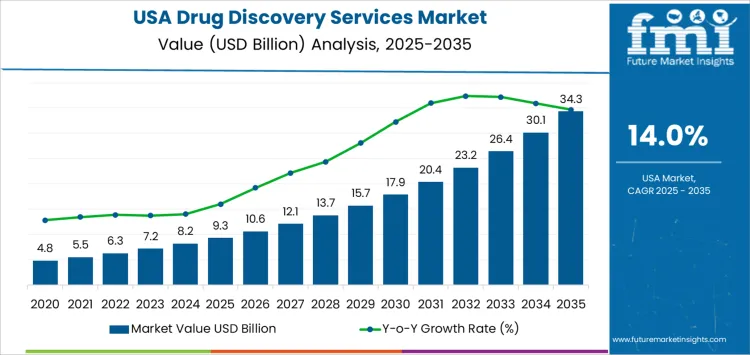
The USA drug discovery services demand is valued at USD 9.3 billion in 2025 and is forecasted to reach USD 34.3 billion by 2035, reflecting a CAGR of 14.0%. Growth is driven by increased outsourcing from pharmaceutical and biotechnology companies seeking cost efficiency, accelerated timelines, and access to specialized research capabilities. Expansion of therapeutic pipelines in oncology, immunology, and rare diseases further strengthens reliance on external discovery partners.
Target selection leads the process segment because early-stage identification of disease-relevant biological pathways underpins successful downstream development. Service providers integrate genomics, proteomics, computational modeling, and AI-enabled analytics to improve precision in selecting viable therapeutic targets. Enhanced biomarker discovery and validation support more efficient candidate progression.
The West, South, and Northeast regions show strong adoption due to concentrated biotech clusters, academic research partnerships, and substantial venture funding activity. Key suppliers include Charles River Laboratories International Inc., Thermo Fisher Scientific Inc., Evotec SE, and Pharmaron Beijing Co., Ltd. Their strategies focus on platform integration, expanded high-throughput screening capacity, and end-to-end discovery solutions that accelerate novel drug development in competitive therapeutic areas.
Saturation Point Analysis for drug discovery services in the United States focuses on the relationship between demand expansion and structural ceilings within research capacity, outsourcing behavior, and therapeutic development priorities. Current demand remains below the saturation threshold because biopharmaceutical pipelines continue to diversify across biologics, gene therapy, and small-molecule programs. This diversification sustains broad reliance on external discovery platforms. Large firms maintain hybrid models that combine in-house capabilities with selective outsourcing, which prevents rapid convergence toward a fixed upper limit.
Smaller biotechnology companies expand service utilization because early research stages require specialized assays, computational modeling, and structural biology resources that are costly to build internally. This dependency extends the distance from saturation because company formation rates remain strong across early-stage innovation clusters. Academic and translational research centers generate additional flow toward service providers through partnership activity that supports preclinical validation.
Operational ceilings appear in areas where highly specialized talent, advanced instrumentation, and regulatory support functions limit rapid expansion of provider capacity. These constraints shape the eventual saturation point because service throughput cannot scale indefinitely without long planning cycles. Price sensitivity across smaller firms also introduces an economic boundary that slows movement toward saturation. The sector remains positioned in a mid-growth zone with considerable headroom due to ongoing scientific and therapeutic diversification.
| Metric | Value |
|---|---|
| USA Drug Discovery Services Sales Value (2025) | USD 9.3 billion |
| USA Drug Discovery Services Forecast Value (2035) | USD 34.3 billion |
| USA Drug Discovery Services Forecast CAGR (2025-2035) | 14.0% |
Demand for drug discovery services in the United States grows as research organizations, biotechnology firms, and pharmaceutical companies expand pipelines targeting complex diseases. Advancements in genomics, cell biology, and computational modeling increase the volume of early stage projects that require specialized screening, assay development, and lead optimization. Small and mid-sized biotechs depend on external discovery partners due to limited in house infrastructure for high throughput studies and preclinical evaluation. Funding for oncology, rare disorders, infectious diseases, and immunology strengthens activity across discovery programs supported by outsourced service models.
Contract research organizations provide integrated platforms that combine chemistry, biology, and data analytics, which supports continuous project progression from hit identification to candidate selection. Mergers and licensing agreements generate additional compound evaluation needs as companies assess potential assets before later development stages. Digital laboratories and AI driven discovery methods create demand for partners with computational capacity and validated experimental workflows. Federal and private investment in translational research expands collaboration between academic centers and service providers. Growth in biologics, peptides, and nucleic acid based therapeutics increases reliance on discovery tools designed for complex molecular classes.
Demand for drug discovery services in the USA is shaped by expanding therapeutic pipelines, outsourcing strategies, and requirements for specialized scientific capabilities across early-stage research. Organizations rely on external expertise to advance molecular targets, validate biological mechanisms, and optimize chemical structures. Distribution reflects workflow complexity, project sequencing, and the need for integrated chemistry and biology support across discovery programs.
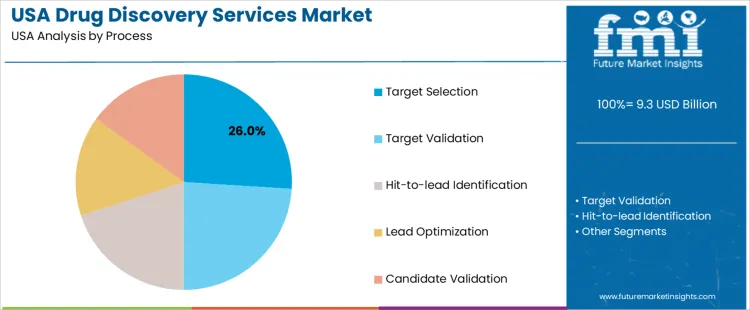
Target selection holds 26.0%, making it the leading process segment in USA demand for drug discovery services. This stage requires extensive bioinformatic analysis, disease-pathway mapping, and feasibility assessment to identify actionable molecular targets. Target validation holds 24.0%, supported by functional studies that determine whether identified targets demonstrate therapeutic relevance. Hit-to-lead identification holds 20.0%, involving screening, compound prioritization, and early profiling. Lead optimization holds 15.0%, focused on improving potency, selectivity, and pharmacokinetic properties. Candidate validation holds 15.0%, confirming preclinical suitability before development. Process distribution reflects the resource intensity and decision-critical nature of early discovery stages.
Key Points:
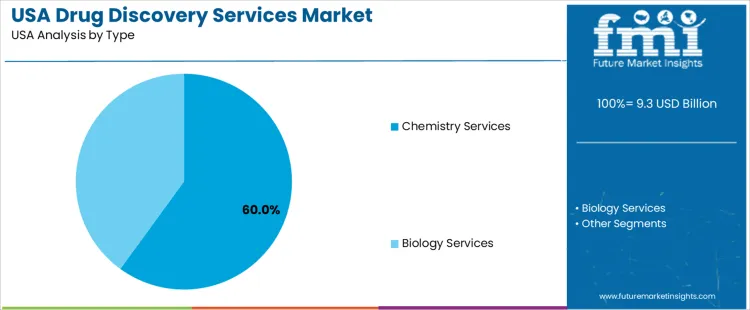
Chemistry services hold 60.0%, making them the leading type segment in USA drug discovery services. These activities support compound synthesis, structure-activity optimization, analytical characterization, and medicinal chemistry workflows. Organizations depend on chemistry partners to accelerate compound design cycles and supply libraries for screening and optimization. Biology services hold 40.0%, supporting mechanism studies, cell-based assays, in-vitro models, and biomarker evaluation. These services validate biological actions and guide chemical refinement decisions. Type distribution reflects the foundational role of chemistry in compound evolution and the parallel need for biological confirmation across discovery programs.
Key Points:
Biopharmaceutical R&D activity, therapeutic pipeline expansion, and specialized expertise are driving demand.
Demand increases as US biopharmaceutical companies outsource early-stage research to accelerate target identification, hit validation, and lead optimization. Startups rely on external discovery partners due to limited internal laboratory capacity. Large drug developers use contract research organizations to manage parallel programs in oncology, immunology, and rare diseases. Venture-backed firms expand preclinical exploration that requires screening libraries, computational modelling, and in vitro assays. Federal funding supports academic-industry collaboration that generates new discovery pathways. Regional clusters in Massachusetts, California, and North Carolina maintain strong utilization of specialized discovery platforms. High development costs encourage outsourcing strategies that enhance pipeline efficiency.
R&D expenditure constraints, regulatory complexity, and talent shortages restrain operational scalability.
Biopharmaceutical firms face budget pressure when prioritizing therapeutic areas with uncertain commercial outcomes, which limits outsourced discovery volume. Regulatory expectations require extensive documentation of assay validation, data quality, and reproducibility, which raises service costs. Several organizations encounter difficulty accessing specialized scientists in structural biology, medicinal chemistry, and computational modelling. Smaller companies report reliance on limited capital pools that influence contract duration and project scope. Data-security requirements increase operational spending for service providers managing sensitive research information. Geographic disparities influence laboratory availability, which restricts scaling of discovery partnerships across regions with fewer research facilities.
AI-driven discovery, integrated platforms, and modality diversification shape current trends.
US drug developers expand use of AI-supported target prediction, virtual screening, and structure-based design to shorten early discovery timelines. Service providers introduce integrated platforms that combine chemistry, biology, and preclinical evaluation into unified workflows. Demand increases for discovery programs focused on cell and gene therapies, RNA-based treatments, and novel biologics. Cloud-based data environments support cross-team collaboration and audit-ready documentation. High-throughput screening advances improve sensitivity for complex target classes. Academic research spin-outs partner with contract organizations to validate novel mechanisms. Pharmaceutical firms evaluate long-term frameworks that standardize external discovery processes across therapeutic portfolios.
Demand for drug discovery services in the USA is increasing due to expanding biopharmaceutical pipelines, higher R&D intensity, and growing reliance on outsourced discovery capabilities. West USA records a CAGR of 16.1% supported by strong biotechnology activity and advanced research ecosystems. South USA shows a 14.4% CAGR driven by expanding pharmaceutical clusters and rising contract research operations. Northeast USA posts a 12.8% CAGR due to dense academic, clinical, and pharmaceutical networks. Midwest USA holds an 11.2% CAGR supported by stable biomedical research output and steady demand for discovery platforms across regional health-science institutions.
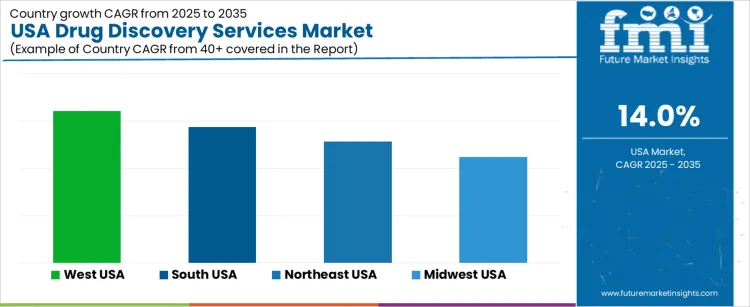
| Region | CAGR (2025-2035) |
|---|---|
| West USA | 16.1% |
| South USA | 14.4% |
| Northeast USA | 12.8% |
| Midwest USA | 11.2% |
West USA drives demand through its strong biotechnology concentration and extensive network of research-driven enterprises requiring specialized discovery capabilities. The region’s CAGR of 16.1% reflects broad engagement in early-stage screening, target identification, and lead optimization supported by advanced laboratory infrastructure. Startups and established biotech firms rely on outsourced platforms to accelerate discovery timelines and manage complex experimental workflows. Academic research centers maintain large volumes of translational studies that require structured support from discovery service providers. High R&D investment patterns sustain continuous activity in computational modeling, high-throughput screening, and assay development aligned with emerging therapeutic modalities.
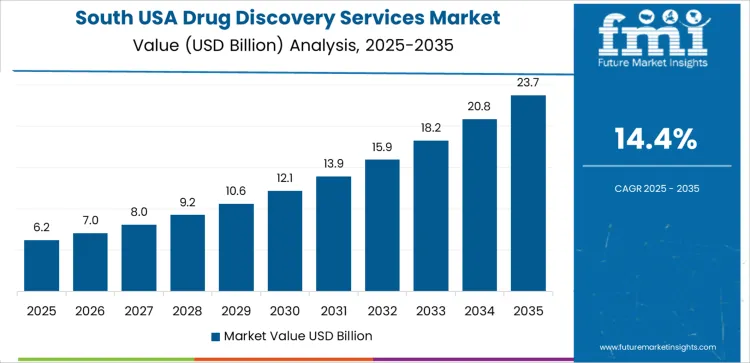
South USA supports rising demand due to its expanding pharmaceutical manufacturing presence and growing ecosystem of contract research organizations. The region’s CAGR of 14.4% reflects increasing dependence on outsourced discovery functions across drug screening, target validation, and preclinical pathway studies. Pharmaceutical clusters use external discovery services to manage pipeline expansion and reduce internal research load. Academic medical centers maintain strong research activity, which drives engagement with specialized discovery partners. Regional expansion of life-science infrastructure supports consistent procurement of high-throughput discovery tools and associated analytical capabilities.
Northeast USA drives demand due to its dense concentration of pharmaceutical companies, academic institutions, and clinical research facilities. The region’s CAGR of 12.8% reflects steady utilization of discovery workflows involving target exploration, compound characterization, and early-stage pharmacology studies. Major research hospitals support translational studies that require structured analytical capabilities from discovery service providers. Established pharmaceutical R&D pipelines create continuous requirements for computational design, screening services, and mechanistic studies. The region maintains consistent activity due to its integration of academic research, commercial R&D, and clinical investigation.
Midwest USA maintains stable demand due to its broad network of biomedical research institutions, healthcare systems, and mid-scale pharmaceutical operations. The region’s CAGR of 11.2% reflects ongoing requirements for early-stage discovery support involving screening, target analysis, and compound assessment. Research universities conduct sustained biomedical studies that generate continuous engagement with discovery platforms. Healthcare-linked research programs rely on specialized services to support molecular studies and early therapeutic exploration. Regional biotech and pharmaceutical firms maintain predictable outsourcing patterns to enhance discovery throughput and support regulated research practices.
Demand for drug discovery services in the USA is shaped by requirements for validated assay platforms, integrated preclinical research, molecular screening capacity, and high-throughput analytical workflows. Biopharmaceutical firms assess data integrity, protocol reproducibility, and scalability across early-stage programs involving target identification, lead optimization, and pharmacology studies. Procurement teams prioritize providers offering multidisciplinary scientific expertise, compliance with regulatory expectations, and stable project management structures supporting complex research portfolios. Engagement patterns reflect the need for specialized instrumentation, bioinformatics capability, and coordinated laboratory networks that reduce internal development timelines. Charles River Laboratories International Inc. participates across discovery biology, in vivo pharmacology, and early safety programs requiring established scientific depth. Thermo Fisher Scientific Inc. maintains visibility through contract research capabilities linked to analytical technologies, custom assay development, and molecular profiling services. Evotec SE contributes through platform-based discovery models involving high-throughput screening, structural biology, and medicinal chemistry programs. Pharmaron Beijing Co., Ltd. supports USA clients through discovery chemistry, DMPK studies, and coordinated project execution using global laboratory resources. Competitive positioning in the USA reflects scientific expertise, data reliability, platform integration, and operational scale supporting diverse early-stage research demands.
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Process | Target Selection, Target Validation, Hit-to-lead Identification, Lead Optimization, Candidate Validation |
| Type | Chemistry Services, Biology Services |
| Drug Type | Small Molecule Drugs, Biologics Drugs |
| Therapeutic Area | Oncology, Neurology, Infectious and Immune Systems Diseases, Digestive System Diseases, Other Therapeutic Areas |
| End User | Pharmaceutical & Biotechnology Companies, Academic Institutes, Manufacturing, Other End Users |
| Regions Covered | West USA, South USA, Northeast USA, Midwest USA |
| Key Companies Profiled | Laboratory Corporation of America Holdings, Charles River Laboratories International Inc., Thermo Fisher Scientific Inc., Evotec SE, Pharmaron Beijing Co., Ltd. |
| Additional Attributes | Dollar sales by process stage, service type, drug type, therapeutic area, and end-user groups; regional adoption trends across West, South, Northeast, and Midwest USA; competitive landscape of CROs specializing in chemistry and biology-based drug discovery; integration with AI-driven screening, high-throughput platforms, and computational drug design; workflow efficiencies in hit identification, lead optimization, and candidate validation for small molecules and biologics. |
How big is the demand for drug discovery services in USA in 2025?
The demand for drug discovery services in USA is estimated to be valued at USD 9.3 billion in 2025.
What will be the size of drug discovery services in USA in 2035?
The market size for the drug discovery services in USA is projected to reach USD 34.3 billion by 2035.
How much will be the demand for drug discovery services in USA growth between 2025 and 2035?
The demand for drug discovery services in USA is expected to grow at a 14.0% CAGR between 2025 and 2035.
What are the key product types in the drug discovery services in USA?
The key product types in drug discovery services in USA are target selection, target validation, hit-to-lead identification, lead optimization and candidate validation.
Which type segment is expected to contribute significant share in the drug discovery services in USA in 2025?
In terms of type, chemistry services segment is expected to command 60.0% share in the drug discovery services in USA in 2025.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.