The USA drug-induced immune hemolytic anemia demand is valued at USD 3.3 billion in 2025 and is forecasted to reach USD 5.8 billion by 2035, reflecting a CAGR of 5.7%. Growth is driven by increased diagnostic testing, rising awareness of rare adverse drug reactions, and broader utilization of medications associated with immune-mediated hemolysis. Hospitals and reference laboratories continue to expand testing panels to support early identification and treatment.
Cephalosporins lead drug-group association because they are widely prescribed antibiotics with known potential to trigger immune-mediated red blood cell destruction in susceptible individuals. Diagnostic workflows emphasize serologic assays, drug-dependent antibody testing, and molecular evaluations designed to distinguish immune hemolysis from other hematologic disorders. High clinical sensitivity and rapid turnaround support improved patient management.
The West, South, and Northeast regions reflect strong demand due to large healthcare networks and high diagnostic service utilization. Key suppliers include ARUP Laboratories, Cyprotex, Creative Biolabs, Machaon Diagnostics, and F. Hoffmann-La Roche Ltd. Their strategies focus on advanced assay development, expanded clinical reference services, and improved characterization of drug-antibody interactions to support accurate diagnosis and therapeutic decision-making.
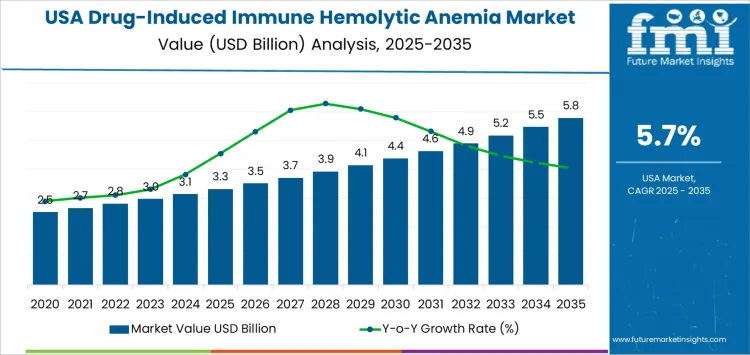
The early vs late growth curve comparison for demand linked to drug-induced immune hemolytic anemia in the United States reflects a transition from sporadic diagnostic activity to more structured testing patterns. Early-curve growth shows limited expansion because case identification depends on clinician familiarity with immune-mediated hemolysis and access to specialized laboratory methods. Testing volumes remain modest in the early phase because incidence is low and diagnostic workflows vary across institutions. Referral patterns to advanced immunohematology laboratories maintain a narrow foundation that defines the initial growth slope.
Late-curve growth shows stronger progression as clinical protocols incorporate structured evaluation for suspected drug-related hemolysis. Wider availability of antibody identification methods increases test ordering consistency across hospitals and specialized diagnostic centers. Pharmacovigilance programs elevate awareness of drug-associated hemolytic events, which increases the likelihood of targeted testing when adverse reactions appear. Consolidation of laboratory networks improves turnaround reliability, creating higher demand stability during the later phase.
The comparison shows a shift from irregular early adoption shaped by clinician experience and limited standardization toward a more consistent late pattern supported by formalized diagnostic pathways and improved laboratory capacity. The late curve maintains higher momentum because coordinated practices increase the frequency of confirmatory testing across relevant clinical settings.
| Metric | Value |
|---|---|
| USA Drug-Induced Immune Hemolytic Anemia Sales Value (2025) | USD 3.3 billion |
| USA Drug-Induced Immune Hemolytic Anemia Forecast Value (2035) | USD 5.8 billion |
| USA Drug-Induced Immune Hemolytic Anemia Forecast CAGR (2025-2035) | 5.7% |
Demand for drug induced immune hemolytic anemia identification and related clinical management in the United States increases due to rising exposure to medications associated with immune mediated red cell destruction. Broader use of antibiotics, anticancer agents, cardiovascular drugs, and anti-inflammatory therapies creates conditions where susceptible individuals experience immune reactions that target erythrocytes. Growth in polypharmacy among older adults elevates cumulative exposure to drug classes linked to hemolytic responses. Expansion of autoimmune disease treatments increases monitoring for hematologic complications that can mimic or trigger immune hemolysis.
Hospitals and diagnostic centers report higher testing volumes as clinicians investigate unexplained anemia, jaundice, and hemolysis markers in patients receiving complex medication regimens. Pharmacovigilance programs strengthen reporting of suspected drug related hematologic events, which raises clinical recognition rates. Broader access to electronic medical records improves detection of temporal associations between medication initiation and hemolytic presentations. Oncology and transplant programs monitor patients for immune mediated hematologic effects generated during intensive therapy cycles. Awareness among healthcare providers increases due to training on adverse drug reactions that require prompt identification and intervention to prevent severe complications.
Demand for drug-induced immune hemolytic anemia evaluation in the USA is shaped by diagnostic complexity, medication exposure patterns, and the need to differentiate immune-mediated hemolysis from other anemia causes. Healthcare providers require confirmation testing for suspected drug-related hemolytic reactions involving antibody formation against red blood cells. Demand distribution reflects drug usage frequency, clinical risk profiles, and institutional responsibility for diagnostic interpretation.
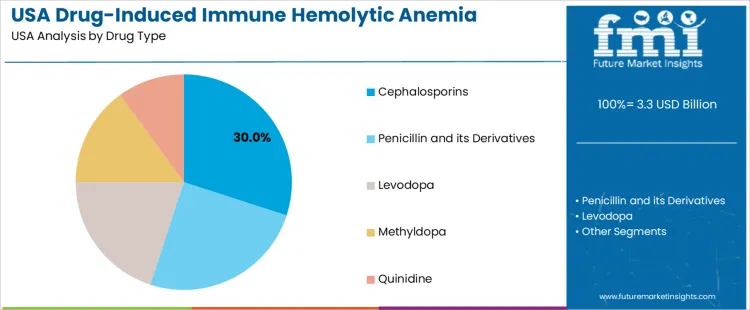
Cephalosporins hold 30.0%, making them the leading drug group associated with immune hemolytic anemia evaluation. Widespread prescription volume and documented antibody-mediated hemolysis cases increase diagnostic frequency. Penicillin and its derivatives hold 25.0%, driven by established links between drug-dependent antibodies and red cell destruction. Levodopa holds 20.0%, reflecting neurological patient populations requiring monitoring for hemolytic responses during long-term therapy. Methyldopa holds 15.0%, associated with autoantibody formation requiring confirmatory testing. Quinidine holds 10.0%, supporting demand through known immune-complex pathways. Distribution reflects drug exposure patterns and documented hemolytic risk across therapeutic categories.
Key Points:
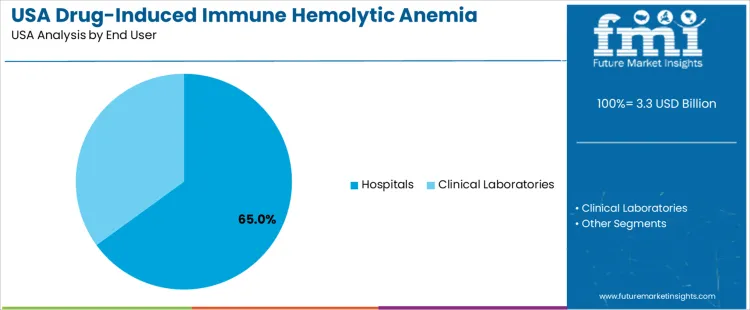
Hospitals hold 65.0%, making them the primary end-user segment for drug-induced immune hemolytic anemia testing in the USA. High-acuity environments manage acute hemolytic events requiring rapid antibody identification and transfusion decision support. Clinical laboratories hold 35.0%, performing specialized testing that includes direct antiglobulin evaluations and drug-dependent antibody detection. These facilities support outpatient clinics, specialty practices, and referral networks requiring confirmatory analysis. End-user distribution aligns with case severity, diagnostic infrastructure, and institutional capacity for immunohematology workflows.
Key Points:
Diagnostic awareness, pharmacovigilance activity, and medication exposure patterns are driving case identification.
Reported cases increase as US hospitals strengthen diagnostic workflows for immune-mediated hematologic conditions linked to medication exposure. Clinical teams monitor patients receiving antibiotics, anticoagulants, and oncology agents that present documented risk of hemolytic reactions. Electronic health record systems support faster recognition of hemolysis indicators such as declining hemoglobin and positive direct antiglobulin tests. Pharmacists expand surveillance of drug interactions that elevate immune-activation risks. Academic centres publish case reviews that improve clinician familiarity with presentation patterns. Transfusion services integrate protocols that guide rapid evaluation of suspected immune-mediated hemolysis. Rising use of complex therapeutic regimens in oncology and chronic illness management increases monitoring frequency.
Limited diagnostic specificity, reporting variability, and therapeutic complexity restrain accurate assessment.
Several healthcare facilities encounter difficulty distinguishing drug-induced immune hemolytic anemia from other hemolytic disorders due to overlapping laboratory features. Reporting patterns vary across states because hospitals follow different internal pharmacovigilance thresholds. Smaller clinics experience limited access to specialized immunohematology testing, which delays confirmation. Treatment pathways require coordination across hematology, pharmacy, and primary care, which increases operational workload. Budget constraints influence availability of advanced antibody detection assays. Patients receiving multiple medications present attribution challenges that complicate clinical decision-making. Discontinuation of suspected agents introduces care continuity issues when alternatives carry different risk profiles.
Advanced diagnostics, structured surveillance, and medication stewardship shape current trends.
US hospitals evaluate expanded use of flow-based assays and enhanced antiglobulin testing to improve detection of drug-dependent antibodies. Health systems integrate structured pharmacovigilance dashboards that track hemolytic events associated with high-risk medications. Hematology programs invest in decision-support tools that guide evaluation of immune-mediated reactions in polypharmacy cases. Academic networks update clinical pathways for managing suspected drug-induced hemolysis using standardized laboratory triggers. Medication stewardship initiatives refine prescribing patterns for agents with documented hemolytic risk. Blood banks introduce protocols that streamline transfusion support for affected patients. Outpatient infusion centres strengthen monitoring procedures for therapies associated with immunologic reactions.
Demand for diagnostics and clinical management related to drug-induced immune hemolytic anemia in the USA is rising due to expanding use of high-risk medications, broader screening activities, and stronger clinical attention to immune-related adverse reactions. West USA records a CAGR of 6.6% supported by intensive therapeutic usage and high patient throughput in large healthcare networks. South USA shows a 5.9% CAGR due to expanding hospital capacity and increasing volumes of drug-related hematologic evaluations. Northeast USA holds a 5.2% CAGR driven by dense specialty care centers. Midwest USA posts a 4.6% CAGR supported by structured diagnostic workflows across widespread clinical systems.
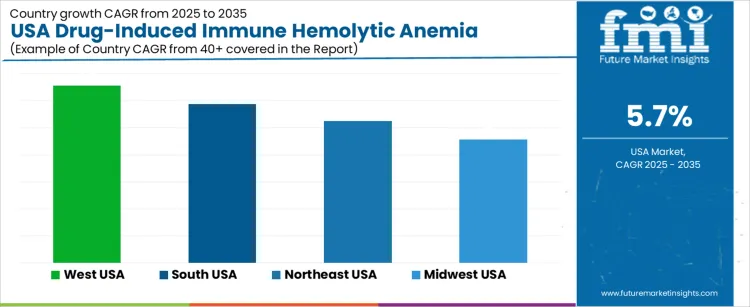
| Region | CAGR (2025-2035) |
|---|---|
| West USA | 6.6% |
| South USA | 5.9% |
| Northeast USA | 5.2% |
| Midwest USA | 4.6% |
West USA drives demand due to the region’s large concentration of hospitals, specialty clinics, and research-oriented medical centers that frequently evaluate patients receiving medications associated with immune-mediated hemolysis. The region’s CAGR of 6.6% reflects consistent testing activity involving direct antiglobulin testing, hematologic monitoring, and immune-reaction assessments. Providers treat diverse patient groups using antimicrobial agents, oncology therapeutics, and cardiovascular drugs that require monitoring for rare but clinically significant hemolytic responses. High outpatient activity supports routine screening for unexplained anemia patterns. Diagnostic laboratories maintain structured workflows for detecting immune-mediated red cell destruction, which contributes to ongoing demand for testing and clinical evaluation.
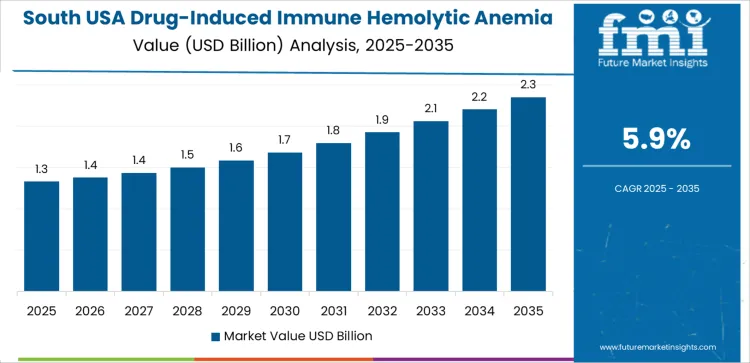
South USA supports rising demand due to expanding healthcare infrastructure and increased utilization of medications linked with immune-hemolytic reactions. The region’s CAGR of 5.9% reflects growing diagnostic engagement across hospitals, outpatient facilities, and hematology practices. Clinical teams conduct regular monitoring of patients on antimicrobials, oncology regimens, and autoimmune therapies that may trigger immune hemolysis. High procedure volumes in regional hospital systems strengthen requirements for confirmatory tests and ongoing hematologic evaluation. Laboratory networks maintain integrated workflows designed to assess antibody-mediated red cell destruction, which supports continuous demand for specialized diagnostic activities.
Northeast USA drives demand due to its dense clinical ecosystem with high reliance on specialty care, academic medical centers, and advanced diagnostic capabilities. The region’s CAGR of 5.2% reflects strong utilization of hematology and immunology tests required to detect immune-mediated hemolysis in patients receiving complex therapeutic regimens. High patient turnover in large hospitals strengthens routine evaluation of anemia patterns, especially where immune reactions are suspected. Diagnostic laboratories conduct regular direct antiglobulin testing supported by structured protocols. Specialist practices maintain steady assessment activities due to the region’s broad adoption of medications with known immune-reaction risk profiles.
Midwest USA supports stable demand through its broad network of hospitals, diagnostic centers, and hematology practices that manage patients receiving medications associated with immune-related hemolytic complications. The region’s CAGR of 4.6% reflects steady testing activity for anemia assessments, immunologic evaluations, and monitoring of drug-associated hemolysis. Healthcare systems across mid-sized cities maintain consistent diagnostic workflows involving antibody detection and red cell integrity assessment. Providers continue monitoring patients using antimicrobial agents, oncology therapies, and chronic-care medications where immune-mediated reactions may occur. Routine hematologic testing supports ongoing demand across regional institutions.
Demand for diagnostics related to drug-induced immune hemolytic anemia in the USA reflects the need for accurate antibody characterization, specialized serologic testing, and advanced analytical platforms supporting differential evaluation of hemolytic events. Clinical laboratories assess assay sensitivity, turnaround consistency, reagent stability, and validated workflows for detecting drug-dependent antibodies linked to hematologic complications. Hospitals and reference laboratories prioritize providers capable of supporting complex immunohematology investigations, including flow cytometry, complement activation assessment, and serologic cross-reactivity studies. Procurement teams focus on dependable testing infrastructure, trained personnel, and alignment with quality management standards required for rare event analysis. Cyprotex participates through in vitro toxicology and mechanistic assays that support research programs examining drug-related hematologic effects. Creative Biolabs contributes with custom antibody development, immune response characterization, and laboratory platforms used in preclinical assessments. Machaon Diagnostics maintains visibility through esoteric hematology testing, complement assays, and rapid analysis supporting acute clinical decision requirements. F. Hoffmann-La Roche Ltd. operates through diagnostic technologies and analytical instruments used in hematology laboratories addressing immune-mediated red cell destruction. Competitive positioning in the USA reflects assay accuracy, interpretive expertise, and laboratory throughput supporting clinicians managing suspected drug-related hemolysis.
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Drug Type | Cephalosporins, Penicillin and its Derivatives, Levodopa, Methyldopa, Quinidine |
| End User | Hospitals, Clinical Laboratories |
| Regions Covered | West USA, South USA, Northeast USA, Midwest USA |
| Key Companies Profiled | ARUP Laboratories, Cyprotex, Creative Biolabs, Machaon Diagnostics, F. Hoffmann-La Roche Ltd. |
| Additional Attributes | Dollar sales by drug class and end-user categories; regional demand variation across West, South, Northeast, and Midwest USA; diagnostic and therapeutic adoption patterns in hospital and clinical laboratory settings; competitive landscape of immunohematology testing, antibody detection services, and hemolysis-related diagnostic platforms; integration of drug-induced hemolytic anemia assays within hematology workflows and adverse drug reaction monitoring programs. |
How big is the demand for drug-induced immune hemolytic anemia in USA in 2025?
The demand for drug-induced immune hemolytic anemia in USA is estimated to be valued at USD 3.3 billion in 2025.
What will be the size of drug-induced immune hemolytic anemia in USA in 2035?
The market size for the drug-induced immune hemolytic anemia in USA is projected to reach USD 5.8 billion by 2035.
How much will be the demand for drug-induced immune hemolytic anemia in USA growth between 2025 and 2035?
The demand for drug-induced immune hemolytic anemia in USA is expected to grow at a 5.7% CAGR between 2025 and 2035.
What are the key product types in the drug-induced immune hemolytic anemia in USA?
The key product types in drug-induced immune hemolytic anemia in USA are cephalosporins, penicillin and its derivatives, levodopa, methyldopa and quinidine.
Which end user segment is expected to contribute significant share in the drug-induced immune hemolytic anemia in USA in 2025?
In terms of end user, hospitals segment is expected to command 65.0% share in the drug-induced immune hemolytic anemia in USA in 2025.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.