The USA drug screening demand is valued at USD 3.6 billion in 2025 and is projected to reach USD 15.3 billion by 2035, reflecting a CAGR of 15.7%. Growth is driven by expanded workplace testing, increased regulatory compliance in safety-sensitive industries, and heightened public health initiatives targeting substance misuse. Broader adoption of rapid screening technologies supports faster detection across clinical, forensic, and occupational settings.
Urine samples lead usage because they offer a non-invasive collection method, established detection windows, and broad test compatibility for multiple drug classes. Laboratories and point-of-care facilities prioritize assays that deliver high sensitivity, reduced turnaround time, and consistent analytical reliability. Advances in immunoassay and chromatographic techniques strengthen the accuracy of confirmatory testing.
The West, South, and Northeast regions display strong demand based on large workforce populations, active healthcare systems, and well-developed laboratory networks. Key suppliers include Laboratory Corporation of America Holdings, Hoffmann-La Roche Ltd, BioMerieux SA, Express Diagnostics Int’l, Inc., and Bio-Rad Laboratories Inc. Their strategies focus on expanding automated testing platforms, enhancing assay panels, and improving digital reporting tools that streamline screening workflows in high-volume environments.
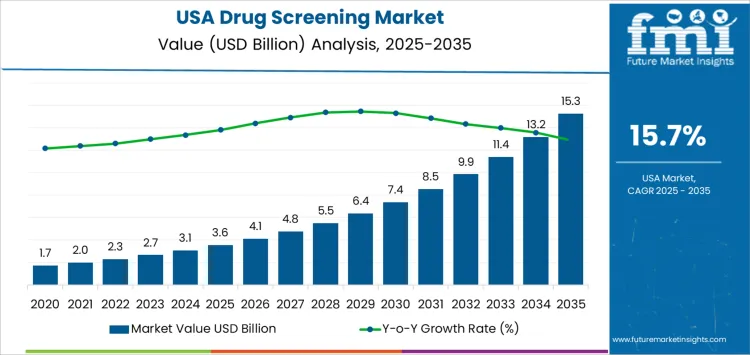
The 10-year growth comparison for drug screening demand in the United States shows a transition from steady early expansion to stronger late-period activity driven by regulatory, workplace, and clinical dynamics. Early-period growth reflects stable utilization across employment testing, probation programs, and clinical toxicology. Testing volumes rise gradually because program structures remain consistent and adoption patterns follow established institutional policies. The early decade shows moderate growth shaped by legacy methodologies, limited panel diversity, and slower integration of high-throughput platforms.
Late-period growth becomes more pronounced as testing requirements expand across safety-sensitive industries and healthcare organizations integrate broader toxicology panels into routine workflows. Increased attention to substance misuse elevates screening frequency across occupational and clinical contexts, creating stronger late-period acceleration. Advancements in analytical sensitivity, multiplex assay capability, and laboratory automation enhance throughput, which supports higher test ordering consistency. Policy updates affecting opioid monitoring programs increase late-period demand across pain management and emergency care settings.
Technology adoption curves also contribute to the comparative shift because later years show wider implementation of mass spectrometry, rapid immunoassays, and electronic reporting systems. The comparison indicates a movement from incremental early growth anchored in long-standing programs to intensified late growth shaped by expanded testing breadth, regulatory alignment, and diagnostic modernization.
| Metric | Value |
|---|---|
| USA Drug Screening Sales Value (2025) | USD 3.6 billion |
| USA Drug Screening Forecast Value (2035) | USD 15.3 billion |
| USA Drug Screening Forecast CAGR (2025-2035) | 15.7% |
Demand for drug screening in the United States rises due to expanding testing requirements across workplaces, healthcare facilities, and legal systems. Employers conduct routine screening to support safety standards in transportation, manufacturing, construction, and logistics. Growth in substance use monitoring programs strengthens adoption of laboratory based and point of care testing methods. Hospitals implement screening protocols to guide clinical decisions, manage overdose cases, and support treatment pathways in emergency and inpatient settings. Physicians use toxicology tests to evaluate medication adherence during pain management and behavioral health therapy.
Courts and correctional institutions apply structured screening to track compliance in probation and rehabilitation programs. Public health agencies monitor drug exposure trends to support community level intervention planning. Wider availability of synthetic substances increases the need for expanded test panels capable of detecting multiple drug classes. Laboratories invest in mass spectrometry and immunoassay platforms that deliver precise results across high sample volumes. Telehealth based treatment programs integrate remote screening models supported by validated collection systems. Insurance policies recognize structured testing within controlled substance management, which maintains consistent demand across clinics and monitoring programs.
Demand for drug screening in the USA is shaped by workplace compliance requirements, clinical toxicology needs, and legal enforcement programs across public and private sectors. Screening workflows depend on specimen stability, detection windows, and ease of collection. Adoption patterns reflect institutional testing policies, device availability, and analytical capacity within laboratories, healthcare providers, and enforcement agencies.
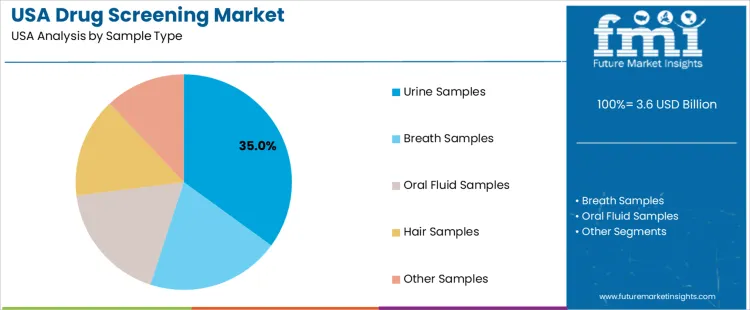
Urine samples hold 35.0%, making them the leading specimen type in USA drug screening. This category supports broad metabolite detection, established regulatory acceptance, and compatibility with automated laboratory workflows. Breath samples hold 20.0%, driven by alcohol-testing programs and portable device usage across law enforcement. Oral fluid samples hold 18.0%, valued for noninvasive collection and rapid screening in workplace and roadside settings. Hair samples hold 15.0%, offering extended detection windows for retrospective drug-use assessment. Other samples hold 12.0%, covering blood, sweat, and alternative matrices used in specialized applications. Distribution reflects detection timelines, specimen handling requirements, and institutional testing protocols.
Key Points:
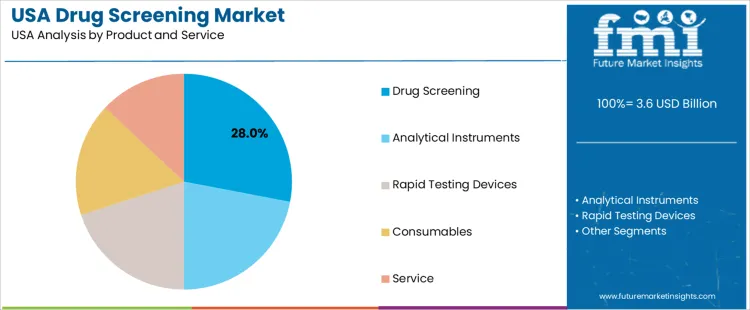
Drug screening holds 28.0%, making it the leading segment across product and service categories in USA demand. This segment includes laboratory-based analyses, confirmatory testing, and integrated screening programs used by employers, healthcare providers, and legal agencies. Analytical instruments hold 22.0%, supporting mass spectrometry, chromatography, and automated immunoassay platforms essential for high-throughput testing. Rapid testing devices hold 20.0%, offering point-of-care capabilities in workplace, clinical, and enforcement environments. Consumables hold 17.0%, covering reagents, cartridges, and specimen-collection materials. Service holds 13.0%, supporting outsourced testing programs and logistics. Distribution reflects testing complexity, throughput requirements, and operational preferences.
Key Points:
Workplace policies, clinical testing needs, and public safety programs are driving demand.
Demand increases as employers across transportation, logistics, construction, and healthcare maintain comprehensive screening protocols aligned with federal and state requirements. Hospitals and outpatient centers expand toxicology testing to support emergency care, behavioural health evaluation, and medication monitoring. Law enforcement agencies use structured screening workflows during roadside assessments and probation management. Substance use treatment programs rely on testing to support counselling and compliance tracking. Educational institutions implement testing policies for athletic programs. Laboratories invest in rapid immunoassay platforms that support high specimen throughput. Insurance providers integrate screening into risk assessment frameworks that govern enrolment and claims review.
Cost variability, operational constraints, and regulatory differences restrain broader adoption.
Facilities experience budget pressure when selecting high-sensitivity analytical methods that require advanced instrumentation. Smaller clinics report limited staffing capacity for sample handling and documentation tasks. Several states maintain differing rules for privacy, consent, and testing conditions, which increases administrative workload for multi-state employers. Turnaround times vary when laboratories manage peaks in specimen volume. Payers apply reimbursement controls that influence clinical testing frequency. Organizations with distributed workforces encounter logistical challenges coordinating specimen collection. Providers face compliance obligations for chain-of-custody standards that increase training requirements and procedural oversight.
Advanced analytics, expanded test panels, and decentralized workflows shape current trends.
US laboratories integrate mass spectrometry methods that improve detection accuracy for emerging synthetic substances. Employers adopt digital platforms that automate scheduling, reporting, and audit documentation. Clinical settings expand oral fluid and hair testing due to longer detection windows and easier collection processes. Point-of-care devices gain traction in urgent care and rehabilitation centers that require immediate results. Public health agencies monitor community exposure patterns to guide prevention programs. Telehealth models incorporate remote sample collection kits for ongoing treatment monitoring. Testing providers introduce configurable panels tailored to occupational risk profiles across industrial, transportation, and healthcare environments.
Demand for drug screening in the USA is rising due to expanding clinical diagnostics, broader workplace testing requirements, and increased healthcare attention to substance-use monitoring. West USA records a CAGR of 18.0% supported by strong biomedical activity and higher utilization of advanced screening platforms. South USA shows a 16.1% CAGR driven by expanding healthcare networks and widespread occupational testing requirements. Northeast USA posts a 14.4% CAGR due to dense clinical infrastructure and strong adoption of analytical technologies. Midwest USA holds a 12.5% CAGR supported by stable institutional testing and broad use of toxicology services across healthcare and employment settings.
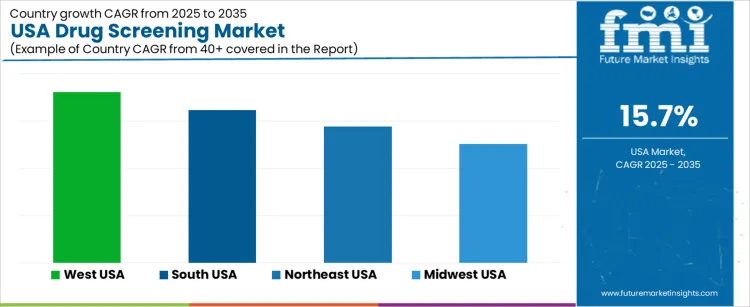
| Region | CAGR (2025-2035) |
|---|---|
| West USA | 18.0% |
| South USA | 16.1% |
| Northeast USA | 14.4% |
| Midwest USA | 12.5% |
West USA drives demand due to its strong concentration of healthcare institutions, research organizations, and diagnostic laboratories that rely on high-throughput screening capabilities. The region’s CAGR of 18.0% reflects continuous use of toxicology panels, advanced analytical assays, and rapid immunoassay formats. Clinical providers maintain regular screening for therapeutic monitoring and substance-use evaluation across high-volume outpatient settings. Research centers engage in pharmacokinetic studies requiring precise analytical workflows. Occupational health programs sustain routine procurement of drug-testing services. The region’s technology-driven diagnostic environment supports consistent integration of mass spectrometry platforms and automated screening systems.
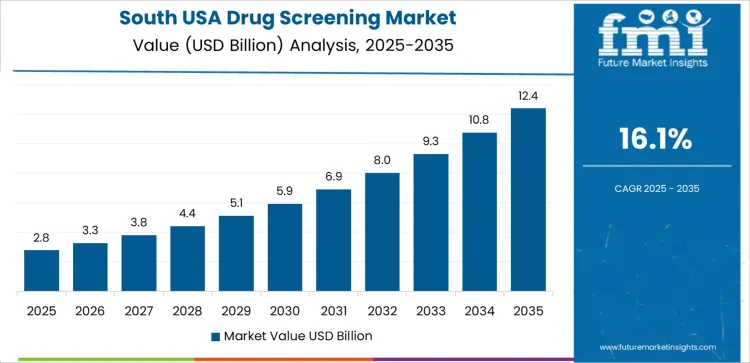
South USA supports rising demand through its large healthcare service base, extensive industrial workforce, and consistent use of employment-related drug testing. The region’s CAGR of 16.1% reflects sustained integration of toxicology panels across hospitals, urgent-care centers, and occupational health programs. Healthcare networks maintain regular screening for substance-use management and medication compliance. Laboratories use automated systems to handle rising testing volumes across urban and suburban areas. Industrial employers rely on screening protocols to support safety requirements. Regional health initiatives encourage continuous usage of analytical workflows across clinical and community settings.
Northeast USA drives demand due to its dense healthcare and research infrastructure, which relies on structured toxicology and medication-monitoring processes. The region’s CAGR of 14.4% reflects steady usage of analytical technologies across research centers, academic hospitals, and clinical laboratories. Providers conduct routine screening for therapeutic oversight, emergency response, and substance-use intervention. Large diagnostic networks maintain integrated workflows for mass spectrometry-based confirmation testing. Occupational health programs support additional testing volumes across major metropolitan employment clusters. The region’s mature clinical environment sustains continuous adoption of standardized drug-screening protocols.
Midwest USA maintains stable demand due to its broad network of hospitals, diagnostic centers, and employment-based testing programs. The region’s CAGR of 12.5% reflects steady integration of immunoassays and confirmatory analytical techniques across clinical settings. Healthcare providers screen for medication adherence, toxic exposures, and substance-use patterns. Regional laboratories support community health programs that require structured screening workflows. Industrial employers rely on routine drug testing to meet safety requirements. Diagnostic networks maintain predictable procurement of consumables and analytical systems used in high-frequency testing operations.
Demand for drug screening in the USA reflects extensive testing requirements across workplace programs, clinical diagnostics, pain management, and public health initiatives. Providers must support high-throughput sample processing, validated immunoassays, chromatographic confirmation methods, and consistent reporting workflows. Buyers evaluate analytical sensitivity, turnaround reliability, assay menu breadth, and integration with electronic medical systems. Procurement teams emphasize accreditation status, chain-of-custody capability, reagent stability, and capacity to manage large multi-site testing volumes. Adoption patterns align with regulatory compliance, occupational safety needs, and structured monitoring within healthcare networks. Hoffmann-La Roche Ltd participates through diagnostic technologies, reagent platforms, and analytical systems incorporated into laboratories performing routine toxicology testing. BioMerieux SA maintains visibility through automated analyzers supporting sample handling and immunoassay workflows used in hospital laboratories. Express Diagnostics Int’l, Inc. provides rapid test kits for point-of-collection screening across occupational and institutional settings. Bio-Rad Laboratories Inc. contributes through reagents, quality controls, and analytical instruments supporting confirmation and validation processes. Competitive positioning in the USA reflects analytical accuracy, instrument reliability, reagent availability, and operational scale supporting varied screening environments.
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Sample Type | Urine Samples, Breath Samples, Oral Fluid Samples, Hair Samples, Other Samples |
| Product and Service | Drug Screening, Analytical Instruments, Rapid Testing Devices, Consumables, Service |
| End User | Drug Testing Laboratories, Criminal Justice and Law Enforcement Agencies, Hospitals, Drug Treatment Centers, Individual Users |
| Regions Covered | West USA, South USA, Northeast USA, Midwest USA |
| Key Companies Profiled | Laboratory Corporation of America Holdings, Hoffmann-La Roche Ltd, BioMerieux SA, Express Diagnostics Int’l, Inc., Bio-Rad Laboratories Inc. |
| Additional Attributes | Dollar sales by sample type, product/service category, and end-user segments; regional demand variations across West, South, Northeast, and Midwest USA; competitive landscape of drug screening system manufacturers and service providers; adoption trends in rapid testing, analytical instrumentation, and multi-panel assays; integration of automated toxicology platforms and digital reporting tools; regulatory influence from workplace testing mandates, forensic standards, and clinical compliance programs. |
How big is the demand for drug screening in USA in 2025?
The demand for drug screening in USA is estimated to be valued at USD 3.6 billion in 2025.
What will be the size of drug screening in USA in 2035?
The market size for the drug screening in USA is projected to reach USD 15.3 billion by 2035.
How much will be the demand for drug screening in USA growth between 2025 and 2035?
The demand for drug screening in USA is expected to grow at a 15.7% CAGR between 2025 and 2035.
What are the key product types in the drug screening in USA?
The key product types in drug screening in USA are urine samples, breath samples, oral fluid samples, hair samples and other samples.
Which product and service segment is expected to contribute significant share in the drug screening in USA in 2025?
In terms of product and service, drug screening segment is expected to command 28.0% share in the drug screening in USA in 2025.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.