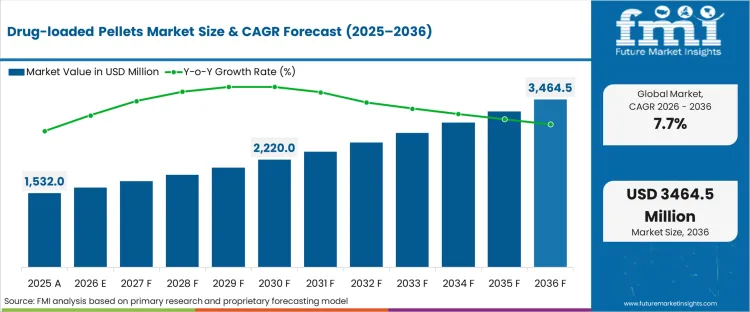
In 2026, the drug-loaded pellets market was valued at USD 1,650.0 million. Based on Future Market Insights analysis, demand for drug-loaded pellets is estimated to grow to USD 3,450.0 million by 2036. FMI projects a CAGR of 7.7% during the forecast period.
Absolute dollar growth of USD 1,800.0 million over the decade signals structural expansion rather than a packaging-led cycle. As per FMI, growth is expected to be supported by wider use of multiparticulate dosing to manage release control and reduce peak–trough variability, while scale-up validation, batch-to-batch uniformity requirements, and tech-transfer complexity are expected to pace adoption in regulated launches. “Aenova’s extended range of development services allows us to support pharmaceutical and biotech companies in accelerating their programs and optimizing their resources, Our Aenovation™ program is a reflection of our commitment to innovation and excellence in pharmaceutical development.” said Florent Bordet, Chief Scientific Officer at Aenova. [1]
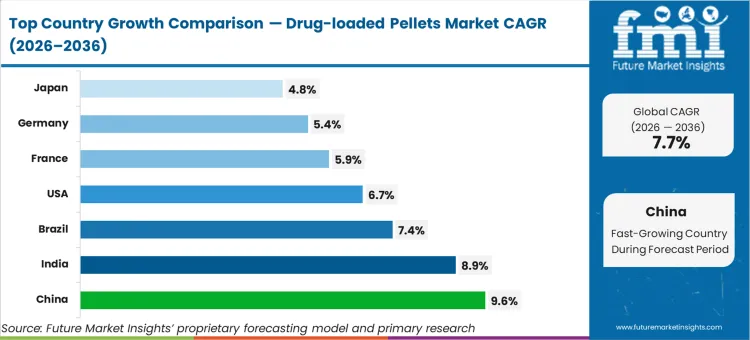
China (9.6% CAGR, supported by expanding oral solid dose capacity and higher adoption of controlled-release formats) and India (8.9% CAGR, driven by rising generic and CDMO output) are expected to lead growth. Brazil (7.4% CAGR) is expected to expand through broader use of modified-release products. The U.S. (6.7% CAGR) is expected to grow through innovator demand for differentiated release profiles. Mature markets including France (5.9%), Germany (5.4%), and Japan (4.8%) are expected to contribute more through replacement demand, constrained by validation timelines and conservative platform switching.
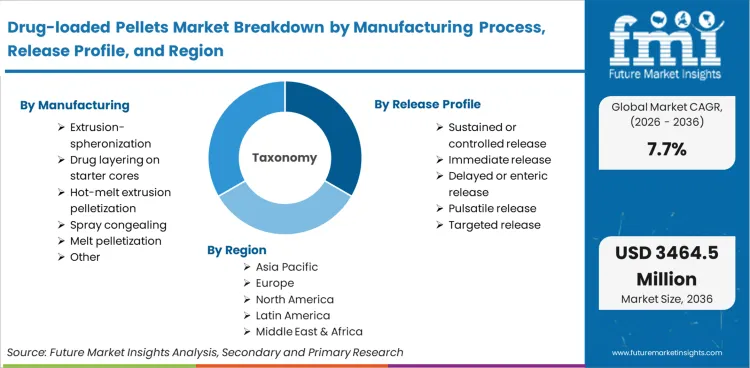
The market includes multiparticulate drug-loaded pellets that are produced as intermediates or finished oral dosage forms designed for the purpose of controlled, delayed, or targeted drug release. The pellets are processed by methods such as extrusion/spheronization, drug coating on starter cores, melt or hot melt extrusion pelletization, and spray congealing and are packaged in capsules, MUPS tablets, sachets/sprinkles, oral suspensions, and other forms. The customers include innovator pharma, generic companies, CDMO/CMO service providers, research institutes, and veterinary pharma companies. The market is driven by the requirements of release profile, manufacturability, and quality specifications related to uniformity and particle size.
The report includes global and regional market sizing and a 10-year forecast for 2026 to 2036. Segment-level sizing is provided by manufacturing process, release profile, therapeutic area, dosage form integration, customer type, and particle size range, with country-level CAGR comparisons across key markets. Competitive positioning is covered for major pellet technology and manufacturing providers, along with analysis of outsourcing patterns, tech-transfer considerations, and quality expectations affecting platform selection, as per FMI analysis.
The scope excludes conventional single-unit oral solids that do not use pelletized multiparticulates, including standard immediate-release tablets and capsules filled with simple powders. It also omits APIs sold as bulk substances without pelletization, and excludes finished drug product revenue where pellets are only a minor internal component and not separately priced. Non-pharma industrial pellets are excluded. The focus remains on drug-loaded pellet intermediates and pellet-integrated dosage formats aligned to the listed segments and customer types.
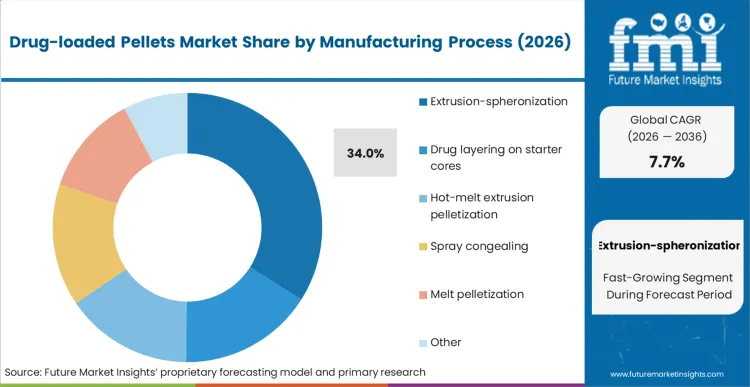
Extrusion-spheronization is projected to command a 34% share in 2026, followed by drug layering on starter cores with a 28% share. Extrusion-spheronization is the leader because it is suited for large-scale production of pellets with controlled size distribution, which is ideal for sustained release formulations that require uniform coating. Drug layering is the second market because it is suited for potent APIs, but cycle time and drug layering efficiency impact scale economies.
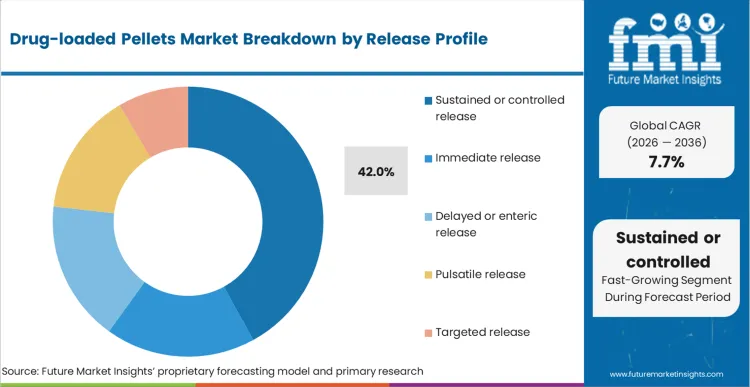
Controlled and sustained release holds the second-largest market share of 42% in 2026, followed by immediate release with a market share of 26%, according to a report by FMI. The dominance of sustained release products is a result of the growing need for a smoother pharmacokinetic profile and less frequent dosing, which is met by the use of coated pellet technology in capsules or MUPS tablets.
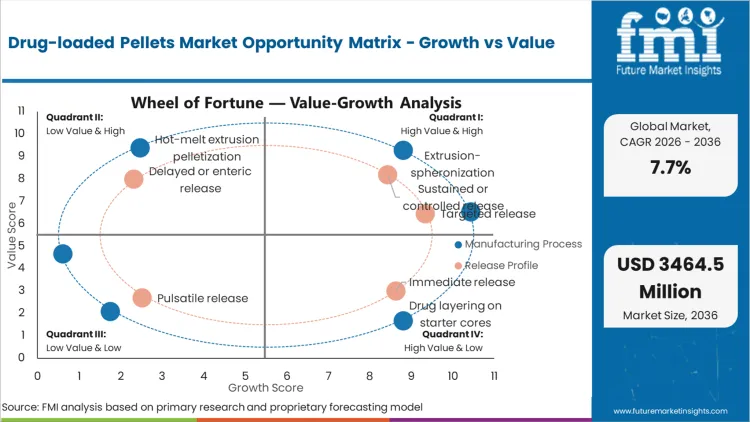
Future Market Insights analysis that historical patterns point at a formulation-led oral solids category where demand is anchored in controlled release design, dissolution curve management, and multi-particulate manufacturing discipline. Adoption is being driven by the need to balance dose loading, swallowability, and release performance within capsules and MUPS tablets, as per FMI.
While growth is being restrained by process complexity, longer development timelines, and tighter dissolution acceptance criteria that raise the cost of change-control, value expansion is being supported by sustained-release dominance and increasing outsourcing to specialized manufacturers. Based on FMI’s report, supplier selection is increasingly being filtered by reproducibility, documentation readiness, and predictable tech-transfer outcomes.
Based on the regional analysis, drug-loaded pellets market is segmented into North America, Latin America, East Asia, South Asia & Pacific, Western Europe, Eastern Europe and Middle East & Africa across 40+ countries. Regional performance is interpreted through adoption of modified-release oral solids, scale-up activity in palletization lines, and outsourcing patterns among CMOs and CDMOs, as per FMI. The full report also offers market attractiveness analysis based on regional trends.
.webp)
| Country | CAGR |
|---|---|
| United States | 6.7% |
| India | 8.9% |
| Germany | 5.4% |
| China | 9.6% |
| Japan | 4.8% |
| France | 5.9% |
| Brazil | 7.4% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
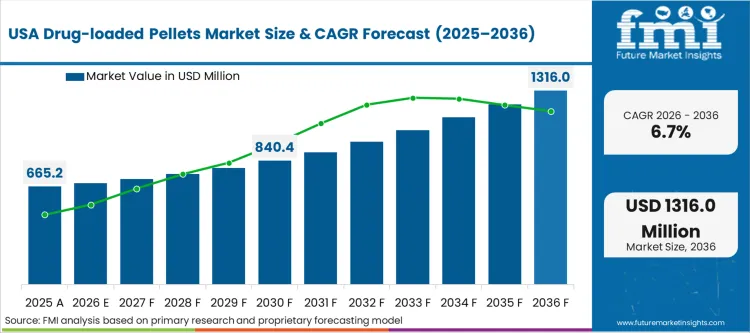
North America is shaped by formulation-led lifecycle management, where pellet-based systems are selected to achieve controlled release, taste masking, and multiparticulate flexibility across capsules and MUPS tablets. Catalent participates through development-to-commercial services that support pelletization and downstream oral solid manufacturing. Thermo Fisher Scientific supports CDMO-linked oral solid capacity that can absorb scale-up work for controlled release programs. Based on FMI’s report, innovation-led demand and generic competition both support continued use of multiparticulate approaches.
FMI’s report includes a detailed analysis of the growth in the North American region, along with a country-wise assessment that includes the U.S., Canada and Mexico. Readers can also find regional trends, regulations, and market growth based on different segments and countries in the North America region.
Latin America remains import-reliant for many high-spec multiparticulate products, with selective local manufacturing for oral solids. Demand is paced by generic uptake, availability of appropriate excipient supply, and access to contracted pelletization capability. Based on FMI’s report, uptake remains concentrated where local manufacturers and regional CDMOs can support sustained release formats at acceptable cost.
The report consists of a detailed analysis for the market in Brazil, Argentina and Rest of Latin America. Readers can find detailed information about several factors, such as the pricing analysis and regional trends, which are impacting growth in the Latin America region.
East Asia is driven by scale-up in complex generics and expanding CDMO capacity, where multiparticulate delivery is used to replicate reference release profiles and improve formulation performance. Evonik supports demand through polymer and formulation inputs aligned to controlled release needs. Lonza and other service providers participate where pelletization and downstream processing are integrated into broader oral solid programs. FMI analysts note that sustained and controlled release remains the lead release profile in many programs due to chronic therapy demand.
The full report analyzes the drug-loaded pellets market across East and South Asia from 2021-2036, covering pricing, trends, and growth drivers in China, Japan, South Korea, India, Indonesia, Malaysia, and Thailand. The assessment highlights trends that dictate regional demand and procurement behaviour.
South Asia & Pacific functions as a formulation and manufacturing scale-up region, where demand is supported by expanding CDMO and generic manufacturer activity and rising use of controlled release oral solids. Colorcon supports adoption through functional coating systems used in modified release pellets and MUPS. Catalent and other service providers influence demand where development support and tech transfer capability is required. FMI analysts note that capsule-based pellets remain dominant due to integration simplicity.
FMI’s report includes a detailed analysis of the growth in the South Asia & Pacific region, along with a country-wise assessment that includes India, ASEAN Countries, Australia & New Zealand and Rest of South Asia. Readers can also find regional trends, regulations, and market growth based on different segments and countries in the South Asia & Pacific region.
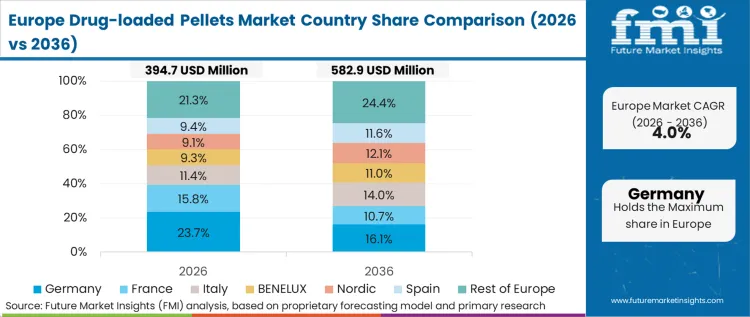
Western Europe is shaped by regulated manufacturing discipline and strong presence of specialty oral solid CMOs, where multiparticulate pellets are used for controlled release and for complex generics that require precise release matching. Evonik participates through excipient and polymer inputs supporting modified release designs. Siegfried and Recipharm maintain relevance through oral solid manufacturing services aligned to regulated markets. Based on FMI’s report, demand remains anchored in sustained and controlled release profiles.
FMI’s analysis of drug-loaded pellets market in Western Europe consists of country-wise assessment that includes Germany, U.K., France, Italy, Spain, BENELUX, Nordic Countries and Rest of Western Europe. Readers can know various regulations and latest trends in the regional market.
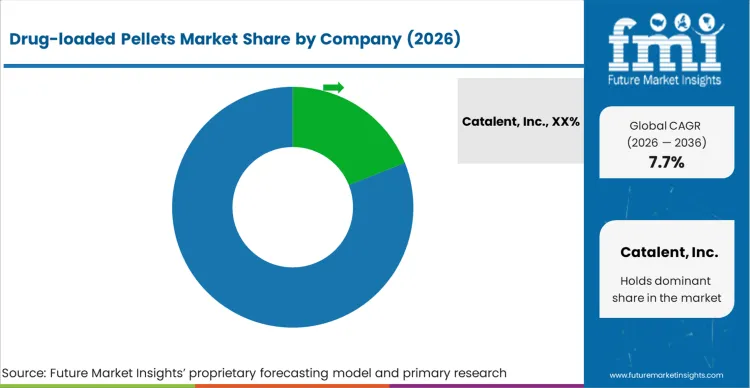
Market structure remains fragmented, yet practical competition is concentrated among a limited set of suppliers that can run validated pelletisation processes, maintain tight batch-to-batch control of drug loading and size distribution, and support regulated customers with documentation that stands up to audit and filing review. Competition is shaped by process reproducibility and scale-up success rather than short-cycle price movement, since switching suppliers can trigger revalidation work and comparability risk for modified release products. Capability across multiple manufacturing routes is a key differentiator because buyers often want a partner that can meet both development and commercial needs under one quality framework, as per FMI.
Companies with structural advantages tend to combine specialized pelletisation know-how with scalable equipment, analytical capability, and quality systems that manage variability in particle size and release performance. Suppliers that can handle sustained or controlled release profiles with consistent dissolution outcomes are better positioned because many programs require stable performance across batches and across sites. Depth in dosage form integration, including capsules and MUPS tablets, also supports account retention because it reduces downstream formulation risk for customers. Providers with broader CDMO footprints can offer redundancy, tech transfer support, and capacity planning that lowers supply risk during commercial ramps, based on FMI’s report.
Customer concentration reinforces buyer leverage. Innovator pharma and large generic manufacturers often qualify more than one supplier or internal route, maintain approved alternates, and use milestone-based contracts that tie volumes to performance and filing progress. CDMOs and CMOs compete on timelines, development support, and regulatory documentation response, which keeps pricing disciplined for standard pelletisation services. Pricing power is stronger where tighter particle size control and complex release profiles raise switching friction and increase the cost of revalidation. This procurement approach supports measured premiums mainly in programs where dissolution and size distribution are critical to bioequivalence or therapeutic performance, Future Market Insights analysis.
Recent Developments
The report includes full coverage of key trends from competitive benchmarking. Some of the recent developments covered in the reports:
| Metric | Value |
|---|---|
| Market Definition | The drug-loaded pellets market covers global production and trade of multiparticulate pellet-based drug delivery intermediates and finished-dose integrations used to achieve immediate, delayed, or controlled release profiles, where demand is shaped by oral solid dose lifecycle management, swallowability and sprinkle-use needs, and scale-up and quality control requirements across innovator, generic, and contract manufacturing settings. |
| Manufacturing Process Segmentation | Extrusion-spheronization, Drug layering on starter cores, Hot-melt extrusion pelletization, Spray congealing, Melt pelletization, Other |
| Release Profile Segmentation | Immediate release, Sustained or controlled release, Delayed or enteric release, Pulsatile release, Targeted release |
| Therapeutic Area Coverage | CNS, Cardiovascular, Gastrointestinal, Oncology, Pain or anti-inflammatory, Endocrine, Other |
| Dosage Form Integration Coverage | Capsules, Tablets (MUPS), Sachets or sprinkles, Oral suspensions, Other (implants etc.) |
| Customer Type Coverage | Innovator pharma, Generic manufacturers, CDMO or CMO, Research institutes, Veterinary pharma |
| Particle Size Range Coverage | <300 µm, 300-600 µm, 600-1000 µm, >1000 µm |
| Regions Covered | North America, Latin America, East Asia, South Asia & Pacific, Western Europe, Eastern Europe, and Middle East & Africa. |
| Countries Covered | United States, Canada, Mexico, Brazil, Argentina, Germany, France, United Kingdom, Italy, Spain, China, India, Japan, South Korea, Indonesia, Australia and 40+ countries |
| Key Companies Profiled | Catalent, Inc., Evonik Industries AG, Colorcon, Inc., Thermo Fisher Scientific Inc., Lonza Group AG, Aenova Group GmbH, Recipharm AB, Siegfried Holding AG, CordenPharma International, Glatt GmbH |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top down and bottom up market modeling validated through primary interviews with formulation teams, pellet technology suppliers, and CDMO stakeholders, supported by capacity and technology mapping and product mix triangulation, as per FMI. |
| Quantitative Units | USD 1,650.0 Mn (2026) to USD 3,450.0 Mn (2036), at a CAGR of 7.7% |
How large is the demand for Drug-Loaded Pellets in the global market in 2026?
Demand for Drug-loaded pellets in the global market is estimated to be valued at USD 1,650.0 Mn in 2026, as per FMI.
What will be the market size of Drug-Loaded Pellets in the global market by 2036?
Market size for Drug-loaded pellets is projected to reach USD 3,450.0 Mn by 2036.
What is the expected demand growth for Drug-Loaded Pellets in the global market between 2026 and 2036?
Demand for Drug-loaded pellets in the global market is expected to grow at a CAGR of 7.7% between 2026 and 2036.
Which manufacturing process is poised to lead global demand by 2026?
Extrusion-spheronization is expected to be the leading process, capturing 34% share in 2026.
Which release profile is expected to account for the largest share in 2026?
Sustained or controlled release is expected to hold the highest share at 42% in 2026.
Which dosage form integration is expected to dominate the 2026 mix?
Capsules are expected to lead dosage form integration with 46% share in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.