The EGFR-mutated lung cancer treatment market opens 2026 at USD 16.1 billion and is expected to reach USD 39.2 billion by 2036 at a 9.30% CAGR. Adoption is driven by patient genetic profiling, treatment line, and response monitoring rather than promotional visibility. Oncologists select therapies based on mutation type, progression risk, and drug tolerability. Once incorporated into institutional protocols, switching requires clinical justification and regulatory approval. Hospitals and specialty oncology centers monitor therapy adherence, infusion schedules, and adverse event management. Procurement teams focus on supply reliability, lot tracking, and cold chain integrity. Care pathway planning ensures timely initiation and continuity.
Market growth is shaped by incidence trends, genetic testing penetration, and access to targeted therapies rather than broad advertising campaigns. Manufacturers coordinate formulation consistency, vial stability, and packaging to maintain efficacy across treatment cycles. Release involves potency confirmation, sterility testing, and documentation of lot history. Distribution centers stage inventory near high-volume oncology centers to prevent therapy interruptions. Clinical education and monitoring support physician confidence. Profitability follows patient adherence, predictable reorder patterns, and secure supply chains rather than aggressive pricing or frequent formulation updates. Program expansion aligns with adoption of biomarker testing and guideline updates.
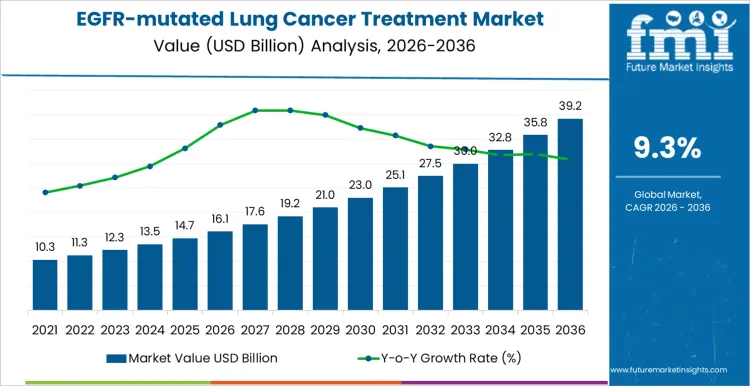
Therapy adoption and clinical guideline alignment drive growth in the EGFR-mutated lung cancer treatment market more than overall lung cancer incidence. In 2026, at approximately USD 16.1 billion, use is concentrated in oncology hospitals, specialized cancer centers, and molecular diagnostic-enabled clinics where treatment selection depends on confirmed EGFR mutation status. Demand is closely tied to biomarker testing capacity, early-stage diagnosis programs, and access to targeted therapies. Expansion occurs as more treatment centers implement first-line targeted regimens, incorporate combination therapy protocols, and widen patient coverage under insurance frameworks. Growth is progressive, reflecting gradual integration of precision medicine pathways rather than sudden surges in patient volume.
Market penetration and patient management frameworks define the later phase of the EGFR-mutated lung cancer treatment market. As value approaches roughly USD 39.2 billion by 2036, stakeholders focus on therapy sequencing, adherence monitoring, and integration of companion diagnostics to optimize outcomes. Pharmaceutical providers that offer bundled support including reimbursement assistance, training for clinical staff, and patient education gain preference among hospital networks. The climb from mid-teens into the mid-forty billion reflects incremental expansion across oncology networks, wider adoption of molecular profiling, and the entry of next-generation EGFR inhibitors, rather than a single breakthrough event. Higher rates of reflex testing and standardized panels are widening access to lung cancer molecular biomarker profiling across community oncology settings. Companies that combine reliable drug supply, clinical support services, and flexible patient access solutions are positioned to maintain long-term contracts across multiple treatment centers.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 16.1 billion |
| Forecast Value (2036) | USD 39.2 billion |
| Forecast CAGR 2026 to 2036 | 9.3% |
EGFR-mutated lung cancer treatments focus on therapies that inhibit specific epidermal growth factor receptor pathways driving tumor growth. Conventional chemotherapy often produced broad cytotoxic effects, limited efficacy, and significant adverse events in this population. Targeted therapies, including first-, second-, and third-generation tyrosine kinase inhibitors, offer mutation-specific intervention, improved progression-free survival, and a better safety profile. Clinical committees often compare new EGFR options against the wider tyrosine kinase inhibitors class to benchmark safety monitoring and resistance expectations. Oncologists select therapies based on mutation subtype, prior treatment history, and resistance mechanisms. Adoption is concentrated in academic hospitals, specialty oncology centers, and molecular testing facilities where rapid genotyping and access to precision medicine influence treatment decisions. Many centers operationalize EGFR pathways within broader non-small cell lung carcinoma (NSCLC) protocols to streamline diagnosis-to-treatment timelines. Clinical outcomes, including tumor response, resistance development, and patient quality of life, directly shape therapy algorithms, protocol design, and follow-up scheduling.
Pharmaceutical manufacturers differentiate products through potency against specific EGFR variants, central nervous system penetration, dosing flexibility, and safety monitoring tools rather than cost per unit alone. Earlier treatment relied on empiric regimens with limited personalization, restricting efficacy and increasing adverse events. Current programs emphasize companion diagnostics, standardized monitoring, and adherence support to optimize patient response. Formulary teams increasingly require linked companion diagnostics evidence to justify first-line targeted selection and reimbursement continuity. Healthcare institutions assess total value through reduced hospitalization, delayed disease progression, and improved functional outcomes, not drug price alone. Support services such as mutation testing, patient education, and adherence monitoring enhance uptake. Market expansion is linked to broader molecular testing, increasing lung cancer incidence, and the integration of targeted therapies into first-line care, providing opportunities for suppliers that offer comprehensive treatment solutions spanning diagnostics and therapeutics.
Treatment selection in the EGFR-mutated Lung Cancer Market is primarily influenced by mutation-specific efficacy, patient safety, and adherence to clinical guidelines rather than short-term hospital procurement cycles. Healthcare providers prioritize therapies that offer superior progression-free survival, tolerable side effect profiles, and integration into treatment protocols. Segment adoption varies by generation of tyrosine kinase inhibitors (TKIs) and combination regimens. Suppliers providing dosing guidance, patient support programs, and reliable drug supply chains can secure long-term adoption. Market growth is concentrated in 1st- and 2nd-line therapy settings, creating predictable demand for manufacturers capable of aligning therapeutic efficacy with clinical support services and regulatory compliance.
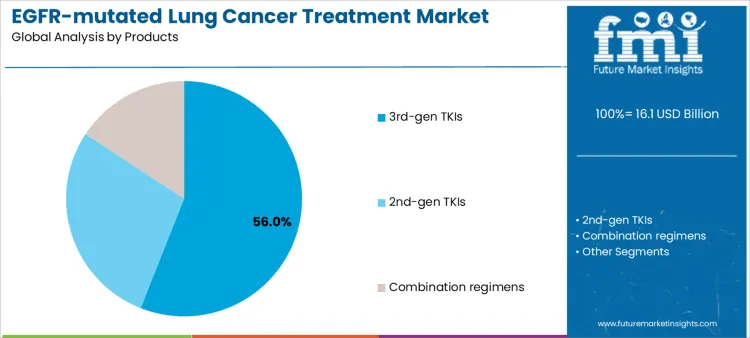
Third-generation TKIs represent approximately 56% of demand due to their efficacy against resistant mutations and ability to be used in first-line therapy. Suppliers supporting these therapies must ensure cold-chain integrity, regulatory documentation, and clinical training for oncology staff. Second-generation TKIs are utilized for patients with specific mutation profiles or as follow-up therapy, requiring supplier guidance on sequencing, monitoring, and patient adherence. Combination regimens address complex or refractory cases, increasing supplier involvement in administration guidance, adverse event management, and integration into clinical protocols. Product choice defines supplier engagement in dosing support, patient management programs, and inventory planning for sustained adoption.
Therapeutic characteristics also shape operational planning and workflow integration. Third-generation TKIs require consistent availability, staff education on mutation-specific dosing, and patient monitoring support, creating recurring supplier touchpoints. Second-generation TKIs necessitate guidance on patient selection, adherence monitoring, and outcome tracking. Combination regimens demand additional training, coordination of multiple therapies, and monitoring of drug-drug interactions. Hospitals that standardize on specific product types rely on supplier support for training, supply reliability, and adverse event management. Providers offering comprehensive support programs, patient education, and consumable logistics can maximize adoption and retention across treatment lines.
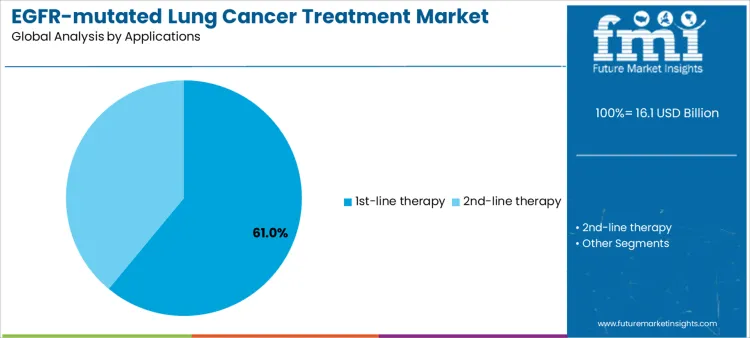
First-line therapy accounts for approximately 61% of market demand due to the high prevalence of newly diagnosed EGFR-mutated lung cancer patients and the need for effective initial management to improve survival outcomes. Second-line therapy, though smaller in volume, involves resistant or progressed cases, creating opportunities for high-value, specialized products and supplier support programs. Application-specific demand informs inventory planning, staff training, and patient support initiatives. Aligning product availability and support services with first- and second-line treatment needs enables suppliers to optimize market penetration, revenue stability, and clinical outcomes.
Treatment stage requirements also influence supplier engagement and service strategy. High-volume first-line programs prioritize consistent drug supply, structured patient support, and clinical education to ensure proper use and adherence. Second-line programs demand technical guidance for sequencing, monitoring for resistance mutations, and management of adverse events. Smaller or combination therapy applications require specialized support, training, and high-touch service. Segment demand is therefore determined by therapy stage, patient population, and clinical complexity rather than short-term procurement trends. Suppliers offering reliable supply, education, and technical support can secure long-term partnerships and maximize adoption.
The EGFR-mutated Lung Cancer Treatment Market is shaped by hospitals and oncology centers managing targeted therapy for patients with EGFR gene mutations. Treatments are evaluated based on response rate, progression-free survival, safety profile, and administration convenience. Selection decisions involve oncologists, pharmacy teams, and hospital procurement. Suppliers that provide therapies with consistent efficacy, robust clinical data, and clear dosing guidance are preferred. This positions treatments as essential components of precision oncology rather than optional interventions, emphasizing clinical reliability, adherence, and patient outcomes.
Healthcare providers increasingly follow evidence-based treatment guidelines to ensure consistent patient outcomes and reduce adverse events. This drives demand in the EGFR-mutated Lung Cancer Treatment Market toward therapies that integrate with standard care protocols, provide predictable response, and offer manageable side effect profiles. Buyers prefer drugs supported by clinical studies, treatment algorithms, and monitoring guidance. Suppliers are evaluated on product consistency, regulatory compliance, and clinical support rather than minor formulation differences. Treatments aligned with standardized protocols are more likely to be adopted across multiple oncology centers.
Recurring demand comes from hospitals, cancer centers, and specialty clinics managing ongoing therapy for patients with confirmed EGFR mutations. Each treatment cycle generates predictable repeat orders. The EGFR-mutated Lung Cancer Treatment Market also benefits from multi-site hospital networks standardizing on approved therapy regimens. Suppliers that provide patient support programs, dosing tools, and reliable product supply secure long-term engagement. Embedding treatments into standard oncology care pathways ensures ongoing demand tied to patient volume and therapy duration.
Targeted therapies carry high per-patient costs and require careful monitoring for adverse events, which can limit adoption in smaller or low-volume centers. The EGFR-mutated Lung Cancer Treatment Market also faces adoption constraints from regulatory approvals, insurance coverage, and hospital formulary processes. Clinicians are cautious about switching therapies mid-course due to patient response and safety concerns. Suppliers entering the market must provide clinical guidance, training, and consistent product availability. Adoption is concentrated in high-volume oncology centers and specialized treatment programs where precision and patient outcomes are critical.
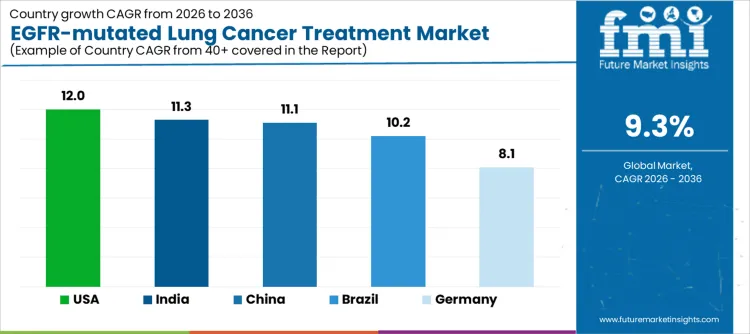
| Country | CAGR |
|---|---|
| USA | 12.0% |
| Brazil | 10.2% |
| China | 11.1% |
| India | 11.3% |
| Germany | 8.1% |
Demand for EGFR mutated lung cancer treatment is rising as hospitals, oncology centers, and specialty clinics expand targeted therapy programs, molecular diagnostics, and personalized treatment regimens. India leads with an 11.3% CAGR, supported by rising lung cancer diagnostics incidence, increasing access to targeted therapies, and growing adoption of precision oncology diagnostics. China follows at 11.1%, driven by expanding cancer treatment infrastructure, higher patient volumes, and integration of molecular testing in standard care. Brazil records 10.2%, reflecting growing access to advanced therapies in private and public hospitals. The USA grows at 12.0%, shaped by adoption of next generation EGFR inhibitors and expanded clinical programs. Germany, at 8.1%, reflects steady demand in a mature healthcare system with established oncology protocols.
United States demand for EGFR-mutated lung cancer treatments is projected to grow at a 12% CAGR as hospital oncology programs expand targeted therapy protocols and molecular testing capabilities. Procurement and therapy adoption are guided by oncology committees, pharmacy directors, and multidisciplinary tumor boards. Adoption emphasizes targeted EGFR inhibitors, personalized treatment regimens, and adherence monitoring. Once selected, therapies are integrated across multiple hospitals and outpatient oncology centers. Clinical trials, guideline adherence, and insurance coverage influence adoption. Import formulations dominate high specification regimens, while domestic suppliers provide standard targeted therapies. Capital allocation aligns with new program launches and patient volume expansion. Replacement cycles follow treatment duration and protocol updates. Commercial success depends on inclusion in hospital formularies, network therapy programs, and supplier support agreements.
Brazil is experiencing a 10.2% CAGR in EGFR-mutated lung cancer treatments due to expansion of private hospital oncology units and specialty lung cancer programs. Procurement authority resides with network pharmacy committees, oncology service directors, and hospital purchasing offices. Adoption focuses on EGFR inhibitor therapies, molecular testing, and outpatient treatment monitoring. Import formulations dominate high specification regimens, while domestic suppliers cover standard therapies. Once validated, therapies are deployed across multiple affiliated hospitals. Training programs for oncologists and clinical staff, as well as adherence monitoring, influence utilization. Capital allocation aligns with hospital network expansion and patient volumes. Replacement cycles follow treatment protocols. Commercial success depends on inclusion in network formularies, reference site adoption, and long-term supply agreements rather than single clinic or department purchases.
China’s EGFR-mutated lung cancer treatment demand is growing at an 11.1% CAGR due to expansion of tertiary hospitals, oncology centers, and molecular testing programs. Large hospital networks control procurement. Adoption emphasizes EGFR inhibitor therapy access, precision dosing, and patient follow-up programs. Domestic suppliers provide standard targeted therapies, while international brands retain positions in advanced regimens. Tender cycles, provincial health programs, and multi-year capital planning govern procurement. Once approved, therapies are administered across multiple hospitals and oncology departments. Physician training, guideline adherence, and patient monitoring influence adoption. Replacement cycles follow treatment duration and therapy updates. Capital allocation aligns with hospital expansion and oncology program growth. Commercial success depends on inclusion in provincial and national formularies rather than single clinic or hospital purchases.
India is witnessing an 11.3% CAGR in EGFR-mutated lung cancer treatments as hospital oncology programs and specialty cancer centers expand targeted therapy services. Procurement authority resides with hospital oncology committees, pharmacy boards, and tumor boards. Adoption focuses on EGFR inhibitor regimens, precision dosing, and patient adherence monitoring. Import formulations dominate high specification cases, while domestic suppliers provide standard therapies. Once approved, therapies are implemented across multiple affiliated hospitals and outpatient centers. Training, protocol standardization, and patient monitoring programs influence adoption. Replacement cycles follow therapy schedules. Budget allocation aligns with hospital expansion and oncology program launches. Commercial success depends on network formulary inclusion, reference site adoption, and long-term supply agreements rather than single clinic or department sales.
Germany is experiencing an 8.1% CAGR in EGFR-mutated lung cancer treatments due to structured clinical guidelines, reimbursement frameworks, and hospital oncology standardization programs. University hospitals and regional cancer centers control most procurement. Adoption emphasizes targeted EGFR therapy, molecular testing, and patient monitoring. Evaluation prioritizes efficacy, safety, and integration with treatment protocols. Procurement follows health technology assessment and capital planning processes. Import products dominate premium regimens while domestic suppliers cover standard therapies. Once approved, deployment spreads across multiple departments and hospitals. Training, protocol adherence, and workflow integration influence utilization. Replacement cycles follow therapy duration and patient management. Budget allocation aligns with oncology program expansion. Commercial success depends on framework inclusion, reference site adoption, and integrated supplier support rather than isolated clinic purchases.
EGFR-mutated lung cancer therapy selection follows clinical guideline alignment, mutation testing protocols, and hospital formulary review rather than product catalog comparison. Oncologists define mutation type, line of therapy, combination regimens, dosing schedule, and safety monitoring before approving a treatment. AstraZeneca, Roche, Pfizer, Takeda, Johnson & Johnson compete at evaluation and guideline integration stages. Once a therapy enters care pathways, prescribing protocols, pharmacy stocking, and patient monitoring routines anchor adoption. AstraZeneca leverages targeted tyrosine kinase inhibitors with clinical evidence and patient support programs. Roche positions therapies through companion diagnostics and molecular profiling integration. Pfizer focuses on oral administration options and tolerability data. Takeda emphasizes long term safety and specialty pharmacy support. Johnson & Johnson aligns therapies with combination treatment strategies and oncology networks.
Competitive positioning reflects efficacy, safety, support programs, and guideline adherence rather than pricing alone. Physicians assess response rates, progression free survival, adverse event profile, and CNS penetration during clinical evaluation. Hospital pharmacists review storage, dispensing logistics, and therapy sequencing. AstraZeneca secures positions where mutation testing and guideline recommendations favor its inhibitors. Roche gains share through companion diagnostic integration and molecular monitoring. Pfizer competes in programs prioritizing adherence and oral dosing convenience. Takeda holds adoption in centers emphasizing long term therapy management. Johnson & Johnson gains placements where combination strategies and clinical protocol alignment drive selection. Market positions vary by hospital type, mutation prevalence, clinical guideline adoption, and payer coverage rules.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD billion |
| Product Type | 3rd-generation TKIs; 2nd-generation TKIs; Combination regimens |
| Application | 1st-line therapy; 2nd-line therapy |
| End User / Sales Channel | Hospital pharmacies; Specialty pharmacies; Retail |
| Region | Asia Pacific; Europe; North America; Latin America; Middle East & Africa |
| Countries Covered | United States, Canada, Mexico, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Brazil, Chile, Saudi Arabia, Turkey, South Africa, Rest of regions |
| Key Companies Profiled | AstraZeneca; Roche; Pfizer; Takeda; Johnson & Johnson |
| Additional Attributes | Dollar by sales by therapy line and product class, 1st versus 2nd line mix, 3rd versus 2nd gen TKI adoption, mutation testing linkage, treatment duration driven demand, adherence and sequencing impact, cold chain and specialty distribution requirements, guideline driven formulary placement, patient support and reimbursement influence |
How big is the egfr-mutated lung cancer treatment market in 2026?
The global egfr-mutated lung cancer treatment market is estimated to be valued at USD 16.1 billion in 2026.
What will be the size of egfr-mutated lung cancer treatment market in 2036?
The market size for the egfr-mutated lung cancer treatment market is projected to reach USD 39.2 billion by 2036.
How much will be the egfr-mutated lung cancer treatment market growth between 2026 and 2036?
The egfr-mutated lung cancer treatment market is expected to grow at a 9.3% CAGR between 2026 and 2036.
What are the key product types in the egfr-mutated lung cancer treatment market?
The key product types in egfr-mutated lung cancer treatment market are 3rd-gen tkis, 2nd-gen tkis and combination regimens.
Which applications segment to contribute significant share in the egfr-mutated lung cancer treatment market in 2026?
In terms of applications, 1st-line therapy segment to command 61.0% share in the egfr-mutated lung cancer treatment market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.