- Figure 3: Global Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 4: Global Market Y-o-Y Growth Comparison by Region, 2025-2035
- Figure 5: Global Market Attractiveness Analysis by Region
- Figure 6: North America Market Incremental Dollar Opportunity, 2025-2035
- Figure 7: Latin America Market Incremental Dollar Opportunity, 2025-2035
- Figure 8: Western Europe Market Incremental Dollar Opportunity, 2025-2035
- Figure 9: Eastern Europe Market Incremental Dollar Opportunity, 2025-2035
- Figure 10: East Asia Market Incremental Dollar Opportunity, 2025-2035
- Figure 11: South Asia and Pacific Market Incremental Dollar Opportunity, 2025-2035
- Figure 12: Middle East & Africa Market Incremental Dollar Opportunity, 2025-2035
- Figure 13: North America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 14: Latin America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 15: Western Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 16: Eastern Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 17: East Asia Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 18: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 19: Middle East & Africa Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 20: Global Market - Tier Structure Analysis
- Figure 21: Global Market - Company Share Analysis

