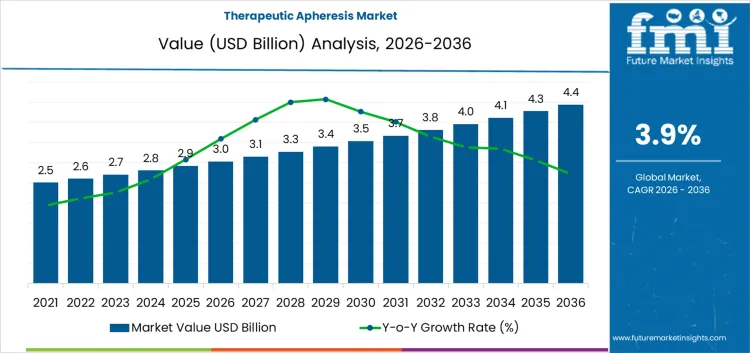
The therapeutic apheresis market is valued at USD 3.0 billion in 2026 and projected to reach USD 4.4 billion by 2036 at a CAGR of 3.9%. Value behavior reflects reliance on procedure-based clinical interventions embedded within hospital and tertiary care treatment protocols. Spending is anchored to disease-specific indications where apheresis serves as a non-substitutable therapeutic option. Capital allocation favors installed base expansion and consumable utilization rather than rapid site proliferation. Revenue concentration remains linked to plasma exchange and related procedures requiring repeat treatment cycles. Procurement patterns prioritize system reliability, regulatory compliance, and compatibility with established transfusion and dialysis infrastructure, reinforcing stable but measured value growth.
Structural and operational constraints define scalability. High per-procedure cost, driven by disposable kits and specialized staffing, limits expansion beyond high-acuity care centers. Reimbursement variability across indications influences procedural volumes and budget planning. Clinical guidelines and evidence thresholds regulate utilization intensity, constraining discretionary adoption. Equipment utilization depends on scheduling capacity and trained personnel availability. Regulatory oversight for blood handling and extracorporeal circulation elevates compliance burden and slows rapid deployment. Geographic value concentration aligns with tertiary hospital density, transplant program activity, and specialist access. Growth persistence depends on protocol entrenchment within chronic and acute disease management pathways rather than broad-based outpatient diffusion.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 3.0 billion |
| Market Forecast Value (2036) | USD 4.4 billion |
| Forecast CAGR 2026 to 2036 | 3.9% |
Demand for therapeutic apheresis solutions is increasing as clinicians seek targeted interventions to remove pathological components from blood in autoimmune, hematologic, and metabolic disorders. Healthcare providers specify apheresis procedures to manage conditions such as Guillain Barré syndrome, thrombotic thrombocytopenic purpura, and severe hyperlipidemia where conventional therapies do not sufficiently modify disease activity. Hospitals and specialty clinics require equipment and disposables that support plasma exchange, leukapheresis, and immunoadsorption with consistent performance and clinical safety. Procurement teams prioritize systems that deliver reproducible separation efficiency, user-friendly interfaces, and robust quality control measures to support procedural efficacy and operator confidence.
Regulatory frameworks for blood management and device safety reinforce demand for certified platforms that integrate with clinical workflows in critical care and outpatient settings. Expansion of diagnostic capabilities and early disease detection is reinforcing uptake of therapeutic apheresis as part of personalized treatment strategies. Healthcare administrators allocate resources to support multidisciplinary care pathways that incorporate apheresis in treatment plans for complex disease states. Advances in disposable kit design and extracorporeal circuit technology support procedural efficiency and reduce risk of adverse events. Training for clinical staff on protocol adherence and patient monitoring enhances operational readiness. These factors are contributing to sustained demand growth for therapeutic apheresis in evolving clinical practice environments.
Demand for therapeutic apheresis is shaped by rising prevalence of immune-mediated disorders, hematologic conditions, and critical care interventions. Clinical adoption reflects need for extracorporeal blood processing supporting rapid removal of pathological components. Treatment protocols emphasize precision, patient safety, and repeatability across acute and chronic care settings. Infrastructure availability and trained personnel influence procedural volume distribution. Segment classification reflects differentiation by procedure complexity and care delivery environment. Structure highlights how procedural focus and end-user setting determine utilization intensity, equipment deployment, and workflow integration across hospital-centered and specialized treatment frameworks.
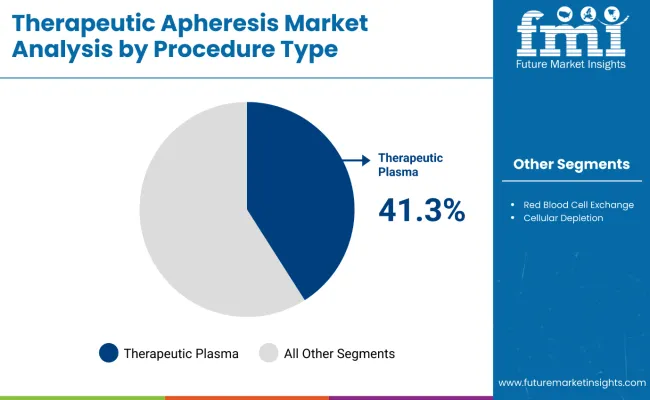
Therapeutic plasma exchange holds 41.3%, representing the largest share among therapeutic apheresis procedures due to broad clinical applicability. TPE is used across neurological, renal, and autoimmune conditions requiring removal of circulating antibodies or toxins. Protocol standardization supports consistent adoption across treatment centers. Procedural flexibility allows application in both acute crisis management and scheduled therapy regimens. Red blood cell exchange, cellular depletions, and other procedures address narrower indications with lower procedural frequency. Procedure segmentation reflects dominance of plasma-based intervention where treatment efficacy and protocol familiarity support sustained clinical utilization.
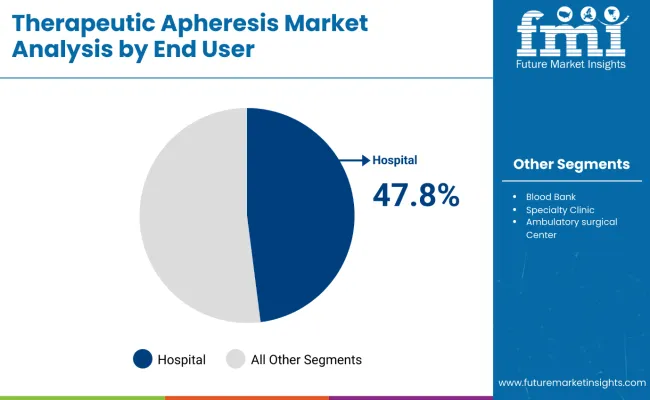
Hospitals account for 47.8%, representing the largest share among end-user settings due to concentration of specialized care services. Inpatient environments support complex apheresis procedures requiring multidisciplinary oversight. Access to intensive care units and transfusion services enables management of high-risk patients. Centralized procurement and scheduling infrastructure sustain higher procedure volumes. Blood banks, specialty clinics, and ambulatory surgical centers perform apheresis within defined scopes and lower aggregate intensity. End-user segmentation reflects concentration of advanced therapeutic capability within hospital settings managing diverse and severe clinical conditions.
Demand for therapeutic apheresis reflects clinical requirements for selective removal of pathogenic blood components in acute and chronic disease management. Adoption spans hospitals, specialty clinics, and blood centers supporting neurology, nephrology, hematology, and autoimmune care. Global scope aligns with protocol-driven interventions requiring extracorporeal circulation and precise plasma or cellular separation. Usage integrates with regulated clinical workflows, trained personnel availability, and device-supported treatment standardization.
How do disease-specific treatment protocols and clinical effectiveness shape adoption?
Therapeutic apheresis supports conditions where rapid reduction of circulating antibodies, lipoproteins, or cellular elements improves patient outcomes. Demand increases across autoimmune disorders, thrombotic microangiopathies, and transplant-related indications where pharmacologic therapy alone shows limited efficacy. Clinicians rely on apheresis for rapid symptom control and crisis intervention. Standardized treatment guidelines support repeat procedures over defined treatment cycles. Compatibility with existing dialysis infrastructure facilitates adoption within nephrology units. Advances in membrane technology and adsorption columns improve selectivity and treatment tolerability. Multidisciplinary clinical acceptance strengthens utilization across specialty departments. Adoption reflects protocol necessity rather than elective therapeutic choice.
How do cost intensity and operational complexity influence market scalability?
Procedures require high-cost equipment, disposable kits, and trained clinical staff, increasing per-treatment expense. Demand sensitivity rises where reimbursement coverage varies by indication and payer policy. Scheduling constraints limit throughput due to procedure duration and equipment availability. Consumable dependency increases recurring operational cost. Infection control and anticoagulation management raise procedural risk considerations. Regulatory oversight for blood handling adds compliance burden. Limited specialist availability restricts service expansion beyond tertiary care centers. Capital allocation priorities within hospitals constrain rapid scaling of apheresis capacity across broader care networks.
Demand for therapeutic apheresis is expanding globally due to increasing diagnosis of immune-mediated, neurological, and hematological disorders requiring extracorporeal blood treatment. Utilization growth reflects broader clinical acceptance of plasma exchange and cell separation therapies within tertiary care settings. Expansion of transplant programs and critical care services supports procedural volume growth. Technology improvements enhance safety and procedural efficiency. Growth rates in India at 4.4%, China at 4.0%, South Korea at 3.8%, USA at 3.7%, and Germany at 3.6% indicate sustained expansion shaped by healthcare access, specialist availability, and treatment protocol adoption.
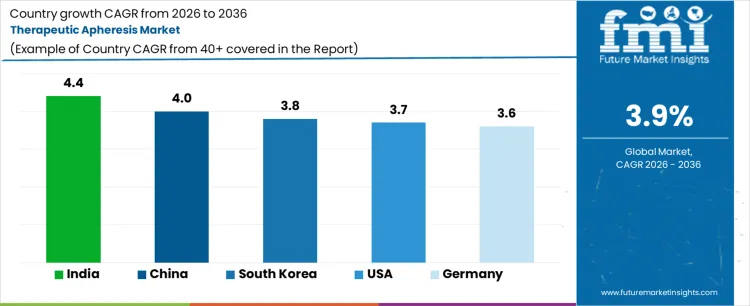
| Country | CAGR (%) |
|---|---|
| India | 4.4% |
| China | 4.0% |
| South Korea | 3.8% |
| USA | 3.7% |
| Germany | 3.6% |
Expanding tertiary care capacity is influencing therapeutic apheresis demand in India, growing at a CAGR of 4.4%. Rising diagnosis of autoimmune and neurological conditions increases referral volumes for plasma exchange therapy. Growth of transplant centers supports demand for pre- and post-transplant apheresis procedures. Private hospital investment improves access to specialized extracorporeal therapies. Limited availability outside major cities moderates broader adoption. Training programs for clinicians and technicians support gradual procedural expansion within high-acuity hospitals.
Hospital-based specialty care expansion underpins therapeutic apheresis demand in China, growing at a CAGR of 4.0%. Large tertiary hospitals integrate apheresis into treatment protocols for hematological and immune conditions. Rising critical care capacity supports procedural utilization. Standardization of clinical guidelines improves adoption consistency. Reimbursement variability influences selective usage across regions. Urban healthcare concentration drives most procedural volume, with limited penetration in lower-tier facilities.
Clinical protocol alignment shapes therapeutic apheresis demand in South Korea, expanding at a CAGR of 3.8%. Neurology and transplant centers routinely incorporate plasma exchange into treatment pathways. Strong clinician training supports procedural consistency and safety. High hospital technology adoption enables efficient treatment delivery. National health coverage supports access while controlling utilization growth. Demand remains stable, driven by evidence-based application rather than procedural expansion.
Specialized clinical application sustains therapeutic apheresis demand in the USA, growing at a CAGR of 3.7%. Established use across hematology, neurology, and transplant medicine supports baseline procedural volumes. Reimbursement scrutiny influences selective application aligned with clinical guidelines. Academic medical centers account for significant usage. Equipment replacement and protocol refinement drive ongoing investment. Growth remains measured due to mature clinical pathways and cost containment priorities.
Regulated clinical practice defines therapeutic apheresis demand in Germany, expanding at a CAGR of 3.6%. Treatment protocols emphasize evidence-based indications and standardized application. Hospitals integrate apheresis within comprehensive care pathways for immune and hematological conditions. Strong reimbursement governance moderates procedural growth. High-quality equipment standards sustain replacement demand. Adoption remains consistent, driven by clinical necessity rather than procedural expansion.
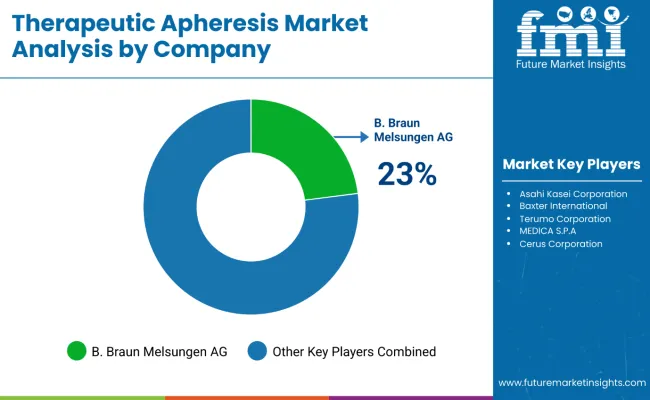
Demand for therapeutic apheresis is driven by rising prevalence of autoimmune disorders, hematological conditions, and indications such as thrombotic thrombocytopenic purpura, myasthenia gravis, and familial hypercholesterolemia. Use cases include plasma exchange, red cell exchange, immunoadsorption, and leukapheresis performed in hospitals and specialty clinics. Buyers evaluate device reliability, separation efficiency, disposable kit quality, treatment protocols, and integration with existing clinical workflows. Procurement teams prioritize suppliers with regulatory approvals, evidence-based clinical support, robust service networks, and training programs for healthcare providers. Trend in the global market reflects adoption of personalized treatment regimens, expansion of outpatient apheresis centers, and emphasis on reducing procedure times and patient risk.
Terumo BCT holds leading positioning through comprehensive therapeutic apheresis platforms and disposables used across plasma exchange and cell collection applications with wide clinical adoption. Fresenius Kabi supports demand with apheresis equipment and consumable systems integrated into hospital therapy portfolios. Asahi Kasei Medical contributes high-performance apheresis systems and cartridges designed for precision separation and clinical versatility. Haemonetics participates with therapeutic device platforms and support services focused on treatment efficacy and operational reliability. B. Braun Melsungen supplies apheresis solutions and disposables used in diverse therapeutic protocols supported by global service infrastructure. Competitive differentiation depends on device performance, clinical evidence, global training support, and ability to meet diverse treatment needs in specialty care settings.
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Procedure Types | Therapeutic plasma exchange (TPE); Red blood cell exchange (RBCX); Cellular depletions; Other procedures |
| Technology | Centrifugation; Membrane filtration |
| Application | Sickle cell disease; Neurology; Nephrology; Familial hypercholesterolemia; Oncology; Autoimmune disease; Graft-versus-host disease (GVHD); Transplant of solid organs |
| End User | Hospitals; Blood banks; Specialty clinics; Ambulatory surgical centers |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, China, South Korea, USA, Germany, and 40+ countries |
| Key Companies Profiled | Terumo BCT; Fresenius Kabi; Asahi Kasei Medical; Haemonetics; B. Braun Melsungen |
| Additional Attributes | Dollar sales by procedure type and technology; adoption trends for TPE and RBCX across hematology, neurology, and transplant indications; treatment efficiency, plasma removal rates, and patient safety performance metrics; suitability across centrifugation and membrane-based platforms; consumables utilization, procedure duration, and staffing requirements; regulatory compliance with transfusion medicine standards and clinical guidelines influencing therapeutic apheresis utilization. |
How big is the therapeutic apheresis market in 2026?
The global therapeutic apheresis market is estimated to be valued at USD 3.0 billion in 2026.
What will be the size of therapeutic apheresis market in 2036?
The market size for the therapeutic apheresis market is projected to reach USD 4.4 billion by 2036.
How much will be the therapeutic apheresis market growth between 2026 and 2036?
The therapeutic apheresis market is expected to grow at a 3.9% CAGR between 2026 and 2036.
What are the key product types in the therapeutic apheresis market?
The key product types in therapeutic apheresis market are therapeutic plasma exchange (tpe), red blood cell exchange (rbcx), cellular depletions and other procedures.
Which end user segment to contribute significant share in the therapeutic apheresis market in 2026?
In terms of end user, hospitals segment to command 47.8% share in the therapeutic apheresis market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.