Drug Resistant Virus Treatment Market
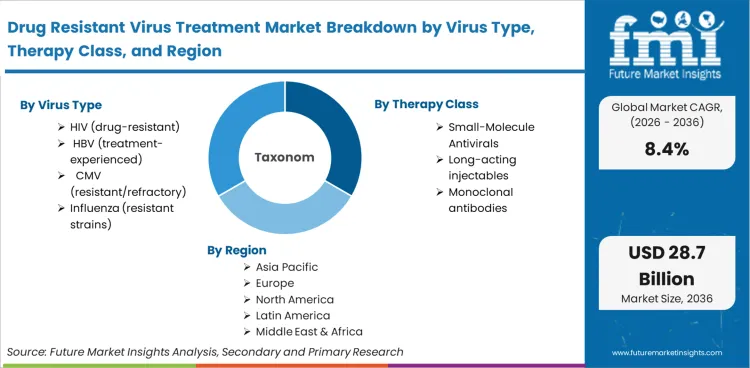
The Drug Resistant Virus Treatment Market is segmented by Virus Type (HIV (drug-resistant), HBV (treatment-experienced), CMV (resistant/refractory), Influenza (resistant strains), HSV/VZV (acyclovir-resistant), Other), Therapy Class (Small-molecule antivirals, Long-acting injectables, Monoclonal antibodies, Host-targeted therapies, Combination regimens), Treatment Setting (Outpatient, Inpatient, Specialty infusion centers), Route of Administration (Oral, Intravenous, Subcutaneous, Intramuscular), Distribution Channel (Hospital pharmacy, Retail pharmacy, Specialty pharmacy, Government / public programs) and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
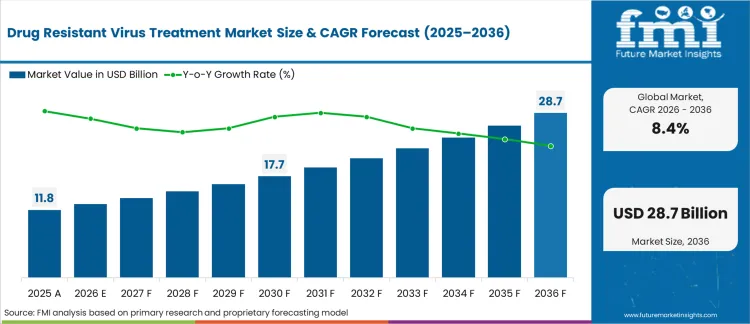
Drug Resistant Virus Treatment Market size, Market Forecast and Outlook By FMI
In 2026, the drug-resistant virus treatment market was valued at USD 12.8 billion. Based on Future Market Insights analysis, demand for drug resistant virus treatment is estimated to grow to USD 28.6 billion by 2036. FMI projects a CAGR of 8.4% during the forecast period.
Absolute dollar growth of USD 1.1 billion over the decade signals a scaling phase rather than a simple volume-led expansion. As per FMI, demand is expected to be supported by major antiviral developers are still investing in new mechanism classes, with capsid targeting presented as a route to expand option sets when resistance limits older regimens. It also reinforces that scale and access execution are being treated as part of development planning, which affects how quickly newer antivirals can be adopted in high-burden countries.
Summary of Drug Resistant Virus Treatment Market
- Drug Resistant Virus Treatment Market Definition
- The industry covers antiviral drugs regimens used to treat resistant or refractory viral infections, supplied through outpatient care, inpatient settings, and specialty infusion centers under protocol-governed prescribing.
- Demand Drivers in the Market
- Rising testing and resistance profiling in HIV and hepatitis care are increasing therapy switching events and repeat treatment cycles.
- Long-term antiviral use is expanding the pool of treatment-experienced patients, increasing demand for salvage and combination regimens.
- Public payer programs and hospital protocols are prioritizing continuity of supply for resistance-linked therapies to avoid rebound and transmission risk.
- Key Segments Analyzed in the FMI Report
- By Virus Type: HIV (drug-resistant) leads at 44.0% share in 2026, followed by HBV (treatment-experienced) at 16.0% share.
- By Therapy Class: Small-molecule antivirals lead at 58.0% share in 2026. followed by Long-acting injectables and periodontitis at 16.0% share.
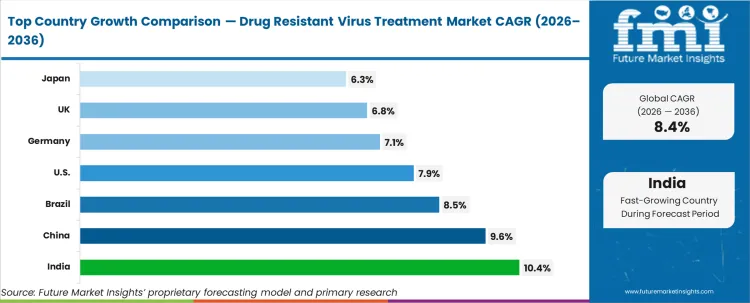
- Geography: India and China show the fastest growth at 10.4% and 9.6% CAGR, supported by expanded diagnosis coverage and program-led treatment access.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Consultant at Future Market Insights, opines, ‘Preference is being decided by resistance barrier, tolerability over long treatment horizons, and assured availability through public channels, since missed doses and forced substitutions can rapidly weaken viral suppression in resistant patients.’
- Strategic Implications and Executive Takeaways
- Protect HIV-resistant positioning through regimen durability evidence and reliable supply performance aligned to program procurement cycles.
- Scale access plans for outpatient settings where repeat dispensing and monitoring are concentrated for chronic antiviral management.
- Build contracting playbooks that match public program tender requirements, including quality documentation and forecast-linked allocation discipline.
- Methodology
- As per FMI, sizing and splits were reconciled using company primary disclosures and therapy-class portfolio mapping across major viruses.
- Shares were validated by linking treatment setting mix to protocol-driven dispensing behavior across outpatient and inpatient pathways.
- Mix sensitivity was tested across therapy classes and routes to confirm revenue movement under realistic regimen substitution patterns.
‘Gilead scientists began to explore whether targeting the capsid of the virus could lead to something new and better for HIV.’ Daniel O’Day, Chairman and CEO of Gilead Sciences, describing the development rationale behind lenacapavir’s capsid-targeting approach. [1]
India (10.4%) CAGR, supported by growing treated populations and expanding specialist access), China (9.6%) CAGR, driven by higher diagnosis coverage and treatment expansion), and Brazil (8.5%) CAGR, linked to public program scale-up) are expected to lead growth. The USA(7.9%) CAGR is expected to expand through regimen optimisation and switch-based demand. Mature markets including Germany (7.1%), the UK (6.8%), and Japan (6.3%) are expected to contribute more through replacement and regimen switching, constrained by strict reimbursement criteria and conservative line-of-therapy progression rules.
Drug Resistant Virus Treatment Market Definition
The market covers pharmacological therapies used to treat viral infections where resistance, treatment experience, or refractory disease limits standard regimens. It includes treatments used for HIV (drug-resistant), HBV (treatment-experienced), CMV (resistant/refractory), influenza resistant strains, and HSV/VZV (acyclovir-resistant), delivered across outpatient, inpatient, and specialty infusion settings. Therapy classes include small-molecule antivirals, long-acting injectables, monoclonal antibodies, host-targeted therapies, and combination regimens. Demand is shaped by resistance testing, prior treatment history, and pathway rules, with revenue generated through product sales via hospital, retail, specialty, and public program channels.
Drug Resistant Virus Treatment Market Inclusions
The report includes global and regional market sizing and a 10-year forecast for 2026 to 2036. It provides segment-level sizing by virus type, therapy class, treatment setting, route of administration, and distribution channel, with country-level CAGR comparisons across major markets. Coverage includes assessment of care pathway controls, switching dynamics, and channel mix that influence treatment uptake and supplier positioning, as per FMI analysts.
Drug Resistant Virus Treatment Market Exclusions
The scope excludes prophylactic vaccines and non-therapeutic prevention programs that do not treat resistant or refractory viral disease. It also omits diagnostic test revenue, including resistance assays and viral load testing, and excludes hospital procedure fees and service revenue. Supportive care products that do not have antiviral treatment action are excluded. The focus remains on treatment products aligned to the listed virus types, therapy classes, settings, and distribution channels.
Drug Resistant Virus Treatment Market Research Methodology
- Primary Research: Interviews were conducted with infectious disease specialists, hospital pharmacists, general physicians, and manufacturer market access teams to confirm usage of pathways, factors driving switching, and purchasing channels.
- Desk Research: Public domain information from regulators, government health authorities, peer-reviewed literature, trade associations, and primary company announcements was analyzed to compare treatment standards and pathways.
- Market-sizing and Forecasting: A hybrid top-down and bottom-up model was used, which includes population at risk, disease epidemiology, country-level treated population with therapy mix by virus type and end use setting, then reconciled through sales of drugs from manufacturer prospectives.
- Data Validation and Update Cycle: Outputs were checked through internal consistency screening, variance review across segment totals, and structured peer checks prior to release.
Segmental Analysis
Drug Resistant Virus Treatment Market Analysis by Virus Type
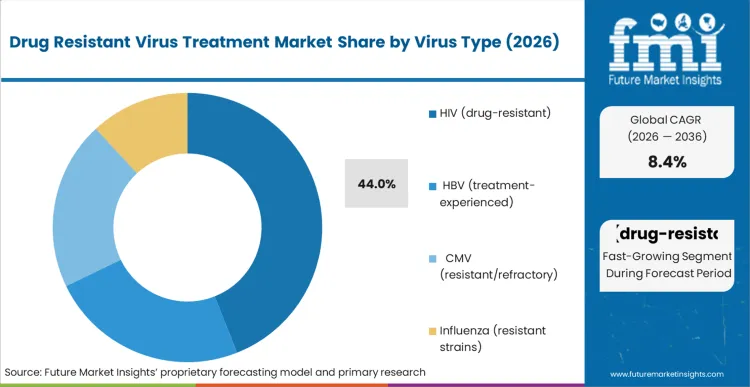
On the basis of FMI’s report, HIV (drug-resistant) is projected to command a share of 44.0% in 2026. This is driven by the chronic nature of HIV treatment and the medical requirement to change treatment regimens when resistance or intolerance occurs. Other forms of resistant viruses also command substantial volume, although these are more episodic in nature, depending on the intensity of outbreaks, referral rates, and availability of specialized testing.
- Routine Resistance Testing Supporting Predictable Switching: The standard practice of HIV care involves organized viral load monitoring and genotypic resistance testing. This allows for appropriate switching, thus establishing a predictable demand pattern. The resistance detection infrastructure supporting the practice of HIV care further ensures a stable market growth in this category.
- High Genetic Barrier & Advanced Mechanism Innovation: The leadership position in the HIV market is maintained through this innovation of treatments that help to keep the effectiveness against the resistant strains of the virus. New molecules that emphasize the differentiated targets in the viral life cycle and possess a higher genetic barrier to resistance are being stressed to improve the treatment durability in the heavily pretreated population. Through this innovation cycle, the premium therapeutic franchise is maintained.
- Closing Prevention and Adherence Gaps to Mitigate Resistance Development: The factors that are associated with the risks of virologic failure and the emergence of drug-resistant strains of HIV include the challenges of long-term treatment adherence, limited access to healthcare, and the burden of the disease among vulnerable populations. This has led to the need for regimen optimization and switching, thus maintaining the need for the development of advanced antiretroviral drugs. The need to improve patient retention in the HIV care continuum and access to resistance testing is being promoted to ensure structured treatment adjustments, repeat prescribing, and extended treatment duration, thus ensuring the continued growth of the drug-resistant HIV treatment market. [2]
Drug Resistant Virus Treatment Market Analysis by Therapy Class
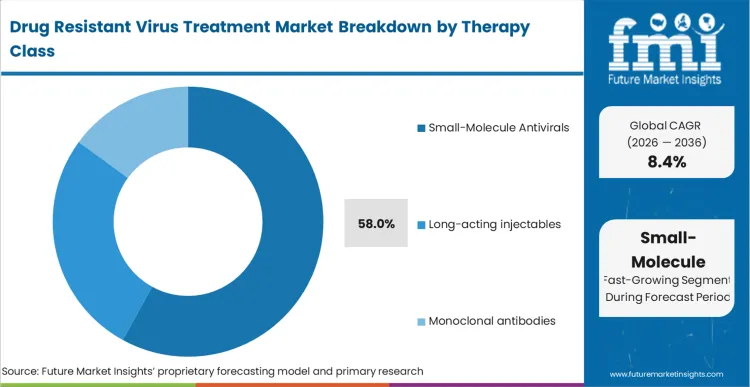
Small molecule antiviral drugs have a 58.0% market share in 2026, according to FMI’s report. This is due to widespread use in multiple resistant virus pathways and the convenience of oral regimens that are aligned with outpatient care. Long-acting injectable and biologic-driven products are used in targeted settings where patient compliance, resistance, and intensity of monitoring support increased complexity, although adoption is gated by facility readiness and reimbursement.
- Broad applicability across Diverse RNA Viruses: Small-molecule antivirals are being actively developed and assessed as critical therapeutic interventions for a range of RNA viruses including HIV, hepatitis viruses, Ebola, Zika, dengue, and SARS-CoV-2 due to their adaptable mechanisms of action, which target and disrupt key stages of viral replication pathways. [3]
- Manufacturing Scalability and Cost Efficiency Advantage: Relative to biologics and long-acting injectable therapies, small molecules have the advantage of simplified manufacturing processes, existing global supply chains, and less reliance on cold chain. This manufacturing advantage will help penetrate geographies, especially in cost-sensitive markets, thus driving overall volume share.
Drug Resistant Virus Treatment Market Drivers, Restraints, and Opportunities
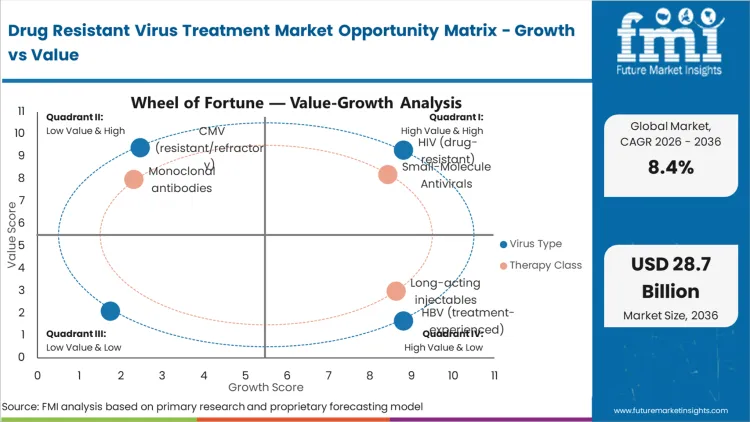
Future Market Insights analysis that historical patterns point at a protocol-governed antiviral category where demand is anchored in chronic suppression goals and regimen switching triggered by resistance, intolerance, or treatment failure. Market structure is being shaped by public program procurement, clinic monitoring cadence, and the need to maintain uninterrupted therapy to prevent rebound and onward transmission, as per FMI.
While scale-up is being restrained by resistance testing access gaps, tender price pressure, and limited site capacity for infusion-based options, growth is being supported by expanding outpatient treatment coverage and wider availability of combination regimens designed for resistant profiles. Based on FMI’s report, supplier selection is increasingly being filtered by quality documentation, forecast responsiveness, and continuity commitments across public and hospital channels.
- Program-led Access: Demand is being expanded through Global Fund Quality Assurance Policy and WHO Prequalification-linked procurement requirements for antiviral supply.
- Tender Compression: Price-led awards and fixed tender cycles restrict margin headroom and slow switching once suppliers are embedded.
- Outpatient Scaling: Growth is being concentrated in outpatient settings where chronic management, refill cadence, and monitoring workflows support repeat utilization.
Regional Analysis
Based on the regional analysis, oral microbiome products market is segmented into North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia and Middle East & Africa across 30+ countries. Regional performance is assessed using country-level demand signals linked to resistance surveillance, treatment guideline adoption, payer coverage, and public programme procurement, as per FMI. The full report also offers market attractiveness analysis based on regional trends.
.webp)
| Country | CAGR |
|---|---|
| USA | 7.9% |
| China | 9.6% |
| India | 10.4% |
| Germany | 7.1% |
| UK | 6.8% |
| Japan | 6.3% |
| Brazil | 8.5% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Drug Resistant Virus Treatment Market Analysis
North America is shaped by protocol-governed care and strong payer influence, where demand is anchored in viral load monitoring, resistance testing access, and guideline-led switching practices. Gilead Sciences holds a leading position in HIV treatment coverage with deep prescriber familiarity and broad access pathways. ViiV Healthcare remains strongly placed in long-acting regimens supported by specialist channel execution. Merck and Pfizer retain relevance through select antivirals and hospital channels. Future Market Insights analysis links regional demand to outpatient dominance and stable reimbursement for chronic viral management.
- United States: Demand for drug resistant virus treatment in the United States is projected to rise at 7.9% CAGR through 2036. Growth is driven by the continued management of HIV cases and increased use of resistance-guided regimen optimization in specialty care, in addition to the sustained demand for treatment-experienced HBV and HSV/VZV patients in targeted cohorts. Demand is sustained by the outpatient delivery model, which includes adherence and specialty pharmacy distribution. Constraint is created by payer prior authorisation and step-therapy rules that can delay switches, which keeps prescribing concentrated in guideline-supported options with strong real-world durability. Based on FMI’s report, hospital use remains important for inpatient antivirals, while long-term volumes remain outpatient-led.
FMI’s report includes a detailed analysis of the growth in the North American region, along with a country-wise assessment that includes the United States. Readers can also find regional trends, regulations, and market growth based on different segments and countries in the North America region.
Asia Pacific Drug Resistant Virus Treatment Market Analysis
The Asia Pacific continues to be the largest growth market, driven by the increasing access to treatment, penetration of diagnostics, and expanding coverage of public programs for chronic viral diseases. Gilead Sciences is represented in the market with HIV and HBV therapies with mixed access. ViiV Healthcare is placed where the adoption of long-acting therapies is underway in the specialist segment. Roche and AbbVie continue to be relevant in the market with more comprehensive antiviral and specialty therapy platforms. According to analysts at FMI, the market is being driven by public buying, domestic guidelines, and the development of specialty infusion capacity.
- China: Demand for drug resistant virus treatment in China is projected to rise at 9.6% CAGR through 2036. Growth is being pulled by expanding access to antiviral therapy through public hospital networks and improving diagnostics coverage in higher-volume urban systems. Demand remains weighted toward outpatient management, while procurement discipline and formulary controls influence regimen availability and switching cadence. Future Market Insights analysis indicates that growth is expected to remain volume-led, with faster uptake where public programme coverage supports earlier initiation and better retention.
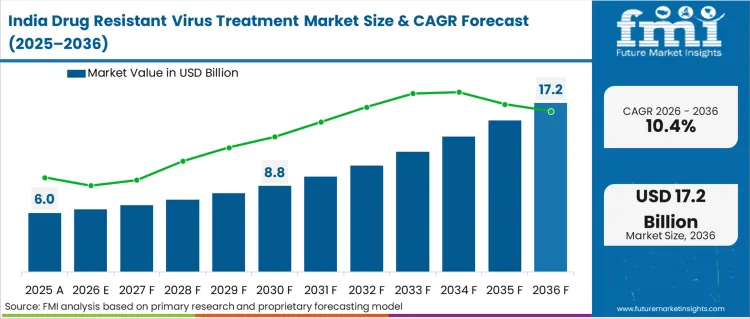
- India: Demand for drug resistant virus treatment in India is projected to rise at 10.4% CAGR through 2036. Growth is supported by expanding access to treatment through public programmes and higher utilisation in private infectious disease and hepatology clinics, with increasing role of specialty pharmacies in metro markets. Demand is also reinforced by wider availability of oral regimens that suit outpatient delivery and reduce facility burden. Constraint is created by uneven access to resistance testing outside major cities, which can delay regimen optimisation for treatment-experienced patients. As per FMI, scale benefits in procurement will continue to shape product mix and pricing.
- Japan: The demand for drug-resistant virus treatment in Japan is expected to grow at 6.3% CAGR till 2036. This is because of the high discipline in protocols and stable coverage structures that support the switching of treatment-experienced patients based on guidelines. The demand for this treatment is mainly driven by specialist hospitals and outpatient facilities. Although the volume of treatment access is mature, the demand is expected to grow moderately. On the other hand, the value growth is driven by higher specification regimens used in complex cases. According to FMI, the demand is expected to be anchored in outpatient settings, while inpatient use is expected to be driven by severe cases.
The full report analyzes the oral microbiome products market across East and South Asia from 2021-2036, covering pricing, trends, and growth drivers in China, Japan, South Korea, India, Australia & New Zealand. The assessment highlights trends that dictate regional demand and procurement behaviour.
Latin America Drug Resistant Virus Treatment Market Analysis
Latin America is fueled by public procurement and mixed access models, where the use of the program is affected by program budgets, tender timing, and the availability of resistance testing in public programs. Gilead Sciences and ViiV Healthcare contribute to the region through HIV programs, and access is affected by country formularies and tender awards. Merck and Pfizer continue to contribute through selected antivirals used in hospital settings. According to the report by FMI, continuity of supply and tender results continue to be important for the stability of regimens in large public programs.
- Brazil: Demand for drug resistant virus treatment in Brazil is projected to rise at 8.5% CAGR through 2036. Growth is supported by public programme coverage for HIV and expanding access to chronic viral management across state-linked health networks. Demand is reinforced by hospital pharmacy and specialty pharmacy channels that support adherence and refills for long-term patients. Constraint is created by tender-driven price pressure and periodic supply variability, which can affect regimen continuity and switching practices in treatment-experienced cohorts. Future Market Insights analysis expects growth to remain anchored in outpatient care, with inpatient utilisation tied to severe infections and comorbidity-driven admissions.
The report consists of a detailed analysis for the market in Brazil, Chile, Argentina, Peru and Rest of Latin America. Readers can find detailed information about several factors, such as the pricing analysis and regional trends, which are impacting growth in the Latin America region.
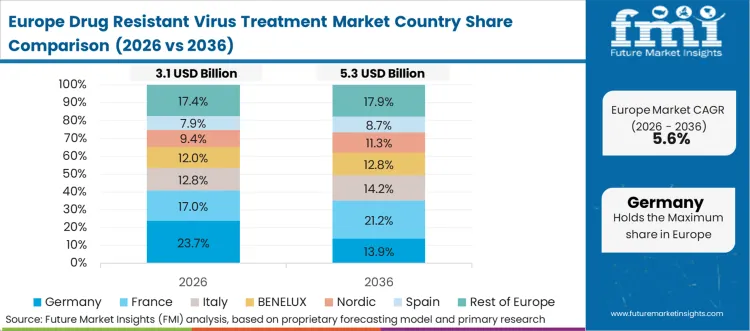
Europe Drug Resistant Virus Treatment Market Analysis
Europe is tender-influenced and guideline-led, where demand is shaped by national reimbursement decisions, specialist prescribing pathways, and structured monitoring that supports resistance-informed switching. Gilead Sciences retains strong access through established HIV and HBV use in multiple countries. ViiV Healthcare is positioned where long-acting options are being adopted within specialist services. Roche and Pfizer remain relevant through broader hospital and infectious disease channels. FMI opines that demand is stabilised by reimbursement governance, with incremental growth tied to treatment-experienced cohorts and higher testing uptake.
- Germany: The demand for drug-resistant virus treatment in Germany is expected to grow at 7.1% CAGR until 2036. This is driven by expert-based HIV treatment and monitoring that allows for early detection of treatment failure and switching. Demand is sustained by robust outpatient specialist access and stable reimbursement for chronic viral treatment. Barriers are introduced by dynamics of price negotiations and prescription management that focus treatment on evidence-based regimens with proven outcomes.
- United Kingdom: Demand for drug resistant virus treatment in the United Kingdom is projected to rise at 6.8% CAGR through 2036. This is due to the presence of specialist commissioning pathways and monitoring that aid in switch therapy for treatment-experienced patients, with outpatient clinics being the primary site of treatment. Demand is shaped by formulary decisions and procurement frameworks that influence product choice and adoption speed. As per FMI, growth remains steady, with value supported by complex-case regimens used in specialist services.
FMI’s analysis of oral microbiome products market in Europe consists of country-wise assessment that includes the Germany, Italy, France, United Kingdom, Spain, Russia, Nordic, Benelux and Rest of Europe. Readers can know various regulations and latest trends in the regional market.
Competitive Aligners for Market Players
Market structure remains fragmented, yet practical competition is concentrated among a limited set of suppliers that can sustain antiviral manufacturing scale, manage resistance-linked lifecycle updates, and maintain compliance-ready supply across outpatient and hospital settings. Practical competition is shaped by formulary access, payer positioning, and continuity under repeat prescribing rather than short-cycle price movement. Product positioning is also shaped by the mix of virus types addressed and the need for predictable response in treatment-experienced patients, where switching decisions are driven by tolerability, interaction profile, and documented resistance considerations, as per FMI.
Manufacturers with structural advantages are generally those with strong antiviral franchises, long-standing relationships with prescribers, and strong expertise in real-world evidence, post-market surveillance, and label maintenance in key geographies. These advantages are only strengthened when a company is able to support combination therapy, mitigate drug-drug interaction constraints, and offer stable access in retail and specialty pharmacy. Firms with narrower portfolios tend to depend on single-franchise performance and local access wins, which increases exposure when guidelines shift, payer rules tighten, or resistance patterns reduce durability for specific regimens, based on FMI’s report.
Customer concentration reinforces buyer leverage. Large payers, national health systems, and hospital networks manage supplier dependency through multi-brand formularies, step edits, prior authorization, and periodic contract resets that keep net pricing disciplined. Specialty pharmacies and public programs also influence allocation through adherence support and channel rules, which can steer volume toward contracted options. Pricing power is therefore constrained where therapeutic alternatives remain acceptable, while stronger pricing positions are retained mainly where resistance constraints, patient complexity, or protocol requirements reduce switching flexibility, Future Market Insights analysis.
Recent Developments
The report includes full coverage of key trends from competitive benchmarking. Some of the recent developments covered in the reports:
- In July 2025, Merck announced the start of Phase 3 trials for MK-8527 a once-monthly HIV PrEP tablet in collaboration with the Gates Foundation, advancing preventive options for HIV at risk of resistant infection. [4]
- In June 2025, Gilead announced that Yeztugo® (lenacapavir) a twice-yearly HIV-1 capsid inhibitor was FDA-approved as the first twice-yearly injectable option for HIV prevention (PrEP), providing expanded long-acting HIV treatment and prevention. [5]
- In December 2024, Pfizer launched a Phase 3 clinical trial for ibuzatrelvir, a next-generation oral antiviral candidate for COVID-19 treatment that could help address variant resistance concerns, reflecting ongoing pipeline innovation.[6]
Key Players in Drug Resistant Virus Treatment Market
- Gilead Sciences
- ViiV Healthcare
- Merck & Co.
- Pfizer
- Roche
- AbbVie
- Astellas Pharma
- Shionogi
- Bristol Myers Squibb
- Johnson & Johnson (Janssen)
- Others
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative units | USD 12.8 billion (2026) to USD 28.6 billion (2036), at a CAGR of 8.4% |
| Market definition | The drug resistant virus treatment market comprises the global production and trade of antiviral therapies used to treat resistant or treatment experienced viral infections, spanning HIV, HBV, CMV, influenza, and HSV or VZV, where demand is shaped by resistance testing access, guideline driven regimen switching, outpatient specialty care scale, and payer and public program coverage for chronic antiviral therapy. |
| Virus Type segmentation | HIV (drug-resistant), HBV (treatment-experienced), CMV (resistant/refractory), Influenza (resistant strains), HSV or VZV (acyclovir-resistant), Other |
| Therapy Class segmentation | Small-molecule antivirals, Long-acting injectables, Monoclonal antibodies, Host-targeted therapies, Combination regimens |
| Treatment Setting coverage | Outpatient, Inpatient, Specialty infusion centers |
| Route of Administration coverage | Oral, Intravenous, Subcutaneous, Intramuscular |
| Distribution Channel Coverage | Hospital pharmacy, Retail pharmacy, Specialty pharmacy, Government or public programs |
| Regions covered | North America, Latin America, East Asia, South Asia, Western Europe, Eastern Europe, Middle East and Africa |
| Countries covered | United States, Canada, Mexico, Brazil, Argentina, Germany, France, United Kingdom, Italy, Spain, China, India, Japan, South Korea, Indonesia, Australia and 40 plus countries |
| Key companies profiled | Gilead Sciences, Inc., ViiV Healthcare Limited, Merck & Co., Inc., Pfizer Inc., F. Hoffmann-La Roche Ltd, AbbVie Inc., Astellas Pharma Inc., Shionogi & Co., Ltd., Bristol-Myers Squibb Company, Johnson & Johnson |
| Forecast period | 2026 to 2036 |
| Approach | Hybrid top down and bottom up market modeling validated through primary interviews with infectious disease clinicians, specialty pharmacies, and manufacturers, supported by treatment population triangulation and guideline mapping, as per FMI. |
Drug Resistant Virus Treatment Market Analysis by Segments
Virus Type:
- HIV (drug-resistant)
- HBV (treatment-experienced)
- CMV (resistant/refractory)
- Influenza (resistant strains)
- HSV/VZV (acyclovir-resistant)
- Other
Therapy Class:
- Small-molecule antivirals
- Long-acting injectables
- Monoclonal antibodies
- Host-targeted therapies
- Combination regimens
Treatment Setting:
- Outpatient
- Inpatient
- Specialty infusion centers
Route of Administration:
- Oral
- Intravenous
- Subcutaneous
- Intramuscular
Distribution Channel:
- Hospital pharmacy
- Retail pharmacy
- Specialty pharmacy
- Government / public programs
Region:
- Asia Pacific
- India
- China
- Japan
- South Korea
- Indonesia
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- Italy
- France
- United Kingdom
- Spain
- Benelux
- Nordics
- Central & Eastern Europe
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Turkey
- Rest of Middle East & Africa
Bibliography
- [1] Gilead Sciences. (2025). An Open Letter From Daniel O’Day, Chairman and CEO. Corporate Communications / Executive Letter.
- [2] Gilead Sciences. (2025). Yeztugo (Lenacapavir) Is Now the First and Only FDA-Approved HIV Prevention Option Offering 6 Months of Protection. Press Release.
- [3] Li, S., Li, H., Lian, R., Xie, J., & Feng, R. (2024). New perspective of small-molecule antiviral drugs development for RNA viruses. Virology. ScienceDirect / Peer-Reviewed Article.
- [4] Merck & Co.. (2025). Merck to Initiate Phase 3 Trials for Investigational Once-Monthly HIV Prevention Pill. Press Release.
- [5] Gilead Sciences. (2025). Gilead Presents New HIV Treatment and Cure Research Data at CROI 2025 Including an Investigational Long-Acting Twice-Yearly Therapy Option. Press Release / Conference Update.
- [6] Pfizer. (2025). Pfizer Launched Phase 3 Trial for Ibuzatrelvir, a New Oral Antiviral for COVID-19 Treatment. Industry News / Clinical Trial Announcement.
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
This report addresses
- Market intelligence to enable structured strategic decision making across oral care innovation, portfolio planning, and channel prioritization
- Market size estimation and 10 year revenue forecasts from 2026 to 2036 for oral microbiome product demand
- Growth opportunity mapping across virus type, therapy class, treatment setting, route of administration, and distribution channel demand pools
- Segment and regional revenue forecasts aligned to the stated taxonomy and covered geographies
- Competition strategy assessment including positioning by product type, formulation, application, and sales channel coverage benchmarking
- Product and compliance tracking focused on quality expectations, labeling discipline, and formulation governance for microbiome positioned products
- Regulatory impact analysis covering how probiotics and biotherapeutic guidance can shape clinical positioning and market access planning
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats for executive and operational use
Frequently Asked Questions
How large is the demand for oral microbiome products in the global market in 2026?
Demand is estimated to be valued at USD 12.8 billion in 2026, as per FMI.
What will be the market size of oral microbiome products in the global market by 2036?
Market size is projected to reach USD 28.6 billion by 2036, based on FMI’s report.
What is the expected demand growth for oral microbiome products in the global market between 2026 and 2036?
Demand is expected to grow at a CAGR of 8.4% between 2026 and 2036.
Which virus type is poised to lead global sales by 2026?
HIV (drug-resistant) is expected to be the dominant product, capturing 44.0% share in 2026.
Which therapy class is expected to account for the largest share in 2026?
Small-molecule antivirals are expected to hold the highest share at 58.0% in 2026.
How significant is Hospital Pharmacies in the 2026 sales channel mix?
Hospital Pharmacies are projected to account for 38.0% share in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Virus Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Virus Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Virus Type , 2026 to 2036
- HIV (drug-resistant)
- HBV (treatment-experienced)
- CMV (resistant/refractory)
- Influenza (resistant strains)
- HIV (drug-resistant)
- Y to o to Y Growth Trend Analysis By Virus Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Virus Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Therapy Class
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Therapy Class, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Therapy Class, 2026 to 2036
- Small-Molecule Antivirals
- Long-acting injectables
- Monoclonal antibodies
- Small-Molecule Antivirals
- Y to o to Y Growth Trend Analysis By Therapy Class, 2021 to 2025
- Absolute $ Opportunity Analysis By Therapy Class, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Virus Type
- By Therapy Class
- By Country
- Market Attractiveness Analysis
- By Country
- By Virus Type
- By Therapy Class
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Virus Type
- By Therapy Class
- By Country
- Market Attractiveness Analysis
- By Country
- By Virus Type
- By Therapy Class
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Virus Type
- By Therapy Class
- By Country
- Market Attractiveness Analysis
- By Country
- By Virus Type
- By Therapy Class
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Virus Type
- By Therapy Class
- By Country
- Market Attractiveness Analysis
- By Country
- By Virus Type
- By Therapy Class
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Virus Type
- By Therapy Class
- By Country
- Market Attractiveness Analysis
- By Country
- By Virus Type
- By Therapy Class
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Virus Type
- By Therapy Class
- By Country
- Market Attractiveness Analysis
- By Country
- By Virus Type
- By Therapy Class
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Virus Type
- By Therapy Class
- By Country
- Market Attractiveness Analysis
- By Country
- By Virus Type
- By Therapy Class
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Virus Type
- By Therapy Class
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Virus Type
- By Therapy Class
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Virus Type
- By Therapy Class
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Virus Type
- By Therapy Class
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Virus Type
- By Therapy Class
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Virus Type
- By Therapy Class
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Virus Type
- By Therapy Class
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Virus Type
- By Therapy Class
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Virus Type
- By Therapy Class
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Virus Type
- By Therapy Class
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Virus Type
- By Therapy Class
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Virus Type
- By Therapy Class
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Virus Type
- By Therapy Class
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Virus Type
- By Therapy Class
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Virus Type
- By Therapy Class
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Virus Type
- By Therapy Class
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Virus Type
- By Therapy Class
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Virus Type
- By Therapy Class
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Virus Type
- By Therapy Class
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Virus Type
- By Therapy Class
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Virus Type
- By Therapy Class
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Virus Type
- By Therapy Class
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Virus Type
- By Therapy Class
- Competition Analysis
- Competition Deep Dive
- Gilead Sciences
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- ViiV Healthcare
- Merck & Co.
- Pfizer
- Roche
- AbbVie
- Astellas Pharma
- Shionogi
- Gilead Sciences
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Virus Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Therapy Class, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Virus Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Therapy Class, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Virus Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Therapy Class, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Virus Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Therapy Class, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Virus Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Therapy Class, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Virus Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Therapy Class, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Virus Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Therapy Class, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Virus Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Therapy Class, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Virus Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Virus Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Virus Type
- Figure 6: Global Market Value Share and BPS Analysis by Therapy Class, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Therapy Class, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Therapy Class
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Virus Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Virus Type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Virus Type
- Figure 23: North America Market Value Share and BPS Analysis by Therapy Class, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Therapy Class, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Therapy Class
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Virus Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Virus Type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Virus Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Therapy Class, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Therapy Class, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Therapy Class
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Virus Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Virus Type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Virus Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Therapy Class, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Therapy Class, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Therapy Class
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Virus Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Virus Type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Virus Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Therapy Class, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Therapy Class, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Therapy Class
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Virus Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Virus Type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Virus Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Therapy Class, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Therapy Class, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Therapy Class
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Virus Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Virus Type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Virus Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Therapy Class, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Therapy Class, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Therapy Class
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Virus Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Virus Type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Virus Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Therapy Class, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Therapy Class, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Therapy Class
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

