Demand for diamond-blackfan anemia (DBA) syndrome therapeutics in the USA is projected to grow from USD 2.0 billion in 2026 to USD 3.1 billion in 2036, reflecting a 4.0% CAGR over the forecast period. This steady growth can be attributed to the increasing awareness and diagnosis of DBA, a rare genetic disorder characterized by bone marrow failure, which is driving demand for specialized therapeutics. The availability of advanced treatments, including gene therapies, corticosteroids, and blood transfusion management, is playing a key role in expanding therapeutic options for DBA patients. As the understanding of the disease deepens and new treatments are developed, the demand for DBA therapeutics is expected to rise gradually. From USD 1.7 billion in 2026, the demand sees consistent increases over the following years, reaching USD 2.0 billion by 2029.
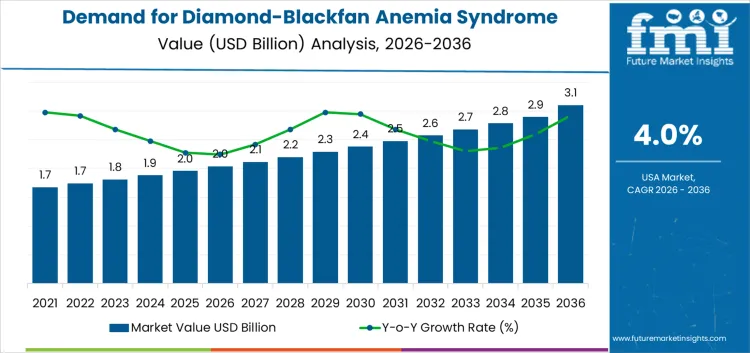
Growth trajectory shows a stable upward trend, with the demand increasing to USD 2.1 billion in 2030, USD 2.2 billion in 2031, and continuing in similar increments through the forecast period. By 2035, the demand for DBA therapeutics is expected to reach USD 2.8 billion, driven by the continued advancements in treatment options and a greater focus on improving patient outcomes. As ongoing research efforts contribute to new therapies and improve current treatments, the DBA therapeutic sector will see sustained growth through 2036, benefiting from ongoing innovation and increased patient awareness.
| Metric | Value |
|---|---|
| Demand for Diamond-Blackfan Anemia Syndrome Therapeutics in USA Value (2026) | USD 2.0 billion |
| Demand for Diamond-Blackfan Anemia Syndrome Therapeutics in USA Forecast Value (2036) | USD 3.1 billion |
| Demand for Diamond-Blackfan Anemia Syndrome Therapeutics in USA Forecast CAGR (2026-2036) | 4.0% |
The demand for therapeutics for diamond-blackfan anemia (DBA) syndrome in the USA is growing due to the increasing awareness of the disease, advancements in genetic research, and the development of more targeted and effective treatments. DBA is a rare, inherited blood disorder characterized by a failure in red blood cell production, leading to anemia and other serious complications. As the understanding of DBA improves, the focus on developing more effective therapies, including corticosteroids, transfusions, and emerging gene therapies, is driving growth in the therapeutic segment. With the expansion of precision medicine and better diagnostic tools, more individuals with DBA are being identified and treated, further increasing demand for therapeutic solutions.
Key drivers for this growth include advancements in research that are leading to the development of new and more effective therapies aimed at treating DBA. These include targeted therapies and investigational treatments that offer the potential for more personalized approaches to care. The growing awareness of rare diseases and the push for increased research funding are also contributing to progress in DBA treatment options. As the prevalence of DBA continues to be better understood, there is a rising demand for treatments to address both the short-term and long-term needs of affected patients, including those who experience complications such as growth failure and congenital abnormalities.
Demand for diamond-blackfan anemia (DBA) syndrome therapeutics in the USA is segmented by treatment type, end-user, and region. By treatment type, corticosteroids lead with 28% of the demand, followed by chelation therapy, blood transfusions, stem cell transplants, and other therapies. In terms of end-user, hospitals account for 55% of the demand, with clinics and others making up the remaining share. Regionally, demand is distributed across West USA, South USA, Northeast USA, and Midwest USA.
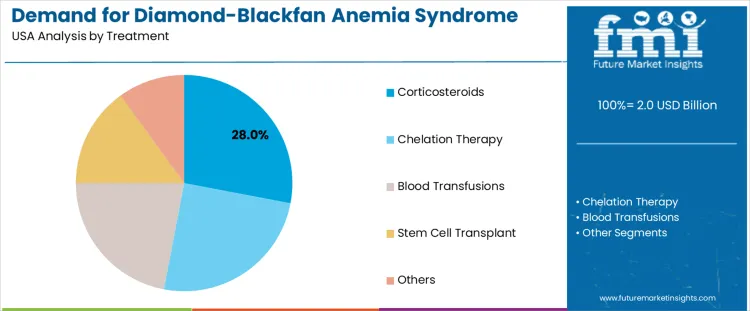
Corticosteroids account for 28% of the demand for Diamond-Blackfan Anemia (DBA) syndrome therapeutics in the USA, driven by their role as the first-line treatment for managing symptoms of the condition. Corticosteroids, such as prednisone, are commonly prescribed to DBA patients to stimulate red blood cell production and reduce the need for frequent blood transfusions. As the most commonly used treatment for DBA, corticosteroids help manage anemia by addressing the underlying issue of insufficient red blood cells. The effectiveness and relatively lower cost of corticosteroids contribute to their widespread use, particularly in pediatric patients. corticosteroids are often used as a bridge therapy while patients await stem cell transplants or other long-term treatments. As awareness of Diamond-Blackfan Anemia and advancements in corticosteroid therapies continue to evolve, corticosteroids will remain a dominant therapeutic option in the DBA treatment landscape.
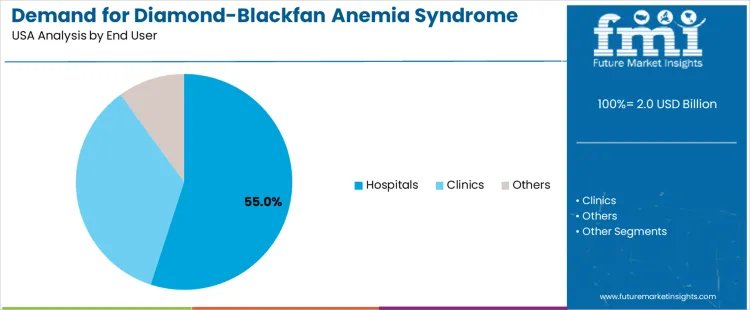
Hospitals account for 55% of the demand for Diamond-Blackfan Anemia (DBA) syndrome therapeutics in the USA, driven by their role as primary care providers for patients with rare and complex conditions. DBA syndrome often requires specialized care, and hospitals are equipped with the necessary resources, such as hematology departments, to provide treatment and management for these patients. Hospitals are typically the first point of contact for diagnosis and initial treatment, where corticosteroids, blood transfusions, and other therapies are administered. Hospitals are also involved in stem cell transplants for eligible patients, making them a key hub for the comprehensive care that DBA patients require. With the growing number of DBA cases being diagnosed, and the increasing need for advanced therapies, hospitals will continue to play a crucial role in managing the disease, thereby driving the demand for DBA therapeutics.
DBA is a rare congenital bone marrow failure disorder that requires treatments such as corticosteroids, blood transfusions, iron chelation, and potentially stem cell transplant or emerging gene therapies. Drivers include unmet medical need, patient advocacy, and research into targeted therapies. Restraints include the rarity of the condition, high cost of treatment, limited approved drugs, and significant side effects associated with current therapies, which may limit broader uptake.
Why is Demand for Diamond‑Blackfan Anemia Syndrome Therapeutics Growing in USA?
Demand for DBA therapeutics in the USA is growing because improved screening and genetic testing identify more affected individuals, while better disease awareness among hematologists supports timely intervention. As survival improves with supportive care, long‑term management becomes critical, increasing therapeutic demand. Patients and families actively seek advanced treatment options to reduce transfusion dependence and mitigate complications like iron overload. Research into novel treatments, including targeted agents and gene therapies, heightens interest in more effective, disease‑modifying options. These factors together sustain demand for existing and emerging DBA therapeutics.
How are Technological and Industry Innovations Driving Diamond‑Blackfan Anemia Syndrome Therapeutic Demand in USA?
Technological and industry innovations are shaping demand for DBA therapeutics in the USA by expanding treatment possibilities. Advances in genetic and molecular research have improved understanding of the ribosomal protein gene mutations underlying DBA, creating pathways for targeted therapies. Progress in gene therapy and gene editing offers potential curative strategies rather than lifelong supportive care. Improved drug delivery systems and better iron chelation therapies enhance patient quality of life. Clinical trials of new agents generate hope and increase demand for participation and access. These innovations encourage healthcare providers and patients to pursue more effective, personalized therapeutic options for DBA.
What are the Key Challenges and Risks That Could Limit Diamond‑Blackfan Anemia Syndrome Therapeutic Demand in USA?
Rarity of the disease restricts patient populations, making large‑scale drug development costly and limiting commercial incentives. High treatment costs, especially for advanced therapies like gene therapy, may not be fully covered by insurance, reducing access. Side effects and long‑term safety concerns with existing treatments can deter use. Regulatory hurdles and lengthy clinical trial requirements delay approval of new therapies. Patient and provider uncertainty about long‑term outcomes with novel treatments may temper adoption until more data become available.
| Region | CAGR (%) |
|---|---|
| West USA | 4.6% |
| South USA | 4.1% |
| Northeast USA | 3.7% |
| Midwest USA | 3.2% |
Demand for diamond-blackfan anemia (DBA) syndrome therapeutics in the USA is steadily growing, with West USA leading at a 4.6% CAGR, driven by its advanced healthcare infrastructure and research focus on rare diseases. South USA follows with a 4.1% CAGR, supported by expanding healthcare facilities and increasing awareness of genetic disorders. Northeast USA shows a 3.7% CAGR, fueled by strong medical research institutions and advancements in gene therapy for rare diseases. Midwest USA experiences a 3.2% CAGR, with steady growth supported by specialized clinics and research in rare blood disorders.
West USA leads the demand for diamond-blackfan anemia (DBA) syndrome therapeutics, growing at a 4.6% CAGR. The region's advanced healthcare infrastructure, combined with significant research institutions and biotechnology companies, is driving the adoption of innovative therapies for rare diseases like DBA. States such as California and Washington are home to leading medical centers and hospitals specializing in genetic disorders, contributing to increased awareness and diagnosis of DBA. Ongoing clinical trials and advancements in gene therapy are helping improve treatment options for DBA patients in the region. The growing focus on personalized medicine and specialized treatments in West USA is further boosting the demand for DBA therapeutics. As genetic research and rare disease treatments continue to gain momentum, West USA is likely to see continued growth in the adoption of DBA therapeutics, ensuring better care for patients with this condition.
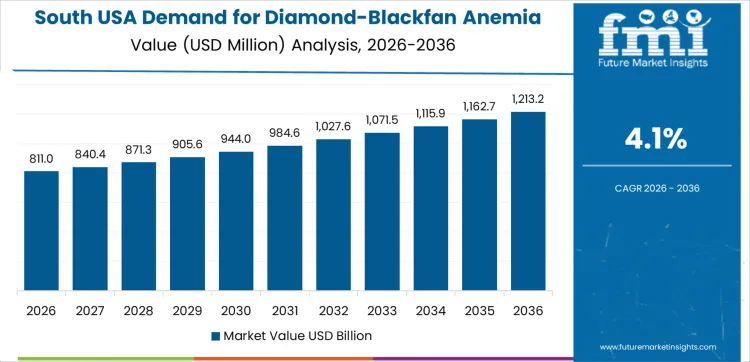
South USA is experiencing steady demand for diamond-blackfan anemia (DBA) syndrome therapeutics, with a 4.1% CAGR. The region’s expanding healthcare sector, along with increasing awareness of rare blood disorders, is contributing to the growth in demand for DBA therapeutics. States like Texas and Florida are seeing a rise in the number of specialized treatment centers and hospitals offering care for DBA patients, leading to better diagnosis and access to therapies. The South's large population and the increasing focus on rare disease management and genetic disorders are helping to drive demand for DBA treatments. The region's growing participation in clinical trials and research programs targeting rare blood disorders is contributing to this demand. As the South continues to prioritize advancements in healthcare and rare disease treatment, the demand for DBA therapeutics is expected to remain strong, helping to improve the quality of life for affected patients.
Northeast USA is seeing steady demand for diamond-blackfan anemia (DBA) syndrome therapeutics, with a 3.7% CAGR. The region’s strong healthcare infrastructure, particularly in states like New York and Pennsylvania, is supporting the growth in the adoption of DBA therapeutics. Major medical institutions and research centers in the Northeast are at the forefront of developing new treatments for genetic disorders like DBA. The increasing awareness of DBA and other rare diseases, coupled with a growing number of specialized clinics and expert healthcare providers, is helping patients receive more accurate diagnoses and better access to treatment. The region’s focus on advanced gene therapies and innovative treatment methods for rare blood disorders is also contributing to the demand for DBA therapeutics. As the region continues to lead in medical research and healthcare excellence, demand for DBA therapeutics is expected to grow steadily, improving treatment outcomes for patients in Northeast USA.
Midwest USA is experiencing moderate demand for diamond-blackfan anemia (DBA) syndrome therapeutics, with a 3.2% CAGR. The region’s healthcare systems, particularly in states like Illinois, Michigan, and Ohio, are increasingly addressing the needs of patients with rare blood disorders, contributing to the growing demand for DBA therapeutics. As the Midwest continues to advance in rare disease research and genetic medicine, new treatment options are becoming more accessible to DBA patients. Specialized medical centers and universities in the region are actively involved in clinical research and trials focused on rare genetic disorders, including DBA, which is driving innovation in therapies. The increasing focus on personalized treatments and genetic-based medicine is also playing a role in this growth. As more healthcare providers and research centers in the Midwest focus on rare disease management, demand for DBA therapeutics will continue to rise steadily, improving patient care and outcomes in the region.
Demand for therapeutics for diamond-blackfan anemia (DBA) syndrome in the USA is increasing as more is understood about this rare, congenital blood disorder. DBA leads to a failure of the bone marrow to produce sufficient red blood cells, often requiring long-term treatment. With ongoing advancements in medical research and the growing focus on rare diseases, the demand for effective DBA therapies is rising, driven by the need for improved treatments to manage symptoms, enhance patient outcomes, and reduce reliance on blood transfusions.
Merck KGaA is a key player in the DBA therapeutics industry, offering innovative treatment options aimed at managing the symptoms of this rare blood disorder. The company’s strategy focuses on developing targeted therapies that address the underlying causes of DBA, helping to improve the quality of life for patients. Their strong commitment to rare disease research and development has positioned them as a significant competitor in the therapeutic landscape.
Other major players include Bayer AG, Cipla Inc., GlaxoSmithKline plc, and AstraZeneca. Bayer AG emphasizes research into rare genetic disorders and provides treatments aimed at managing DBA-related anemia. Cipla Inc. is actively involved in bringing affordable and accessible therapeutics for rare diseases, expanding its presence in the DBA therapeutic area. GlaxoSmithKline and AstraZeneca are also focused on the rare disease industry, with research into anemia-related conditions, aiming to offer more effective, long-term solutions for managing DBA symptoms. As competition intensifies, innovation in therapies, patient access programs, and tailored treatments will play a critical role in shaping the future of DBA syndrome therapeutics in the USA.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 2.0 billion |
| Key Segments | Corticosteroids, Chelation Therapy, Blood Transfusions, Stem Cell Transplant, Others |
| End User | Hospitals, Clinics, Others |
| Region | West USA, South USA, Northeast USA, Midwest USA |
| Key Players Profiled | Merck KGaA, Bayer AG, Cipla Inc., GlaxoSmithKline plc, AstraZeneca |
| Additional Attributes | Dollar sales by treatment type and end-user; regional CAGR and growth trends in Diamond-Blackfan Anemia (DBA) Syndrome therapeutics in the USA |
How big is the demand for diamond-blackfan anemia syndrome therapeutics in USA in 2026?
The demand for diamond-blackfan anemia syndrome therapeutics in USA is estimated to be valued at USD 2.0 billion in 2026.
What will be the size of diamond-blackfan anemia syndrome therapeutics in USA in 2036?
The market size for the diamond-blackfan anemia syndrome therapeutics in USA is projected to reach USD 3.1 billion by 2036.
How much will be the demand for diamond-blackfan anemia syndrome therapeutics in USA growth between 2026 and 2036?
The demand for diamond-blackfan anemia syndrome therapeutics in USA is expected to grow at a 4.0% CAGR between 2026 and 2036.
What are the key product types in the diamond-blackfan anemia syndrome therapeutics in USA?
The key product types in diamond-blackfan anemia syndrome therapeutics in USA are corticosteroids, chelation therapy, blood transfusions, stem cell transplant and others.
Which end user segment is expected to contribute significant share in the diamond-blackfan anemia syndrome therapeutics in USA in 2026?
In terms of end user, hospitals segment is expected to command 55.0% share in the diamond-blackfan anemia syndrome therapeutics in USA in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.