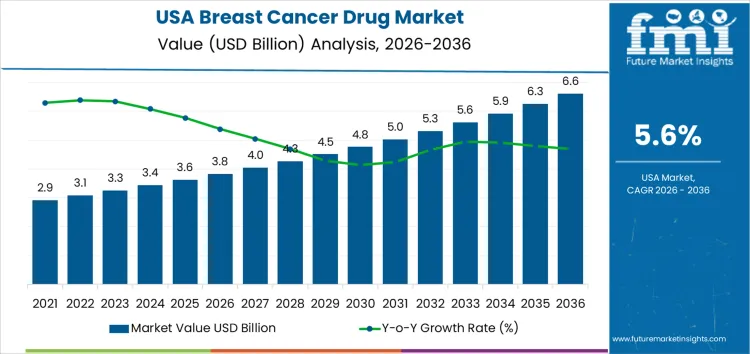
Demand for breast cancer drug in USA is valued at USD 3.8 billion in 2026 and is projected to reach USD 6.6 billion by 2036, reflecting a CAGR of 5.6%. Demand remains relatively stable due to high treatment penetration, established screening programs, and long-term maintenance therapy use. Incremental growth is constrained by generic availability and maturation of key therapy classes, despite ongoing clinical need.
Aromatase inhibitors–based therapies lead drug-class usage because these agents form a standard of care for hormone receptor–positive breast cancer in postmenopausal patients. Clinicians favor aromatase inhibitors for proven efficacy in recurrence reduction and favorable long-term outcomes compared with older endocrine therapies. Continued use in adjuvant and extended treatment settings sustains baseline demand.
West USA, South USA, Northeast USA, and Midwest USA represent key growth regions supported by comprehensive oncology networks, specialist availability, and access to diagnostic and treatment infrastructure. Genentech (Roche), Novartis, Pfizer, Eli Lilly, and Bristol-Myers Squibb anchor competitive activity through branded oncology portfolios, lifecycle management strategies, and combination therapy development aligned with evolving clinical guidelines and patient management practices across the United States.
| Metric | Value |
|---|---|
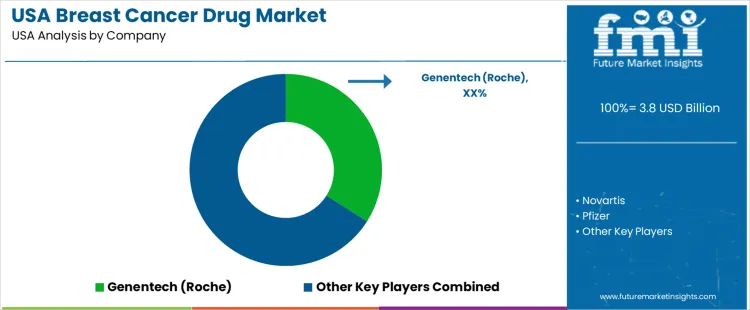
| USA Breast Cancer Drug Sales Value (2026) | USD 3.8 billion |
| USA Breast Cancer Drug Forecast Value (2036) | USD 6.6 billion |
| USA Breast Cancer Drug Forecast CAGR (2026 to 2036) | 5.6% |
Demand for breast cancer drugs in the United States grows due to rising incidence rates, improved screening, and extended survival across patient groups. Early detection through mammography and genetic testing increases the number of patients entering long term treatment pathways. Aging population trends contribute to higher prevalence, since breast cancer risk increases with age. Advances in tumor profiling support personalized therapy selection, which expands use of targeted drugs and hormone based treatments. Clinical guidelines emphasize continuous therapy across early stage, metastatic, and recurrent disease settings, increasing treatment duration.
Expansion of HER2 positive, hormone receptor positive, and triple negative breast cancer classifications drives development and adoption of indication specific drugs. Improved survival outcomes result in prolonged medication use and sequential therapy lines. Broad insurance coverage and oncology benefit programs support patient access to advanced treatments. Growth in outpatient oncology centers increases prescription volume through integrated care delivery models. Ongoing clinical research introduces new drug combinations and novel mechanisms that expand therapeutic options. Patient awareness and adherence programs improve continuity of treatment across care settings. Increasing focus on quality of life and disease control sustains long term demand for breast cancer drug therapies nationwide.
Demand for breast cancer drugs in USA is shaped by screening intensity, hormone receptor testing practices, and long-term adjuvant therapy protocols. Clinicians evaluate mechanism specificity, tolerability, resistance management, and compatibility with combination regimens. Adoption patterns reflect structured oncology pathways, centralized initiation in hospitals, and continued therapy management across outpatient settings under established reimbursement and clinical monitoring frameworks.
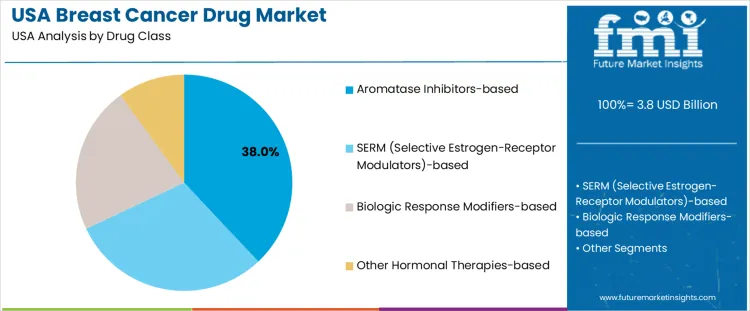
Aromatase inhibitors-based therapies hold 38.0%, representing the largest drug class share in USA demand. These therapies suppress estrogen synthesis, supporting treatment of hormone receptor–positive breast cancer in postmenopausal patients. SERM-based therapies hold 30.0%, modulating estrogen receptor activity and supporting long-term recurrence prevention. Biologic response modifiers–based therapies account for 22.0%, addressing immune-mediated and targeted treatment pathways. Other hormonal therapies hold 10.0%, serving specific clinical profiles and treatment sequences. Drug class distribution reflects emphasis on endocrine-based management aligned with diagnostic stratification.
Key Points
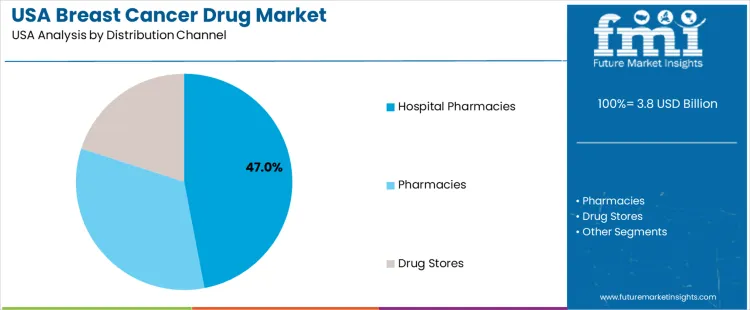
Hospital pharmacies hold 47.0%, accounting for the highest utilization in USA demand. These pharmacies manage therapy initiation, protocol-based dispensing, and coordination with oncology departments. Pharmacies hold 33.0%, supporting ongoing access for patients receiving maintenance or oral therapies. Drug stores account for 20.0%, providing additional access points for refills and long-term treatment adherence. Distribution channel patterns reflect centralized initiation within hospitals followed by decentralized access for continued therapy management.
Key Points
Demand rises as breast cancer remains one of the most commonly diagnosed cancers among women in USA and treatment protocols expand across early, advanced, and metastatic stages. Clinicians prescribe targeted therapies, hormone modulators, chemotherapeutic agents, and immunotherapies based on tumor subtype, receptor status, and patient risk profiles. Adoption aligns with advanced diagnostic practices, multidisciplinary oncology care, and structured treatment pathways within hospital and outpatient oncology networks.
USA breast cancer care emphasizes early detection through mammography and genetic risk assessment, increasing identification of hormone receptor–positive, HER2-positive, and triple-negative subtypes. Oncologists tailor therapy regimens using endocrine therapies, HER2-targeted agents, CDK4/6 inhibitors, and antibody–drug conjugates according to subtype and stage. Clinical guidelines from oncology societies shape sequential therapy decisions to extend progression-free and overall survival. High adoption of precision diagnostics supports targeted drug utilization. Cancer centers and community oncology practices maintain formularies aligned with evidence-based protocols. Multi-agent regimens and maintenance therapies extend treatment duration, supporting sustained drug demand across patient journeys.
Breast cancer therapies often carry high cost, and access depends on private insurance, Medicare, Medicaid, and patient assistance programs. Prior authorization, formulary placement, and cost-sharing affect regimen selection and adherence. Rapid introduction of new agents requires clinicians to balance clinical benefit with payer restrictions. Biosimilars for established biologics influence pricing dynamics and expand access for certain regimens. Clinical trial participation provides early access but also shapes real-world demand post-approval. Patient adherence and management of side effects in long-term therapy influence actual utilization. Demand remains robust due to clinical necessity and guideline-driven care, while growth depends on pricing transparency, equitable access, and continued innovation in breast cancer therapeutics within USA healthcare systems.
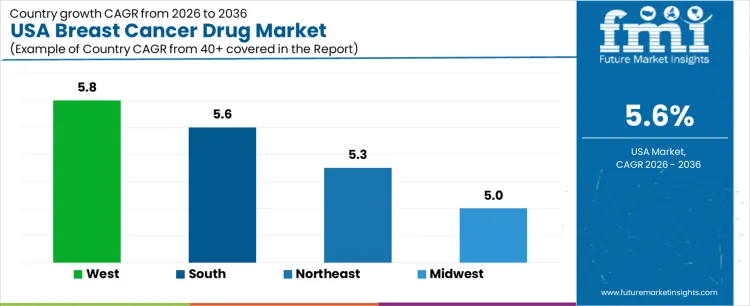
Demand for breast cancer drug in the USA is increasing slowly due to mature treatment protocols, widespread screening, and long-term maintenance therapy use. West USA leads with a 1.2% CAGR, supported by early diagnosis and broad access to oncology care. South USA follows at 1.1%, driven by population growth and expanded treatment coverage. Northeast USA records a 5.6% CAGR, shaped by academic oncology practices and guideline-driven prescribing. Midwest USA posts 0.8%, reflecting integrated health systems and conservative therapy escalation. Regional variation reflects screening penetration, care delivery models, reimbursement discipline, and emphasis on long-duration disease management across the USA.
| Region | CAGR (2026 to 2036) |
|---|---|
| West USA | 1.2% |
| South USA | 1.1% |
| Northeast USA | 1.0% |
| Midwest USA | 0.8% |
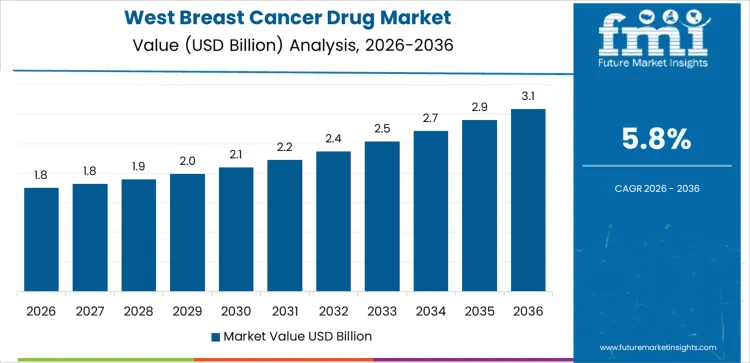
West USA drives demand through strong screening programs, early-stage diagnosis, and high oncology specialist density. Region’s CAGR of 1.2% reflects sustained use of hormonal therapies, targeted agents, and adjuvant treatments over extended treatment periods. Health systems emphasize personalized therapy selection based on biomarker testing. Outpatient oncology centers manage long-term therapy adherence and monitoring. Survivorship programs support continued drug utilization during maintenance phases. Demand favors established therapies with strong real-world evidence and predictable tolerability. Growth remains stability-driven, aligned with early detection and prolonged treatment duration rather than rising incidence.
South USA demand is shaped by population expansion, broader insurance coverage, and improving access to oncology services. Region’s CAGR of 1.1% reflects steady uptake of standard breast cancer drug regimens across community oncology practices. Treatment focuses on widely accepted hormonal and chemotherapy agents. Health systems expand outpatient infusion and oral therapy management. Adherence programs support continuity of care across large geographic areas. Demand growth remains volume-driven, supported by demographic trends and gradual improvements in screening and referral rather than therapy innovation intensity.
Northeast USA demand reflects academic medical centers, evidence-based oncology care, and structured treatment pathways. Region’s CAGR of 1.0% is supported by guideline-aligned prescribing and cautious adoption of new therapies. Clinicians emphasize long-term outcomes, toxicity management, and cost-effectiveness. Clinical trial participation supports early exposure to novel drugs, though broad uptake remains measured. Formularies prioritize therapies with established benefit. Demand growth aligns with patient survival extension and maintenance therapy continuation rather than expansion of treated populations.
Midwest USA demand is anchored in integrated health systems, standardized oncology pathways, and cost-managed care delivery. Region’s CAGR of 0.8% reflects disciplined use of breast cancer drugs within established protocols. Providers prioritize formulary control, treatment consistency, and patient adherence. Screening rates remain stable, supporting predictable case volumes. Therapy escalation occurs cautiously, aligned with clinical necessity. Demand growth remains modest and system-driven, reflecting mature treatment landscapes and focus on long-term disease control rather than expansion of drug utilization.
Demand for breast cancer drugs in the USA is driven by screening protocols, personalized treatment strategies, and expanded therapeutic options for hormone receptor-positive, HER2-positive, and triple-negative subtypes. Buyers evaluate clinical efficacy, safety profiles, biomarker-guided treatment alignment, dosing convenience, and combination therapy potential. Procurement teams prioritize FDA-approved agents with robust trial evidence, support programs for patient adherence, and stable supply chain performance. Trend in the USA market reflects increased use of targeted therapies, immunotherapy integration, and development of agents addressing resistance mechanisms that improve long-term outcomes.
Genentech, a member of the Roche Group, holds a leading position in the USA through a broad portfolio of breast cancer drugs including HER2-targeted therapies supported by extensive clinical evidence and adoption across oncology care settings. Novartis participates with hormone receptor-targeted agents and CDK4/6 inhibitors used widely in hormone-positive disease. Pfizer supports demand through targeted therapies and antibody-drug conjugates integrated into standard treatment regimens. Eli Lilly contributes with HER2 and hormone pathway inhibitors prescribed in both adjuvant and metastatic settings. Bristol-Myers Squibb maintains visibility with immunotherapy combinations employed in select breast cancer populations. Competitive positioning in the USA reflects regulatory alignment, clinical trial outcomes, biomarker-based therapy matching, and comprehensive patient-support services enhancing treatment access and adherence.
| Items | Details |
|---|---|
| Quantitative Units | USD billion |
| Drug Class | Aromatase Inhibitors-based; SERM (Selective Estrogen-Receptor Modulators)-based; Biologic Response Modifiers-based; Other Hormonal Therapies-based |
| Distribution Channel | Hospital Pharmacies; Pharmacies; Drug Stores |
| Regions Covered | West USA; South USA; Northeast USA; Midwest USA |
| Key Companies Profiled | Genentech (Roche); Novartis; Pfizer; Eli Lilly; Bristol-Myers Squibb |
| Additional Attributes | Demand reflects high prevalence, long treatment duration, and adoption of targeted and hormonal therapies. Hospital pharmacies lead oncology dispensing, supported by specialty retail channels. Regional demand aligns with cancer center density, screening rates, reimbursement structures, and access to advanced oncology treatment protocols across the United States. |
How big is the demand for breast cancer drug in USA in 2026?
The demand for breast cancer drug in USA is estimated to be valued at USD 3.8 billion in 2026.
What will be the size of breast cancer drug in USA in 2036?
The market size for the breast cancer drug in USA is projected to reach USD 6.6 billion by 2036.
How much will be the demand for breast cancer drug in USA growth between 2026 and 2036?
The demand for breast cancer drug in USA is expected to grow at a 5.6% CAGR between 2026 and 2036.
What are the key product types in the breast cancer drug in USA?
The key product types in breast cancer drug in USA are aromatase inhibitors-based, serm (selective estrogen-receptor modulators)-based, biologic response modifiers-based and other hormonal therapies-based.
Which distribution channel segment is expected to contribute significant share in the breast cancer drug in USA in 2026?
In terms of distribution channel, hospital pharmacies segment is expected to command 47.0% share in the breast cancer drug in USA in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.