Demand for drug screening in Japan is valued at USD 494.1 million in 2026 and is projected to reach USD 1,586.8 million by 2036, reflecting a CAGR of 12.4%. The rise from USD 275.7 million in 2020 to USD 494.1 million in 2026 is supported by expanding use of urine, breath, oral fluid, and hair testing across laboratories, hospitals, and criminal justice agencies. Analytical instruments, rapid testing devices, and consumables anchor early procurement as testing volumes widen across regulated settings in Japan.
From 2027, values increase from USD 555.2 million to USD 885.4 million by 2031, then reach USD 1,586.8 million by 2036 through widening annual increments. Drug testing laboratories remain the primary end users, while law enforcement and treatment centers sustain predictable growth in screening activity. Hospitals contribute steady demand linked to diagnostic workflows. Service based offerings expand as organizations outsource testing functions. Long term growth reflects heightened monitoring needs, broader sample type adoption, and consistent policy driven testing across Japan.
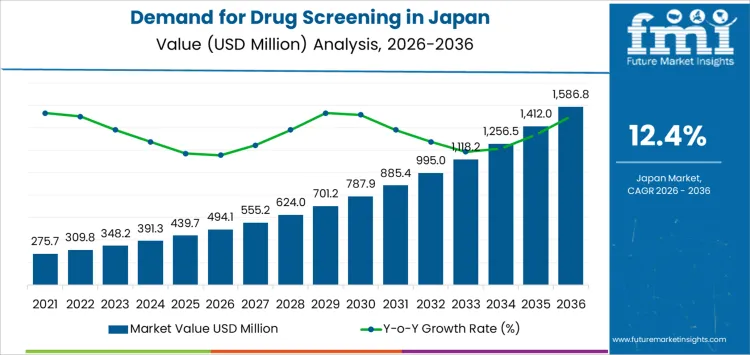
Between 2026 and 2031, demand for drug screening in Japan rises from USD 494.1 million to USD 885.4 million, marking a significant increase driven by expanding diagnostic workloads, broader toxicology testing requirements, and greater institutional reliance on structured screening protocols. Growth in this phase reflects heightened pharmaceutical development activity, stronger occupational health monitoring, and increased use of screening tools in clinical decision pathways. Earlier demand was shaped by compliance-driven testing, while the near-term pattern shows a pivot toward more comprehensive screening panels, higher test frequencies, and wider deployment of automated analytical platforms across hospital and laboratory networks.
From 2031 to 2036, demand advances from USD 885.4 million to USD 1,586.8 million, showing an even sharper rise as screening becomes embedded in preventive medicine workflows, clinical trial enrollment procedures, and advanced therapeutic safety assessments. Expansion in this period is associated with higher compound diversity in drug pipelines, integration of high-throughput toxicology technologies, and growing dependence on external screening partners for rapid turnaround testing. Earlier progress reflected operational scaling, but later growth is shaped by structurally higher testing volumes, precision-oriented analytical requirements, and broader application of screening in personalized care frameworks across Japan’s evolving healthcare and research environments.
| Metric | Value |
|---|---|
| Industry Value (2026) | USD 494.1 million |
| Forecast Value (2036) | USD 1,586.8 million |
| Forecast CAGR (2026-2036) | 12.4% |
Demand for drug screening in Japan has grown with expanding clinical trial activity, regulatory emphasis on safety, and broader use of therapeutic monitoring in healthcare settings. Pharmaceutical sponsors and contract research organisations increased screening for candidate compounds to assess toxicity, efficacy markers, and pharmacokinetic profiles. Hospitals and diagnostic labs adopted screening tools to monitor patient responses and detect adverse effects during treatment. Historical demand aligned with growth in domestic R&D activity, clinical trial volume, and stricter preclinical safety standards that required comprehensive compound evaluation before human studies.
From 2026 to 2036 demand is expected to strengthen as precision medicine, biologics, and complex therapeutic classes such as cell and gene therapies gain prominence. Sponsors seek advanced screening platforms that integrate high-throughput assays, multi-omics data, and predictive models to reduce late-stage failures. Healthcare providers increase use of therapeutic drug monitoring to personalise regimens for chronic conditions and oncology treatments. Buyers prioritise analytical accuracy, turnaround times, and integration with electronic health records during procurement. Demand also reflects expansion of centralised laboratory services and partnerships with specialised drug screening facilities to handle larger sample volumes and regulatory documentation requirements nationwide.
The demand for drug screening in Japan from 2026 to 2036 reflects expanded occupational testing, stricter compliance programs, and broader adoption of multi-sample screening frameworks. Urine samples lead due to established collection procedures and wide analytical compatibility. Drug screening products hold the highest share among product-service categories as institutions rely on validated kits and platforms that support routine workplace testing, clinical assessments, and structured monitoring programs across regulated environments.
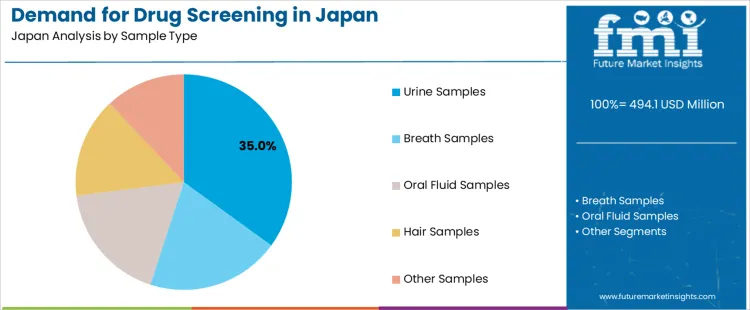
Urine samples account for 35% of Japan’s demand because this sample type remains widely accepted, easy to collect, and compatible with high-throughput laboratory instruments. Consumption rises across employment testing, rehabilitation monitoring, and clinical evaluations requiring established cut-off thresholds and consistent detection windows. Usage stays steady since most laboratories maintain standardized workflows optimized for urine-based assays. Procurement is guided by facilities selecting collection kits, reagents, and transport containers that support secure chain-of-custody handling. Price sensitivity remains moderate because urine testing offers balanced costs while supporting broad drug panels. Specification control focuses on sample stability, contamination prevention, and compatibility with automated screening platforms. Demand continues through 2026 to 2036 as institutions maintain structured testing policies grounded in predictable sample processing and reliable detection performance.
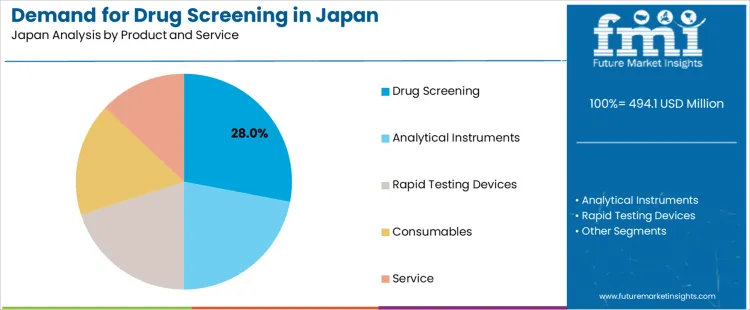
Drug screening products represent 28.0% of the demand because workplaces, clinics, and compliance programs rely on ready-to-use kits that support routine testing with minimal setup. Consumption remains strong in organizations conducting scheduled monitoring where standardized test formats improve consistency across batches. Usage stays steady since these products integrate fixed reagent volumes, preset detection thresholds, and clear visual or digital outputs suited to varied testing environments. Procurement decisions center on lot reproducibility, documentation support, and compatibility with existing screening policies. Price sensitivity remains balanced as organizations evaluate product durability and testing frequency. Specification control highlights accuracy across different drug classes, ease of interpretation, and shelf-life stability. Demand holds from 2026 to 2036 as institutions prioritize predictable tools that streamline testing without requiring extensive analytical infrastructure.
Demand rises through routine programs in Japanese hospitals, outpatient clinics, and public laboratories that monitor medication safety and treatment adherence. Hospitals in Osaka request targeted tests during reviews of complex regimens. Community clinics in Chiba use screening when patients report symptoms linked to recent prescriptions. Prefectural labs in Miyagi process samples tied to monitoring programs for residents on long term therapies. University centers in Nagoya incorporate screening into projects funded through local research schemes. These patterns reflect practical needs across Japan’s care network, where screening supports daily decision making in clinical and research settings.
Drivers emerge from medication oversight practices shaped by Japan’s aging population and structured care pathways. Hospitals in Sapporo request screening during evaluations of older patients with overlapping prescriptions. Clinics in Fukuoka use rapid tests when side effects appear shortly after routine treatments. Research groups in Kyoto conduct screening during candidate refinement supported by regional grants. Community health centers in Toyama use defined panels during seasonal monitoring programs. These activities demonstrate how local routines, patient profiles, and research timetables shape steady demand within diagnostic and scientific environments across Japan.
Barriers relate to capacity, budgets, and existing workflows. Clinics in rural prefectures avoid comprehensive panels because testing budgets remain limited. Hospitals in older facilities face equipment space limits that slow installation of advanced analyzers. University labs in Ehime continue to rely on established internal methods that restrict outsourcing. Public health centers working with fixed annual allocations focus on high volume tests, which delays adoption of specialized panels. Transport times from isolated areas to metropolitan laboratories can be long, reducing timely access. These factors create uneven uptake across regions with varied infrastructure and resources.
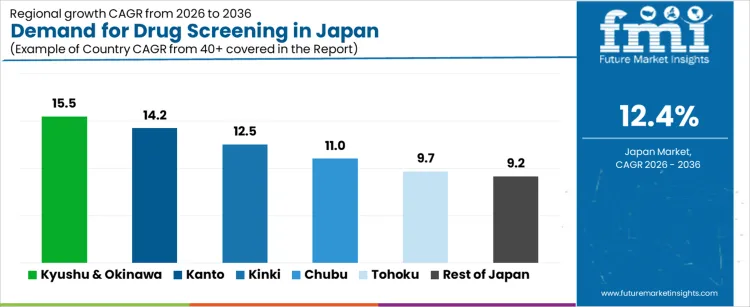
| Region | CAGR (%) |
|---|---|
| Kyushu & Okinawa | 15.5% |
| Kanto | 14.2% |
| Kinki | 12.5% |
| Chubu | 11.0% |
| Tohoku | 9.7% |
| Rest of Japan | 9.2% |
The demand for drug screening in Japan is rising rapidly across clinical diagnostics, workplace testing, forensic laboratories, and research programs, with Kyushu and Okinawa leading at a 15.5% CAGR. Growth in this region is driven by expanded healthcare networks, higher testing volumes across hospitals, and adoption of advanced analytical platforms. Kanto follows at 14.2%, supported by major diagnostic laboratories, dense urban populations, and strong integration of high throughput screening systems. Kinki shows 12.5% growth, reflecting increased utilization in clinical centers and university hospitals. Chubu at 11.0% demonstrates steady adoption aligned with regional healthcare expansions and industrial workplace testing requirements. Tohoku and the Rest of Japan, at 9.7% and 9.2%, record slower yet stable growth influenced by smaller testing infrastructures and more conservative deployment of advanced screening technologies.
Demand for drug screening in Kyushu and Okinawa is increasing at a CAGR of 15.5% from 2026 to 2036 as universities, biotech firms, and medical centers expand early stage discovery programs. Research groups rely on outsourced screening to evaluate compound behavior. Biotech firms use external platforms to manage capacity limits. Hospitals with research units integrate screening support into translational studies. Growth reflects concentrated academic activity supported by external analytical capabilities used across varied scientific tasks.
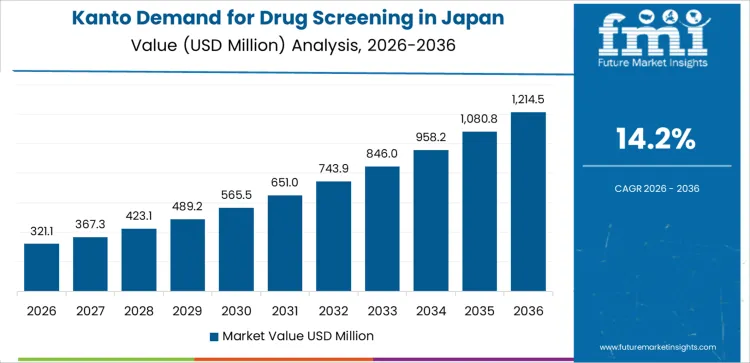
Demand for drug screening in Kanto is advancing at a CAGR of 14.2% from 2026 to 2036 as large pharmaceutical companies, hospital laboratories, and research institutes coordinate screening activities through specialized providers. Pharmaceutical firms conduct structured screening to progress pipeline programs. Hospital labs engage external partners for targeted molecular assessments. University groups manage exploratory research with support from screening providers. Growth aligns with dense scientific activity and active participation across public and private institutions.
Demand for drug screening in Kinki is rising at a CAGR of 12.5% from 2026 to 2036 as universities, clinical research groups, and biotech firms incorporate external capabilities into research cycles. Universities rely on screening partners to evaluate compound behavior across early studies. Clinical research units coordinate focused assessments tied to therapeutic programs. Biotech firms use external workflows to manage laboratory limits. Growth reflects balanced engagement across institutions that use screening support for planned research tasks.
Demand for drug screening in Chubu is progressing at a CAGR of 11.0% from 2026 to 2036 as hospitals, research centers, and contract labs integrate screening into investigative activity. Hospitals engage providers for molecular evaluations linked to internal projects. Research centers rely on external screening for exploratory studies. Contract labs assist institutions that require scalable analytical support. Growth reflects methodical research structures across a region that relies on coordinated screening tasks to advance scientific work.
Demand for drug screening in Tohoku is growing at a CAGR of 9.7% from 2026 to 2036 as universities, medical laboratories, and public research groups use screening providers to support scientific programs. Universities require screening assistance for complex evaluations. Medical laboratories employ external partners for selected research roles. Regional research groups rely on structured screening to support limited internal capacity. Growth reflects steady participation across dispersed institutions with focused scientific activity.
Demand for drug screening in Rest of Japan is expanding at a CAGR of 9.2% from 2026 to 2036 as hospitals, local research units, and smaller universities outsource screening to manage resource restrictions. Hospitals incorporate screening support for molecular assessments. Local research units outsource early evaluations to maintain workflow continuity. Smaller universities depend on providers to carry out structured screening tasks. Growth reflects practical research activity across diverse institutions that use external capabilities to sustain scientific development.
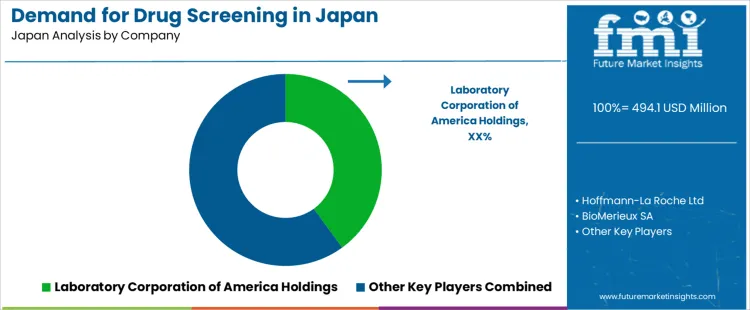
The demand for drug screening in Japan is shaped by workplace compliance programs, hospital based toxicology needs, and increased monitoring of prescription and over the counter medication use in aging populations. Domestic diagnostic firms supply much of the routine testing infrastructure through immunoassay platforms used in hospital laboratories and occupational health centers. Laboratory Corporation of America Holdings influences testing standards through widely referenced toxicology methods used by Japanese clinicians for complex confirmations. Hoffmann La Roche Ltd supports demand through automated analyzers and reagents suited to high throughput hospital environments. Bio Rad Laboratories Inc participates through quality controls and reagents adopted for consistent assay performance across large diagnostic labs. BioMerieux SA contributes through analyzers and immunoassay systems used in multi panel toxicology screening. Express Diagnostics Int l Inc reaches Japan through rapid test kits used in workplace and community screening programs.
Test selection in Japan is governed by analytical sensitivity, cross reactivity control, clear interpretation ranges, and alignment with national occupational health guidance. Hospitals emphasize rapid turnaround for emergency cases and reliable confirmation pathways for complex drug exposures. Employers rely on simple point of care tests for pre-employment and periodic screening in safety sensitive sectors. Buyers prefer systems with robust documentation, local service networks, and stable reagent supply. Demand visibility tracks growth in hospital emergency visits linked to polypharmacy, expansion of workplace testing in logistics and transport companies, and ongoing refinement of toxicology protocols across clinical laboratories and health management organizations.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD million |
| Sample Type | Urine Samples; Breath Samples; Oral Fluid Samples; Hair Samples; Other Samples |
| Product and Service | Drug Screening; Analytical Instruments; Rapid Testing Devices; Consumables; Service |
| End User | Drug Testing Laboratories; Criminal Justice and Law Enforcement Agencies; Hospitals; Drug Treatment Centers; Individual Users |
| Regions Covered | Kyushu & Okinawa; Kanto; Kansai; Chubu; Tohoku; Rest of Japan |
| Countries Covered | Japan |
| Key Companies Profiled | Laboratory Corporation of America Holdings; Hoffmann-La Roche Ltd; BioMerieux SA; Express Diagnostics Int’l, Inc.; Bio-Rad Laboratories Inc. |
| Additional Attributes | Dollar by sales across sample types and product-service categories; test throughput and turnaround time metrics; panel breadth and sensitivity thresholds; reagent and consumable replacement rates; confirmatory testing share; point-of-care versus central lab split; chain-of-custody and regulatory compliance requirements; outsourcing ratio for screening services; geographic lab capacity and distribution. |
How big is the demand for drug screening in Japan in 2026?
The demand for drug screening in Japan is estimated to be valued at USD 494.1 million in 2026.
What will be the size of drug screening in Japan in 2036?
The market size for the drug screening in Japan is projected to reach USD 1,586.8 million by 2036.
How much will be the demand for drug screening in Japan growth between 2026 and 2036?
The demand for drug screening in Japan is expected to grow at a 12.4% CAGR between 2026 and 2036.
What are the key product types in the drug screening in Japan?
The key product types in drug screening in Japan are urine samples, breath samples, oral fluid samples, hair samples and other samples.
Which product and service segment is expected to contribute significant share in the drug screening in Japan in 2026?
In terms of product and service, drug screening segment is expected to command 28.0% share in the drug screening in Japan in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.