Therapeutic Drug Monitoring Market
The Therapeutic Drug Monitoring Market is segmented by Product, Drug Class, Sample Type, End User, Technology, and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
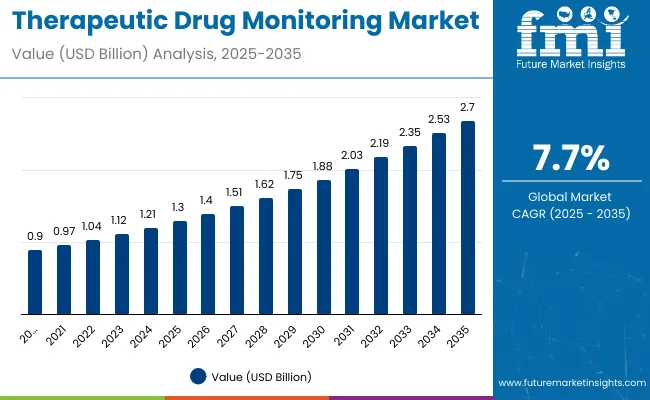
Therapeutic Drug Monitoring Market Size, Market Forecast and Outlook By FMI
The therapeutic drug monitoring market was valued at USD 1.4 billion in 2025. The market is estimated to reach USD 1.5 billion by 2026-end and expand at a CAGR of 7.5% between 2026 and 2036 to reach USD 2.9 billion by 2036. Blood is expected to hold a 75.0% sample type share, and hospitals are expected to lead the end user segment with a 48.0% share.
Summary of the Therapeutic Drug Monitoring Market
- Demand and Growth Drivers
- Narrow therapeutic index medicines increase repeat testing need as small dose changes can affect patient safety.
- Transplant treatment reinforces demand for immunosuppressant monitoring as patients need repeated drug concentration checks.
- Antiepileptic therapy keeps test use stable as dose response varies across patients and treatment settings.
- Product and Segment View
- Immunoassays are projected to capture 45.0% share of the product segment in 2026 as routine laboratories rely on automated assay menus for repeat drug concentration checks.
- By 2026, blood-based samples are anticipated to represent 75.0% of sample type demand since most therapeutic ranges are interpreted through serum and plasma results.
- Hospital laboratories are forecast to account for 48.0% of end user demand during the same year as inpatient teams need faster results for dosage review.
- Geography and Competitive Outlook
- Germany is anticipated to post 8.6% CAGR through 2036, with specialist hospitals using drug-level testing more often in transplant and neurology programs.
- Structured dose review across UK pathology networks is expected to lift the market at 8.3% CAGR by 2036.
- Companies combining immunoassay menus with chromatography capability hold stronger positions during the forecast period.
- Analyst Opinion
- Sabyasachi Ghosh, Principal Consultant for Healthcare at FMI, says, “Therapeutic drug monitoring is now routine for medicines with narrow safety margins. Hospital laboratories need fast results to support dose adjustment. Transplant and epilepsy services sustain testing volumes. Reagent use and analyzer upkeep pressure costs while assay depth reduces retesting.”
- Therapeutic Drug Monitoring Market Value Analysis
- The therapeutic drug monitoring market is shifting from selective test use to a structured clinical monitoring base.
- Safety requirements across antiepileptic drugs and immunosuppressive therapies are raising use of therapeutic drug monitoring.
- Higher use of clinical diagnostics across hospital laboratories and specialist test networks is strengthening therapeutic drug monitoring uptake.
- Test records matter more as clinicians need measured drug-level evidence before adjusting dosage within accepted treatment ranges.
Dose safety is the main buying requirement in therapeutic drug monitoring. FDA’s 2024 guidance states that small differences in dose or blood concentration can cause serious treatment failure or side effects. Laboratories need assay precision to support clinical dose decisions. Transplant follow-up adds a repeat testing layer as HRSA reported more than 48,000 USA organ transplants in 2024 with a 3.3% increase from 2023. Immunosuppressant panels are a contract priority for hospitals that manage tacrolimus and cyclosporine patients after surgery. Automation is changing supplier competition as Roche stated in December 2024 that cobas Mass Spec would offer over 60 analytes across immunosuppressant drugs and therapeutic drug monitoring. Hospitals gain a clearer path to bring selected complex assays into routine workflows. Suppliers need reliable reagent access and service coverage to convert mass spectrometry capability into repeat clinical use.
Therapeutic Drug Monitoring Market Definition
The therapeutic drug monitoring market includes laboratory tests used to measure drug concentration in patient samples. These tests help clinicians adjust dosage for medicines with narrow safety ranges. Scope includes immunoassays, radioimmunoassays, chromatography, and other analytical methods used for therapeutic dose review. General toxicology screening and routine wellness testing are outside the scope.
Therapeutic Drug Monitoring Market Inclusions
Market scope covers all commercially traded therapeutic drug monitoring products and services categorized by product, including immunoassays, radioimmunoassays, chromatography, and other analytical methods; drug class, including antiepileptic drugs, immunosuppressive drugs, cardioactive drugs, psychoactive drugs, antibiotics, and others; sample type, including blood, urine, saliva, and other specimens; end users, including hospitals, diagnostic laboratories, academic research institutes, and other clinical settings; and technologies, including conventional techniques, advanced techniques, and point-of-care technologies. Revenue scope covers 2026 to 2036. Market participants include diagnostic assay manufacturers, clinical chemistry platform providers, hospital laboratories, reference laboratories, transplant centers, neurology clinics, pharmaceutical companies, laboratory equipment suppliers, reagent distributors, healthcare providers, research institutes, and regulatory bodies.
Therapeutic Drug Monitoring Market Exclusions
Scope does not include forensic toxicology, workplace drug screening, non-therapeutic wellness tests, drug discovery bioanalysis, or clinical trial testing outside routine patient dose monitoring.
Therapeutic Drug Monitoring Market Research Methodology
- Primary Research: FMI analysts interviewed hospital laboratories, diagnostic chains, clinical chemistry specialists, and assay distributors in selected markets.
- Desk Research: FMI analysts combined data from regulatory guidance, clinical testing references, company portfolios, and diagnostic laboratory workflow indicators.
- Market sizing and forecasting: Modeled test use across products, drug classes, sample types, end users, and country-level demand groups from the bottom up.
- Data validation: FMI cross-checked every three months against treatment patterns, test menu expansion, laboratory platform use, and company service coverage.
Why is the Therapeutic Drug Monitoring Market Growing?
- Hospitals are using therapeutic drug monitoring to reduce dose-related safety risk in high-risk medicines.
- Transplant programs need immunosuppressant testing as drug levels affect rejection risk and toxicity.
- Neurology care uses antiepileptic monitoring as seizure control and adverse effects vary by patient.
Patient safety concerns are keeping therapeutic drug monitoring important for medicines with tight treatment windows. FDA stated in February 2024 that narrow therapeutic index drugs can cause serious treatment failure or side effects from small concentration changes. Therapeutic monitoring is used to measure drug amounts in blood and guide safer dosing decisions. Therapeutic monitoring keeps testing value close to clinical risk instead of general diagnostic volume. Laboratories with stable assay controls can protect clinicians from delayed or unclear dose decisions.
Epilepsy care creates a recurring testing base for antiepileptic drug monitoring. CDC reported in May 2024 that about 2.9 million USA adults had active epilepsy during 2021 and 2022. Large treated populations increase the need for test methods to guide dosage checks during long-term therapy. Immunoassay platforms and neurology immunoassay workflows help laboratories manage repeat testing with controlled turnaround. Vendors with dependable reagent supply and clear calibration records can gain repeat hospital contracts.
Market Segmentation Analysis
- Immunoassays are projected to capture 45.0% of product revenue in 2026 as routine hospital laboratories rely on automated assay menus for repeat drug-level checks.
- Transplant care keeps immunosuppressive drugs central to monitored therapy, with 25.0% share forecast for the drug class segment by 2026.
- Blood-based samples are anticipated to hold 75.0% share as serum and plasma results guide most therapeutic range decisions.
- Hospitals are forecast to contribute 48.0% of end user revenue during 2026 since inpatient teams need faster results for dosage review.
- Conventional techniques are expected to represent 46.0% technology share as established immunoassay workflows already match laboratory staffing and controls.
The market for therapeutic drug monitoring is divided into five primary segment groups based on product, drug class, sample type, end user, and technology. Product includes immunoassays, radioimmunoassays, chromatography, and other analytical methods. Drug class includes antiepileptic drugs, immunosuppressive drugs, cardioactive drugs, psychoactive drugs, antibiotics, and others. Sample type includes blood, urine, saliva, and others. End user includes hospitals, diagnostic laboratories, academic research institutes, and others. Technology includes conventional techniques, advanced techniques, and point-of-care technologies.
Insights into Therapeutic Drug Monitoring Market by Product
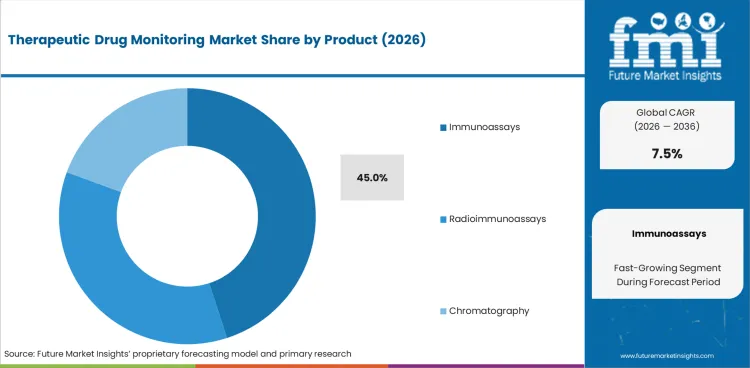
- In 2026, immunoassays are expected to make up 45.0% of product demand across hospital and diagnostic laboratory workflows. Automated assay menus help clinical teams receive repeatable drug concentration results during dose review and therapy adjustment.
- Chromatography and radioimmunoassays address selected complex cases needing higher method control. Their value improves in low-concentration testing and panels requiring stronger analyte separation.
Insights into Therapeutic Drug Monitoring Market by Drug Class
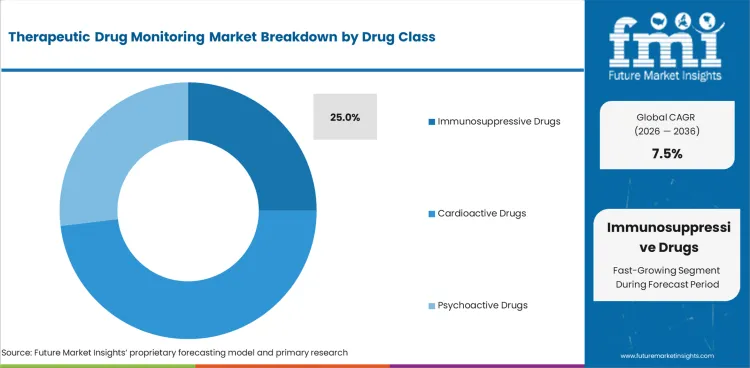
- Immunosuppressive drugs are projected to account for 25.0% of drug class segment in 2026. Transplant programs need repeated drug concentration checks to manage rejection risk and toxicity risk after surgery.
- Antiepileptic drugs serve a steady monitoring base across neurology care. Their use increases in long-term treatment programs requiring dosage review and patient response tracking.
Insights into Therapeutic Drug Monitoring Market by Sample Type
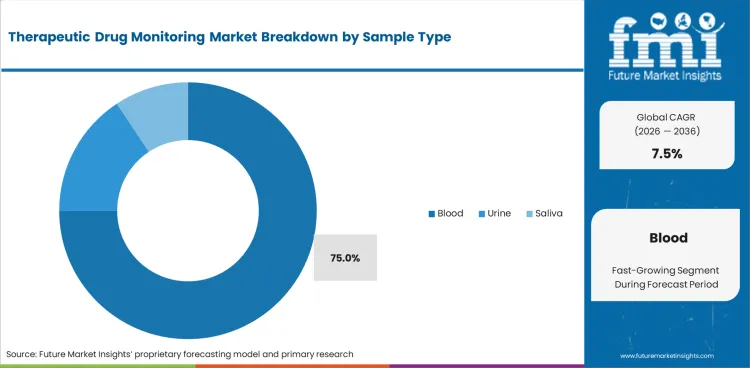
- Blood is estimated to account for 75.0% of sample type segment in 2026. Serum and plasma samples provide the main reference base for therapeutic range interpretation across monitored medicines.
- Urine and saliva options address selected monitoring needs in specific care settings. Their use improves in cases requiring easier collection and noninvasive sample handling.
Insights into Therapeutic Drug Monitoring Market by End User
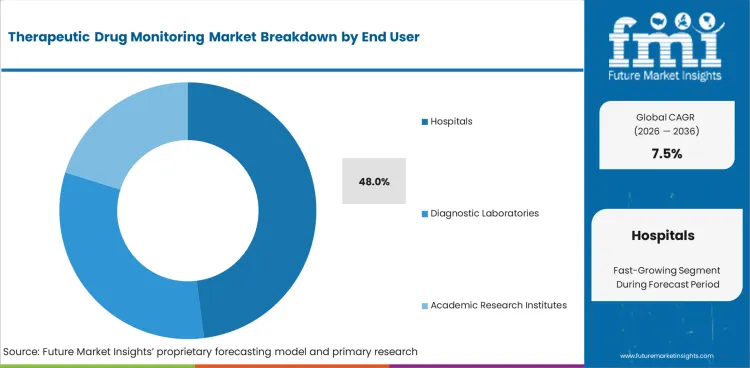
- In 2026, hospitals are projected to hold 48.0% share of end user segment. Inpatient teams need faster access to test results during acute care and specialist dose adjustment.
- Diagnostic laboratories serve outsourced panels and complex analytical work for healthcare providers. Academic research institutes contribute through pharmacokinetic studies and method development activities.
Insights into Therapeutic Drug Monitoring Market by Technology
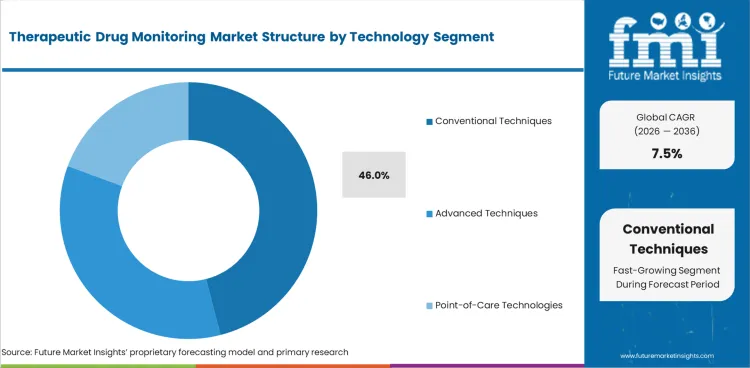
- Conventional techniques are expected to represent 46.0% share of technology demand in 2026. Established immunoassay and chromatography workflows help laboratories manage testing volume with trained staff and validated controls.
- Advanced techniques serve complex panels requiring higher sensitivity and stronger analyte separation. Point-of-care technologies address selected bedside needs requiring faster clinical review and shorter turnaround time.
Therapeutic Drug Monitoring Market Drivers, Restraints, and Opportunities
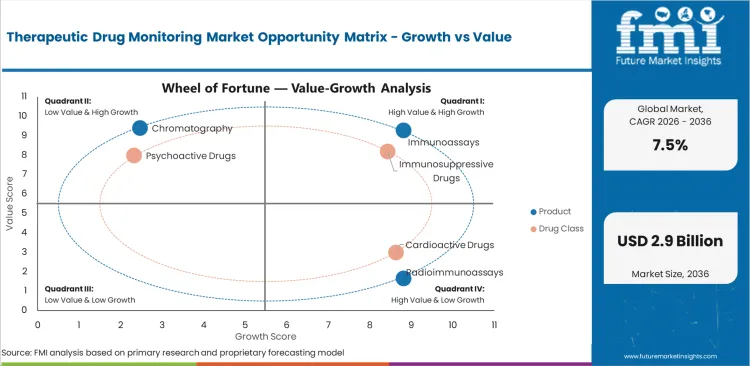
- Narrow therapeutic index medicines are creating structured demand for drug concentration testing across hospitals.
- Reagent cost and analyzer service needs increase pricing pressure for smaller diagnostic laboratories.
- Automated mass spectrometry improves service opportunity through multi-drug testing and better analyte separation.
Therapeutic drug monitoring continues to expand as hospitals need measured drug-level results for safer dosage decisions. Reagent cost and analyzer maintenance can slow higher use across smaller laboratories. Use continues through immunoassay platforms and transplant monitoring programs.
Labor Tightness Is Supporting Higher Test Charges
TDM pricing faces pressure from skilled-labor availability because many drug-level assays require validated workflows, quality control, instrument maintenance, and interpretation-ready reporting. According to the BLS August 2025 report, clinical laboratory technologists and technicians had a median annual wage of USD 61,890 in May 2024, with 22,600 openings projected each year from 2024 to 2034 despite only 2% employment growth. Labor availability matters for TDM because low-volume or urgent assays cannot always absorb labor costs through automation alone. Labs are more likely to defend rush fees and minimum test charges for transplant and infectious disease drug monitoring panels.
Medicare Payment Rules Are Holding TDM Price Floors
Therapeutic drug monitoring pricing is anchored by reimbursement stability instead of broad test inflation. In May 2026, CMS stated that the current CLFS reporting period runs from May 1, 2026 to July 31, 2026, using private payer data from January 1, 2025 to June 30, 2025. CMS confirmed no phase-in payment reduction in 2026, and any reductions from 2027 to 2029 cannot exceed 15% per year. Therapeutic drug monitoring assays such as tacrolimus and sirolimus testing support steadier hospital and reference lab pricing due to delayed reimbursement resets. Commercial buyers should expect negotiated prices to stay disciplined but not collapse because Medicare floors continue to influence payer contracting.
Analysis of Therapeutic Drug Monitoring Market By Key Countries
.webp)
| Country | CAGR |
|---|---|
| Germany | 8.6% |
| UK | 8.3% |
| USA | 8.0% |
| China | 7.9% |
| India | 7.3% |
| Thailand | 7.2% |
| Brazil | 5.3% |
Source: Future Market Insights, 2026.
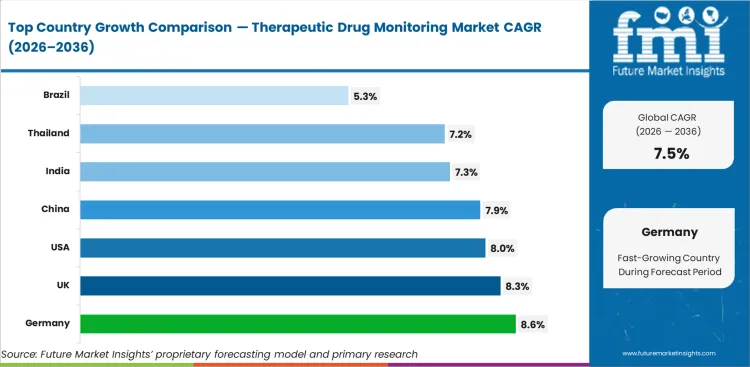
Therapeutic Drug Monitoring Market CAGR Analysis By Country
- Germany records the strongest listed forecast at 8.6% CAGR through 2036 as specialist hospitals deepen drug-level testing for transplant follow-up and neurology care.
- Structured pathology networks place the UK at 8.3% CAGR by 2036 with stronger use of hospital-based dosage review.
- Large treated patient pools and advanced clinical chemistry capacity are expected to lift USA demand at 8.0% CAGR by 2036.
- China is projected at 7.9% CAGR as hospital capacity expands and transplant care leads to a stronger need for immunosuppressant monitoring.
- India is projected at 7.3% CAGR as private hospitals widen monitoring access.
- Thailand is expected to record 7.2% CAGR by 2036 as specialist care supports repeat testing.
- Brazil is forecast at 5.3% CAGR over the forecast period as public transplant programs sustain monitored therapy use.
Demand for therapeutic drug monitoring is forecast to rise at 7.5% CAGR from 2026 to 2036. Country-level analysis covers the major markets contributing to this forecast, with the key markets listed below.
Demand Outlook for Therapeutic Drug Monitoring Market in Germany
Therapeutic drug monitoring industry in Germany is projected to record a CAGR of 8.6% through 2036. DSO reported 3,701 organs transplanted in Germany in 2024. The transplant base increases repeat need for immunosuppressant concentration testing after surgery and during long-term follow-up. Local laboratories can gain from hospitals that require stable assay performance and faster clinical reporting.
- Specialist hospital laboratories boost demand for immunosuppressant testing and antiepileptic drug monitoring.
- Transplant follow-up increases repeat testing need across tacrolimus and cyclosporine therapy.
- Advanced laboratory infrastructure favors chromatography and mass spectrometry use in complex cases.
Sales Analysis of Therapeutic Drug Monitoring Market in the United Kingdom
The therapeutic drug monitoring sector in the UK is expected to expand at a CAGR of 8.3% during the assessment period. NHS Blood and Transplant reported 4,581 organ transplants carried out during 2024 to 2025. The transplant base supports demand for UK therapeutic drug monitoring as transplant patients need ongoing immunosuppressant review. Hospital laboratories benefit from clear clinical pathways and centralized test governance.
- Hospital pathology networks help routine monitoring programs serve inpatient and outpatient care.
- Transplant aftercare increases demand for immunosuppressant testing and consistent result reporting.
- Automated mass spectrometry can serve complex panels in large regional laboratory settings.
Demand Outlook for Therapeutic Drug Monitoring Market in the United States
The USA therapeutic drug monitoring industry is forecast to grow at a CAGR of 8.0% by 2036. Organ transplants in the United States exceeded 48,000 in 2024, according to HRSA’s January 2025 report. The clinical base raises repeated use of immunosuppressant monitoring and hospital dose review. Vendors with validated assays and service coverage can serve both hospital laboratories and reference laboratories.
- Large transplant activity enhances demand for immunosuppressant drug monitoring across follow-up care.
- Epilepsy treatment enhances repeat testing use for antiepileptic drugs across neurology settings.
- Advanced analytical laboratories favor LC-MS workflows for multi-drug and metabolite testing.
Opportunity Analysis of Therapeutic Drug Monitoring Market in China
Therapeutic drug monitoring industry in China is expected to advance at 7.9% CAGR during the assessment period. National Health Commission rules published in 2024 required transplant institutions to show at least 10 successful deceased donation cases each year for two consecutive years or over 25 cases across two years. The requirements raise the importance of organized transplant programs and reliable post-transplant monitoring. Local laboratories need assay systems to fit hospital scale and specialist drug review.
- Hospital expansion favors broader use of drug concentration testing across tertiary care settings.
- Transplant regulation favors stronger need for structured follow-up and immunosuppressant monitoring.
- Local diagnostic suppliers can compete on reagent availability and platform service coverage.
Future Outlook for Therapeutic Drug Monitoring Market in India
India’s therapeutic drug monitoring industry is projected to rise at 7.3% CAGR by 2036. According to the NOTTO report, India reached 18,911 organ transplants in 2024. The clinical base lifts demand for therapeutic drug monitoring in India as immunosuppressant use expands with transplant care. Larger private hospitals and tertiary centers need test platforms to reduce send-out testing delays.
- Transplant activity improves demand for tacrolimus and cyclosporine monitoring across specialist hospitals.
- Private hospital networks raise the demand for faster in-house test turnaround.
- Diagnostic chains can add monitoring panels as chronic disease care and specialist treatment expand.
Demand Outlook for Therapeutic Drug Monitoring Market in Thailand
Thailand’s therapeutic drug monitoring industry is forecast at 7.2% CAGR from 2026 to 2036. Thai Transplant Society continues to publish annual transplant registry coverage, including its 2024 reporting structure for organ transplantation activity. Specialist hospitals make up the main base for immunosuppressant testing and antiepileptic monitoring. Laboratories with reliable sample handling and reference ranges can serve transplant centers and tertiary care networks.
- Specialist hospital networks support demand for immunosuppressant and antiepileptic monitoring.
- Kidney transplant follow-up raises repeat testing need across long-term patient management.
- Reference laboratories can gain from complex cases that need chromatography-based methods.
Sales Analysis of Therapeutic Drug Monitoring Market in Brazil
Brazil’s therapeutic drug monitoring industry is expected to post 5.3% CAGR through 2036. Brazil’s Ministry of Health reported in June 2025 that more than 30,000 transplants were performed through SUS in 2024. The public system base supports demand for therapeutic drug monitoring in transplant follow-up and hospital care. Budget control keeps the market focused on dependable methods and manageable reagent cost.
- Public transplant coverage creates repeat need for immunosuppressant monitoring across SUS care.
- Hospital laboratories need cost-controlled assays to serve high-volume patient groups.
- Reference laboratories can serve specialized testing needs in complex drug panels.
Competitive Landscape and Strategic Positioning
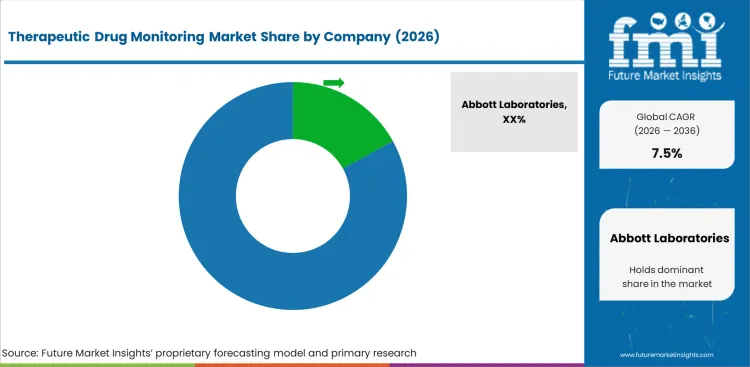
- Abbott Laboratories competes from a strong immunoassay base and long clinical laboratory relationships across routine monitoring accounts.
- Roche and Siemens Healthineers use automated analyzer systems for repeated drug concentration testing in hospital laboratories.
- Specialist providers concentrate on chromatography kits and mass spectrometry workflows for complex therapeutic panels needing higher method control.
Competition in therapeutic drug monitoring is shaped by assay menu depth and laboratory workflow strength across routine and complex testing. Abbott Laboratories holds a strong position through routine immunoassay systems and established hospital account access. Roche brings automation depth through clinical chemistry platforms and mass spectrometry expansion for higher-volume laboratories. Siemens Healthineers emphasizes analyzer workflow and laboratory integration across hospital testing settings. Thermo Fisher Scientific strengthens advanced testing through mass spectrometry systems used in complex therapeutic panels. Danaher adds chromatography capability for laboratories managing specialized drug monitoring and analytical testing needs. Randox Laboratories and Bio-Rad Laboratories support laboratories through specialist test workflows and quality control products.
Assay validation cost and skilled staff needs raise entry barriers across complex analytical methods. Competitive priorities center on broader test menus and faster reporting across long treatment cycles. Hospitals prefer partners offering reliable drug concentration results and dependable service coverage for dosage review. Companies with broad assay coverage and stable instrument access are best placed to reduce coordination burden for clinical laboratories.
Key Companies in the Therapeutic Drug Monitoring Market
Major companies active in the therapeutic drug monitoring market include:
- Abbott Laboratories, Thermo Fisher Scientific Inc., and F. Hoffmann-La Roche Ltd. have strong testing infrastructure for routine and advanced therapeutic monitoring.
- Siemens Healthineers AG, Bio-Rad Laboratories Inc., and Danaher Corporation serve laboratories through analyzer systems, reagents, and analytical platforms.
- Randox Laboratories Ltd., Beckman Coulter Inc., and Chromsystems Instruments & Chemicals GmbH hold specialist positions across quality control, clinical chemistry, and LC-MS testing kits.
Competitive Benchmarking: Therapeutic Drug Monitoring Market
| Company | Assay Menu Depth | Analyzer Workflow Coverage | Advanced Analytical Capability | Geographic Footprint |
|---|---|---|---|---|
| Abbott Laboratories | High | High | Medium | Broad global presence across over 160 countries with strong access across North America, Europe, Asia Pacific, and Latin America. |
| Thermo Fisher Scientific Inc. | High | Medium | Strong | Global operating reach across the Americas, APAC, and EMEA with strong laboratory instrument placement in advanced testing markets. |
| F. Hoffmann-La Roche Ltd. | High | High | Strong | Broad diagnostics presence through worldwide locations and regional sites across over 70 countries. |
| Siemens Healthineers AG | High | High | Medium | Global healthcare presence across major regions with country operations in the USA, Europe, China, India, and other diagnostic markets. |
| Bio-Rad Laboratories Inc. | Medium | Medium | Medium | Global clinical diagnostics reach with offices and manufacturing presence across the Americas, EMEA, and Asia Pacific. |
| Danaher Corporation | High | High | Strong | Operates across 50 countries and 700 global locations through diagnostics, life sciences, and biotechnology businesses. |
| Randox Laboratories Ltd. | Medium | Medium | Medium | International diagnostics footprint with offices in the UK, USA, China, Czech Republic, France, and other markets. |
| Chromsystems Instruments & Chemicals GmbH | High | Medium | Strong | Germany-based specialist supplier serving laboratories worldwide through HPLC and LC-MS/MS diagnostic product coverage. |
Source: Future Market Insights, 2026.
Key Developments in Therapeutic Drug Monitoring Market
- In December 2024, Roche announced CE mark approval for the cobas Mass Spec solution. The platform launch included mass spectrometry automation and planned menu expansion for immunosuppressant drugs and therapeutic drug monitoring.
- In June 2024, Thermo Fisher Scientific introduced the Stellar mass spectrometer for clinical research and targeted quantitation workflows. The product launch strengthens advanced analytical capacity for laboratories handling complex drug panels.
Key Players in the Therapeutic Drug Monitoring Market
Major Global Players
- Abbott Laboratories
- Thermo Fisher Scientific Inc.
- F. Hoffmann-La Roche Ltd.
- Siemens Healthineers AG
- Bio-Rad Laboratories Inc.
- Danaher Corporation
- Beckman Coulter Inc.
Specialist Players
- Randox Laboratories Ltd.
- Chromsystems Instruments & Chemicals GmbH
- Specialty LC-MS Kit Providers
Report Scope and Coverage
| Parameter | Details |
|---|---|
| Quantitative Units | USD 1.4 billion to USD 2.9 billion, at a CAGR of 7.5% |
| Market Definition | The therapeutic drug monitoring market includes laboratory tests used to measure drug concentration for dose adjustment and patient safety review. |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | USA, UK, Germany, China, India, Thailand, Brazil, Japan, South Korea, and 30 plus countries |
| Key Companies Profiled | Abbott Laboratories, Thermo Fisher Scientific Inc., F. Hoffmann-La Roche Ltd., Siemens Healthineers AG, Bio-Rad Laboratories Inc., Danaher Corporation, Randox Laboratories Ltd., Chromsystems Instruments & Chemicals GmbH |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down methodology starting with test demand and verified clinical use patterns. |
Source: Future Market Insights, 2026.
Therapeutic Drug Monitoring Market by Segments
By Product
- Immunoassays
- Radioimmunoassays
- Chromatography
- Other Analytical Methods
By Drug Class
- Antiepileptic Drugs
- Immunosuppressive Drugs
- Cardioactive Drugs
- Psychoactive Drugs
- Antibiotics
- Others
By Sample Type
- Blood
- Urine
- Saliva
- Others
By End User
- Hospitals
- Diagnostic Laboratories
- Academic Research Institutes
- Others
By Technology
- Conventional Techniques
- Advanced Techniques
- Point-of-Care Technologies
Therapeutic Drug Monitoring Market by Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Europe
- Germany
- UK
- Italy
- Spain
- France
- Rest of Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East and Africa
- Saudi Arabia
- Other GCC Countries
- South Africa
- Rest of Middle East and Africa
Research Sources and Bibliography
- Centers for Medicare & Medicaid Services. (2026, May 1). Clinical Laboratory Fee Schedule. USA Department of Health & Human Services.
- USA Bureau of Labor Statistics. (2025, August 28). Clinical Laboratory Technologists and Technicians: Occupational Outlook Handbook. USA Department of Labor.
- USA Food and Drug Administration. (2024, February). Setting and implementing standards for narrow therapeutic index drugs.
- Centers for Disease Control and Prevention. (2024, May). Epilepsy facts and stats.
- Health Resources and Services Administration. (2025, January). Organ transplants exceeded 48,000 in 2024.
- Deutsche Stiftung Organtransplantation. (2025, April). DSO-Jahresbericht 2024.
- NHS Blood and Transplant. (2025, July). Annual report and accounts 2024 to 2025.
- National Organ and Tissue Transplant Organization. (2025, August). Annual report 2024-25.
- National Health Commission of the People’s Republic of China. (2024, June). Human organ transplant medical institution management requirements.
- Ministério da Saúde. (2025, June). Brasil bate recorde de transplantes e anuncia medidas para modernizar sistema e aumentar doações.
- Roche. (2024, December). Roche transforms mass spectrometry diagnostics with launch of cobas Mass Spec solution.
- Thermo Fisher Scientific. (2024, June). Thermo Fisher Scientific introduces innovative mass spectrometer to advance clinical research.
This Report Answers
- What is the current and future size of the therapeutic drug monitoring market?
- How fast is the therapeutic drug monitoring market expected to expand between 2026 and 2036?
- Which product segment is expected to lead the market by 2026?Which sample type is expected to account for the strongest demand by 2026?
- What factors are driving demand for therapeutic drug monitoring globally?
- How are narrow therapeutic index drugs influencing clinical test use?
- Why are hospitals the main demand base for therapeutic drug monitoring?
- How are advanced analytical methods creating demand for specialized drug monitoring?
- Which countries are projected to record faster expansion through 2036?
- What is driving market expansion in Germany and the UK?
- Who are the key companies active in the therapeutic drug monitoring market?
- How does FMI estimate and validate the market forecast?
Frequently Asked Questions
What is the global market demand for Therapeutic Drug Monitoring in 2026?
In 2026, the global market for therapeutic drug monitoring is expected to be worth nearly USD 1.4 billion.
How big will the market for Therapeutic Drug Monitoring be in 2036?
By 2036, the therapeutic drug monitoring market is expected to be worth USD 2.9 billion.
How much is demand for Therapeutic Drug Monitoring expected to expand between 2026 and 2036?
Between 2026 and 2036, demand for therapeutic drug monitoring is expected to expand at a CAGR of 7.5%.
Which product segment is likely to be the leading product globally by 2026?
Immunoassays will make up 45.0% of the product segment in 2026.
What is causing demand to rise in Germany?
The Germany therapeutic drug monitoring market is projected to record 8.6% CAGR through 2036, backed by specialist hospital testing and transplant follow-up.
What is causing demand to rise in the UK?
The UK therapeutic drug monitoring market is projected to expand at 8.3% CAGR through 2036, backed by hospital pathology networks and transplant care.
What does this report mean by Therapeutic Drug Monitoring Market definition?
The therapeutic drug monitoring market includes laboratory testing used to measure drug concentration for dose adjustment and patient safety review.
How does FMI make the Therapeutic Drug Monitoring forecast and check it?
Forecasting models use a hybrid bottom-up and top-down approach, starting with test demand and checking it against clinical use patterns.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Restraints
- Opportunity
- Trends
- Desk Research Programme (Secondary Evidence)
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product , 2026 to 2036
- Immunoassays
- Radioimmunoassays
- Chromatography
- Immunoassays
- Y to o to Y Growth Trend Analysis By Product , 2021 to 2025
- Absolute $ Opportunity Analysis By Product , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Drug Class
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Drug Class, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Drug Class, 2026 to 2036
- Immunosuppressive Drugs
- Cardioactive Drugs
- Psychoactive Drugs
- Immunosuppressive Drugs
- Y to o to Y Growth Trend Analysis By Drug Class, 2021 to 2025
- Absolute $ Opportunity Analysis By Drug Class, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Sample Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Sample Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Sample Type, 2026 to 2036
- Blood
- Urine
- Saliva
- Blood
- Y to o to Y Growth Trend Analysis By Sample Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Sample Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospitals
- Diagnostic Laboratories
- Academic Research Institutes
- Hospitals
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technology, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technology, 2026 to 2036
- Conventional Techniques
- Advanced Techniques
- Point-of-Care Technologies
- Conventional Techniques
- Y to o to Y Growth Trend Analysis By Technology, 2021 to 2025
- Absolute $ Opportunity Analysis By Technology, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Drug Class
- By Sample Type
- By End User
- By Technology
- Competition Analysis
- Competition Deep Dive
- Abbott Laboratories
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Thermo Fisher Scientific Inc.
- F. Hoffmann-La Roche Ltd.
- Siemens Healthineers AG
- Bio-Rad Laboratories Inc.
- Danaher Corporation
- Beckman Coulter Inc.
- Abbott Laboratories
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Sample Type, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Sample Type, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Sample Type, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Sample Type, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Sample Type, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Sample Type, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Sample Type, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Sample Type, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Technology, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product
- Figure 6: Global Market Value Share and BPS Analysis by Drug Class, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Drug Class, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Drug Class
- Figure 9: Global Market Value Share and BPS Analysis by Sample Type, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Sample Type, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Sample Type
- Figure 12: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by End User
- Figure 15: Global Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Technology
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Product
- Figure 32: North America Market Value Share and BPS Analysis by Drug Class, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Drug Class, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Drug Class
- Figure 35: North America Market Value Share and BPS Analysis by Sample Type, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Sample Type, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Sample Type
- Figure 38: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by End User
- Figure 41: North America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Technology
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Product
- Figure 48: Latin America Market Value Share and BPS Analysis by Drug Class, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Drug Class, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Drug Class
- Figure 51: Latin America Market Value Share and BPS Analysis by Sample Type, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Sample Type, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Sample Type
- Figure 54: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by End User
- Figure 57: Latin America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Technology
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Product
- Figure 64: Western Europe Market Value Share and BPS Analysis by Drug Class, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Drug Class, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Drug Class
- Figure 67: Western Europe Market Value Share and BPS Analysis by Sample Type, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Sample Type, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Sample Type
- Figure 70: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by End User
- Figure 73: Western Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Technology
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Product
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Drug Class, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Drug Class, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Drug Class
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Sample Type, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Sample Type, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Sample Type
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by End User
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Technology
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Product
- Figure 96: East Asia Market Value Share and BPS Analysis by Drug Class, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Drug Class, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Drug Class
- Figure 99: East Asia Market Value Share and BPS Analysis by Sample Type, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Sample Type, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Sample Type
- Figure 102: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by End User
- Figure 105: East Asia Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Technology
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Drug Class, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Drug Class, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Drug Class
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Sample Type, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Sample Type, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Sample Type
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Technology
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Drug Class, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Drug Class, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Drug Class
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Sample Type, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Sample Type, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Sample Type
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Technology
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

