AI-enabled Drug Discovery Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
AI-enabled Drug Discovery Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
AI-enabled Drug Discovery Market Forecast and Outlook 2026 to 2036
The AI-enabled drug discovery market is expected to grow from USD 8,176.5 million in 2026 to USD 33,951.6 million by 2036, at a CAGR of 15.3%. This market’s economics are shaped by how financial risk is redistributed between drug developers and technology partners. AI platforms absorb higher upfront development and validation costs, while pharmaceutical companies gain flexibility by converting fixed R&D spend into performance-linked engagements. Pricing structures are evolving away from license fees toward milestone-based and asset-participation models, concentrating margins around validated outputs rather than software access.
Cost efficiency is driven by the ability to reuse trained models, pipelines, and datasets across multiple programs, creating operating leverage for platform owners. Smaller biotechs and CROs rely on AI to compete on speed rather than scale, but capture less downstream value. Over the forecast period, durable margin expansion favors AI providers that integrate discovery insight directly into clinical asset generation, not those dependent on standalone computational services.
Quick Stats for AI-Enabled Drug Discovery Market
- AI-Enabled Drug Discovery Market Value (2026): USD 8,176.5 Million
- AI-Enabled Drug Discovery Market Forecast Value (2036): USD 33,951.6 Million
- AI-Enabled Drug Discovery Market Forecast CAGR (2026-2036): 15.3%
- Leading Platform in AI-Enabled Drug Discovery Market: AI Target Identification Platforms (41% share)
- Key Regions in AI-Enabled Drug Discovery Market: Asia Pacific, Europe, North America, Latin America, Middle East & Africa
- Top Key Players in AI-Enabled Drug Discovery Market: Exscientia, Recursion Pharmaceuticals, Insilico Medicine, Schrödinger, Atomwise
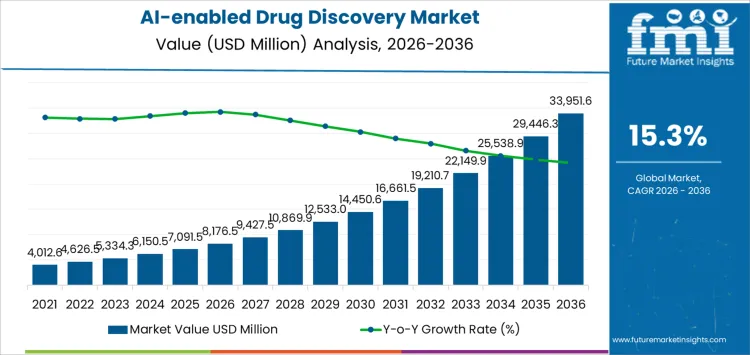
AI-enabled Drug Discovery Market Key Takeaways
| Metric | Value |
|---|---|
| AI-enabled Drug Discovery Market Value (2026) | USD 8,176.5 Million |
| AI-enabled Drug Discovery Market Forecast Value (2036) | USD 33,951.6 Million |
| AI-enabled Drug Discovery Market Forecast CAGR (2026-2036) | 15.3% |
How Are Pipeline Productivity Pressures and Platform Economics Accelerating the AI-enabled Drug Discovery Market?
The AI-enabled drug discovery market is expanding rapidly as pharmaceutical and biotechnology companies seek to improve pipeline efficiency amid rising R&D costs and declining success rates. AI platforms are increasingly deployed to accelerate target identification, hit discovery, and lead optimization, and candidate selection by analyzing vast biological, chemical, and clinical datasets. For drug developers, the primary value lies in reducing early-stage attrition, shortening discovery timelines, and prioritizing high-probability assets before entering costly preclinical and clinical phases.
Commercial adoption is being shaped by platform scalability and integration depth rather than algorithm novelty alone. Pharma companies are favoring AI solutions that integrate seamlessly with existing discovery workflows, wet-lab validation processes, and proprietary data environments. This is driving demand for end-to-end discovery platforms, hybrid AI-human models, and collaborative partnerships where AI providers share risk and upside through milestone-based or asset-linked agreements. Milestone-based and asset-participation models including shared-risk contracting, shape how discovery work is priced and governed across external partners. Vendors that can demonstrate real-world pipeline impact and reproducible outcomes are gaining stronger traction than standalone tool providers.
Ecosystem economics and data strategy are further strengthening market momentum. Expansion of multimodal datasets, improved compute infrastructure, and increasing regulatory comfort with AI-supported discovery are lowering adoption barriers. In parallel, transcriptomics technologies are strengthening model inputs by mapping expression changes that sharpen disease-pathway inference and target confidence. At the same time, CROs and biotech startups are using AI platforms to compete with larger players by accelerating asset generation and out-licensing opportunities. For stakeholders across the value chain, competitive advantage in the AI-enabled drug discovery market increasingly depends on data access, validation credibility, and partnership models that translate computational insight into tangible pipeline and revenue outcomes.
How Is the AI-Enabled Drug Discovery Market Structured Across Key Segments?
The AI-enabled drug discovery market is segmented by platform and application, reflecting how artificial intelligence is applied across different stages of the drug development pipeline. By platform, Ai target identification platforms lead adoption, as early-stage target discovery determines downstream success rates and development timelines. Other platforms include Al lead optimization platforms and Ai clinical trial design tools, which support molecule refinement and trial efficiency. By application, small-molecule drug discovery represents the largest use case, driven by extensive compound libraries and well-established medicinal chemistry workflows. Biologics and antibodies, drug repurposing, and rare disease programs expand adoption across diverse therapeutic strategies.
Why Do AI Target Identification Platforms Lead Platform Adoption in the AI-Enabled Drug Discovery Market?
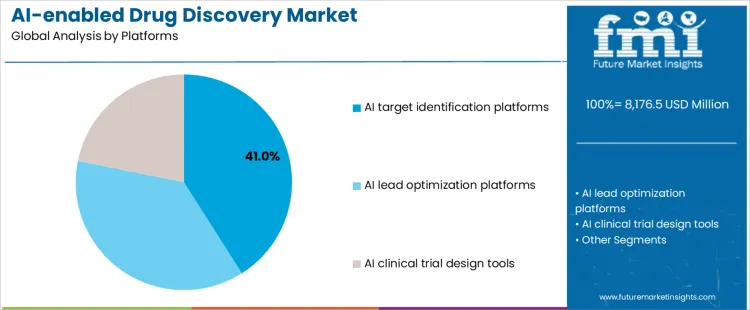
Ai target identification platforms account for 41% share of the ai-enabled drug discovery market because identifying viable biological targets remains the most critical and resource-intensive phase of drug development. These platforms analyze genomic, proteomic, and disease pathway data to uncover novel targets with higher clinical relevance. Pharmaceutical companies use ai-driven target discovery to reduce false positives and focus research investment on mechanisms with stronger validation signals. Early integration of ai improves pipeline efficiency and increases the probability of downstream success. For platform providers, target identification tools enable deep data integration and long-term partnerships with drug developers. These scientific and commercial advantages explain their leading position.
Why Does Small-Molecule Drug Discovery Dominate Application Demand In The AI-Enabled Drug Discovery Market?
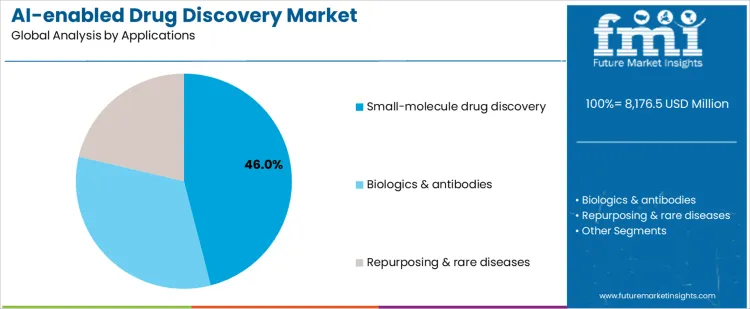
Small-molecule drug discovery holds 46% share of the ai-enabled drug discovery market because it represents the largest and most mature segment of pharmaceutical R&D. Extensive historical datasets on chemical structures, bioactivity, and toxicity allow ai models to deliver accurate predictions. Ai tools accelerate hit identification, structure-activity relationship analysis, and lead optimization within small-molecule programs. Pharmaceutical companies prioritize small molecules due to scalable manufacturing and broad therapeutic applicability. High program volumes generate continuous demand for ai-driven screening and design platforms. These data availability, scalability, and development efficiency factors explain why small-molecule drug discovery remains the dominant application segment.
What Are The Key Dynamics Of The AI-Enabled Drug Discovery Market?
The AI-enabled drug discovery market is driven by pharmaceutical and biotechnology companies seeking to accelerate R&D timelines, reduce discovery costs, and improve success rates of candidate molecules. AI technologies such as machine learning, deep learning, and natural language processing are applied to target identification, virtual screening, de-novo molecular design, and predictive toxicology. Integration with high-throughput screening data, omics datasets, and real-world evidence enhances model training and predictive accuracy. For solution providers, the ability to deliver validated algorithms, interoperable data pipelines, and regulatory readiness support are key differentiators. Partnerships with academia, CROs, and large pharma expand use cases and increase technology adoption across discovery stages.
How Are Technological Trends Shaping Adoption In This Market?
Technological trends are shaping the AI-enabled drug discovery market through improved algorithm sophistication, richer biomedical datasets, and expanded automation. As validation workloads increase, AI in laboratory solution deployments are being evaluated to streamline data capture, QC, and experiment scheduling across discovery teams. Graph neural networks and generative models are enabling more nuanced prediction of protein-ligand interactions and de-novo molecule generation with desired properties. Integration of AI with high-content screening, phenotypic assay data, and structural biology accelerates identification of high-value targets and optimized leads. Cloud computing and scalable infrastructure support large-scale model training and real-time analytics. For AI platform providers, offering tailored workflows, secure data governance, and explainable AI outputs strengthens trust with discovery teams and facilitates broader enterprise deployment.
Why Do Data, Validation, And Regulatory Challenges Restrain Market Growth?
Data quality, validation requirements, and regulatory uncertainty restrain growth in the AI-enabled drug discovery market. Machine learning models require large volumes of curated, high-quality biomedical data, but data heterogeneity, silos, and inconsistent metadata standards limit model performance. Prospective validation of AI-generated candidates through laboratory experiments remains costly and time-intensive, requiring integrated workflows between computational and wet-lab teams. Regulatory frameworks for AI-assisted discovery outputs are still evolving, which complicates adoption of AI platforms in regulated R&D environments where auditability and traceability are essential. This pushes some sponsors to lean more on early phase clinical trial outsourcing partners that already operate under established compliance systems for protocol execution and documentation. These challenges increase implementation risk, extend technology qualification cycles, and limit uptake among risk-averse organizations.
What is The Country-Level Overview And Growth Outlook For The AI-Enabled Drug Discovery Market?
The AI-enabled drug discovery market is expanding rapidly as pharmaceutical companies integrate machine learning, deep learning, and data-driven modeling into target identification, lead optimization, and clinical candidate selection. These platforms reduce discovery timelines, improve success probabilities, and lower R&D costs by analyzing large-scale biological, chemical, and clinical datasets. Country-level growth varies based on pharmaceutical R&D intensity, availability of high-quality biomedical data, regulatory openness to AI-driven workflows, and maturity of digital health ecosystems. Advanced markets emphasize model validation, integration with existing discovery pipelines, and collaboration between AI vendors and large pharma, while high-growth regions focus on rapid platform adoption, contract research integration, and scaling AI tools across multiple therapeutic areas.
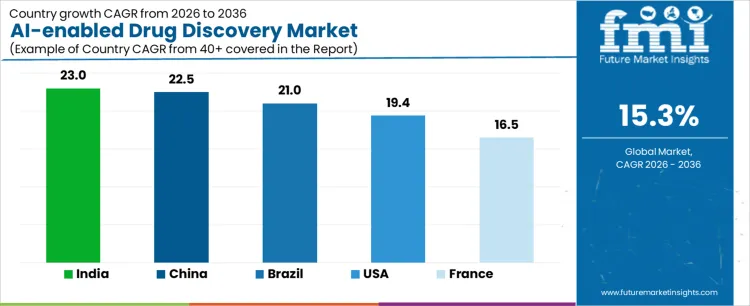
| Country | CAGR (%) |
|---|---|
| India | 23.0 |
| China | 22.5 |
| Brazil | 21.0 |
| USA | 19.4 |
| France | 16.5 |
How is The United States Commercializing AI-Enabled Drug Discovery at Scale?
The AI-enabled drug discovery market in the United States is expanding at a CAGR of 19.4% during 2026-2036, driven by deep integration of AI platforms into established pharmaceutical R&D pipelines. Large pharma companies and biotech firms deploy AI to accelerate target identification, molecular design, and lead optimization across oncology, immunology, and rare disease programs. Buyers emphasize model accuracy, explainability, and seamless integration with existing wet-lab and clinical workflows. Adoption is collaboration-led, with AI vendors working closely with discovery teams rather than operating as standalone solution providers. Qualification cycles focus on demonstrated predictive performance and reproducibility. Procurement decisions prioritize platforms with strong validation records, proprietary datasets, and regulatory awareness. Market value is created through measurable reductions in discovery timelines and attrition rates. The USA represents a maturity-driven market where success depends on scientific credibility, data depth, and the ability to deliver repeatable discovery outcomes at scale.
Why is Brazil Emerging As A Fast-Growing AI Drug Discovery Market?
Demand for AI-enabled drug discovery in Brazil is growing at a CAGR of 21.0% during 2026-2036, supported by expanding pharmaceutical research activity and increasing adoption of digital tools by CROs and academic institutions. AI platforms are primarily used to enhance early-stage discovery efficiency, virtual screening, and repurposing of known compounds. Buyers focus on affordability, usability, and rapid deployment rather than highly customized solutions. Adoption is partnership-driven, often involving public research institutes, local biopharma companies, and international technology providers. Data availability is improving, enabling broader AI application across infectious diseases, metabolic disorders, and neglected diseases. Procurement decisions favor flexible platforms that can integrate with limited in-house infrastructure. Qualification cycles are relatively short, encouraging experimentation and iterative use. Brazil represents a developing but fast-moving market where growth is driven by accessibility, collaboration, and the ability to democratize AI-driven discovery capabilities.
Why is China Accelerating AI-Enabled Drug Discovery Adoption Rapidly?
Adoption of AI-enabled drug discovery platforms in China is expanding at a CAGR of 22.5% during 2026-2036, driven by strong government support, large biomedical datasets, and rapid digitization of pharmaceutical R&D. Chinese biopharma companies deploy AI to shorten discovery cycles, improve candidate selection, and compete globally in innovative drug development. Buyers emphasize speed, scalability, and the ability to process massive genomic and clinical datasets. Adoption is aggressive, with AI platforms embedded early in discovery pipelines across multiple therapeutic areas. Local AI firms play a significant role, supported by strong computational infrastructure and talent availability. Procurement favors high-throughput platforms, localized development, and fast iteration. Qualification cycles are compressed, with performance validated through rapid progression into preclinical stages. China represents a scale- and speed-driven market where competitive advantage depends on data access, computational power, and rapid translation from algorithms to drug candidates.
Why is India The Fastest-Growing Market For AI-Enabled Drug Discovery?
AI-enabled drug discovery market in India is projected to grow at a CAGR of 23.0% during 2026-2036, driven by the convergence of a strong IT ecosystem and a large pharmaceutical manufacturing base. AI platforms are increasingly adopted by generic drug makers, biotech startups, and CROs to enhance target discovery, molecule screening, and cost-efficient R&D. Buyers prioritize affordability, flexible deployment, and compatibility with outsourced research models. Adoption is largely greenfield-led, with AI integrated early into new discovery programs rather than retrofitted into legacy systems. Qualification cycles are short, enabling rapid experimentation across multiple disease areas. Procurement decisions favor vendors offering modular platforms, local technical support, and fast onboarding. Market value is driven by cost efficiency, speed, and the ability to scale discovery efforts across diverse pipelines. India represents a high-growth market where accessibility and execution speed define success.
How is France Applying Structured Validation To AI-Enabled Drug Discovery?
Demand for AI-enabled drug discovery in France is growing at a CAGR of 16.5% during 2026-2036, characterized by cautious, research-driven adoption. French pharmaceutical companies and research institutions emphasize scientific validation, data governance, and regulatory alignment when integrating AI into discovery workflows. AI tools are used extensively in target identification, biomarker discovery, and molecule optimization, often within collaborative research frameworks. Buyers prioritize transparency, explainability of models, and compatibility with academic and clinical research standards. Adoption progresses through pilot programs and consortium-led initiatives rather than rapid enterprise-wide rollout. Procurement decisions favor vendors with strong scientific credentials, peer-reviewed validation, and long-term collaboration models. Qualification timelines are deliberate, reflecting a risk-aware approach. France represents a rigor-driven market where trust, reproducibility, and regulatory readiness outweigh speed of commercialization.
What Shapes Competition in the AI-Enabled Drug Discovery Market?

Competition in the AI-enabled drug discovery market is driven by algorithm accuracy, data integration strength, and demonstrated translational impact captured in official scientific publications and company product information. Exscientia positions itself with platform capabilities that combine deep learning with human-in-the-loop design, showcasing AI-designed molecules that progress rapidly into clinical stages. Its materials emphasize target-agnostic generative design engines and automated optimization loops that reduce cycle times from hit to lead.
Recursion Pharmaceuticals competes with a phenotype-driven AI platform that fuses high-content imaging with machine learning to identify novel biological signatures. Company literature highlights systematic pattern recognition across massive biological datasets and validated programs advancing into therapeutic pipelines. Insilico Medicine differentiates through integrated generative chemistry, bioinformatics, and multi-omics analytics that target unexplored chemical space; official research collateral underscores AI-predicted pathways with rapid lead scoring and synthesis prioritization.
Physics-based and hybrid modeling approaches define another competitive axis. Schrödinger leverages its proprietary computational physics engines combined with machine learning, with documentation emphasizing accurate binding affinity predictions and physics-guided candidate ranking that support medicinal chemistry workflows. Atomwise brings deep convolutional neural network models to virtual screening, with published data demonstrating scalable prediction of ligand-target interactions across diverse target classes.
Key Players in AI-enabled Drug Discovery Market
- Exscientia
- Recursion Pharmaceuticals
- Insilico Medicine
- Schrodinger
- Atomwise
- Others
Scope of Report
| Attribute | Description |
|---|---|
| Quantitative Unit | USD Million |
| Platforms | AI Target Identification Platforms, AI Lead Optimization Platforms, AI Clinical Trial Design Tools |
| Applications | Small-Molecule Drug Discovery, Biologics & Antibodies, Repurposing & Rare Diseases |
| Countries Covered | China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Rest of Asia Pacific, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, Rest of Europe, United States, Canada, Mexico, Brazil, Chile, Rest of Latin America, Kingdom of Saudi Arabia, Other GCC Countries, Turkey, South Africa, Other African Union, Rest of Middle East & Africa |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Key Companies Profiled | Exscientia, Recursion Pharmaceuticals, Insilico Medicine, Schrödinger, Atomwise, Others |
| Additional Attributes | Dollar sales by platform and application are evaluated across AI-driven pharmaceutical R&D workflows. The report includes country-level demand assessment, growth projections for 2026-2036, analysis of model accuracy and data integration, evaluation of pipeline acceleration benefits, competitive benchmarking of AI platforms, and assessment of adoption trends across biotech, pharma, and research organizations. |
AI-enabled Drug Discovery Market by Key Segments
Platforms
- AI target identification platforms
- AI lead optimization platforms
- AI clinical trial design tools
Applications
- Small-molecule drug discovery
- Biologics & antibodies
- Repurposing & rare diseases
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- European Medicines Agency. (2024, September 9). Reflection paper on the use of artificial intelligence (AI) in the medicinal product lifecycle (EMA/CHMP/CVMP/83833/2023). European Medicines Agency.
- Organisation for Economic Co-operation and Development. (2023). Artificial intelligence in science: Challenges, opportunities and the future of research. OECD Publishing. doi:10.1787/a8d820bd-en
- U.S. Food and Drug Administration. (2025, February). Using artificial intelligence & machine learning in the development of drug & biological products: Discussion paper and request for feedback (May 2023, revised February 2025). U.S. Food and Drug Administration.
Frequently Asked Questions
How big is the ai-enabled drug discovery market in 2026?
The global ai-enabled drug discovery market is estimated to be valued at USD 8,176.5 million in 2026.
What will be the size of ai-enabled drug discovery market in 2036?
The market size for the ai-enabled drug discovery market is projected to reach USD 33,951.6 million by 2036.
How much will be the ai-enabled drug discovery market growth between 2026 and 2036?
The ai-enabled drug discovery market is expected to grow at a 15.3% CAGR between 2026 and 2036.
What are the key product types in the ai-enabled drug discovery market?
The key product types in ai-enabled drug discovery market are ai target identification platforms, ai lead optimization platforms and ai clinical trial design tools.
Which applications segment to contribute significant share in the ai-enabled drug discovery market in 2026?
In terms of applications, small-molecule drug discovery segment to command 46.0% share in the ai-enabled drug discovery market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Platforms
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Platforms , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Platforms , 2026 to 2036
- AI target identification platforms
- AI lead optimization platforms
- AI clinical trial design tools
- AI target identification platforms
- Y to o to Y Growth Trend Analysis By Platforms , 2021 to 2025
- Absolute $ Opportunity Analysis By Platforms , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Applications
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Applications, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Applications, 2026 to 2036
- Small-molecule drug discovery
- Biologics & antibodies
- Repurposing & rare diseases
- Small-molecule drug discovery
- Y to o to Y Growth Trend Analysis By Applications, 2021 to 2025
- Absolute $ Opportunity Analysis By Applications, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Platforms
- By Applications
- By Country
- Market Attractiveness Analysis
- By Country
- By Platforms
- By Applications
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Platforms
- By Applications
- By Country
- Market Attractiveness Analysis
- By Country
- By Platforms
- By Applications
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Platforms
- By Applications
- By Country
- Market Attractiveness Analysis
- By Country
- By Platforms
- By Applications
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Platforms
- By Applications
- By Country
- Market Attractiveness Analysis
- By Country
- By Platforms
- By Applications
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Platforms
- By Applications
- By Country
- Market Attractiveness Analysis
- By Country
- By Platforms
- By Applications
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Platforms
- By Applications
- By Country
- Market Attractiveness Analysis
- By Country
- By Platforms
- By Applications
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Platforms
- By Applications
- By Country
- Market Attractiveness Analysis
- By Country
- By Platforms
- By Applications
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Platforms
- By Applications
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Platforms
- By Applications
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Platforms
- By Applications
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Platforms
- By Applications
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Platforms
- By Applications
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Platforms
- By Applications
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Platforms
- By Applications
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Platforms
- By Applications
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Platforms
- By Applications
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Platforms
- By Applications
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Platforms
- By Applications
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Platforms
- By Applications
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Platforms
- By Applications
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Platforms
- By Applications
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Platforms
- By Applications
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Platforms
- By Applications
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Platforms
- By Applications
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Platforms
- By Applications
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Platforms
- By Applications
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Platforms
- By Applications
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Platforms
- By Applications
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Platforms
- By Applications
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Platforms
- By Applications
- Competition Analysis
- Competition Deep Dive
- Exscientia
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Recursion Pharmaceuticals
- Insilico Medicine
- Schrodinger
- Atomwise
- Others
- Exscientia
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Platforms , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Platforms , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Platforms , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Platforms , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Platforms , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Platforms , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Platforms , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Platforms , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Applications, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Platforms , 2026 and 2036
- Figure 4: Global Market Y to o to Y Growth Comparison by Platforms , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Platforms
- Figure 6: Global Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 7: Global Market Y to o to Y Growth Comparison by Applications, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Applications
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y to o to Y Growth Comparison by Region, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Platforms , 2026 and 2036
- Figure 21: North America Market Y to o to Y Growth Comparison by Platforms , 2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by Platforms
- Figure 23: North America Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 24: North America Market Y to o to Y Growth Comparison by Applications, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Applications
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Platforms , 2026 and 2036
- Figure 28: Latin America Market Y to o to Y Growth Comparison by Platforms , 2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by Platforms
- Figure 30: Latin America Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 31: Latin America Market Y to o to Y Growth Comparison by Applications, 2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by Applications
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Platforms , 2026 and 2036
- Figure 35: Western Europe Market Y to o to Y Growth Comparison by Platforms , 2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by Platforms
- Figure 37: Western Europe Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 38: Western Europe Market Y to o to Y Growth Comparison by Applications, 2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by Applications
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Platforms , 2026 and 2036
- Figure 42: Eastern Europe Market Y to o to Y Growth Comparison by Platforms , 2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Platforms
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 45: Eastern Europe Market Y to o to Y Growth Comparison by Applications, 2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Applications
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Platforms , 2026 and 2036
- Figure 49: East Asia Market Y to o to Y Growth Comparison by Platforms , 2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by Platforms
- Figure 51: East Asia Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 52: East Asia Market Y to o to Y Growth Comparison by Applications, 2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by Applications
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Platforms , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y to o to Y Growth Comparison by Platforms , 2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Platforms
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y to o to Y Growth Comparison by Applications, 2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Applications
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Platforms , 2026 and 2036
- Figure 63: Middle East & Africa Market Y to o to Y Growth Comparison by Platforms , 2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Platforms
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 66: Middle East & Africa Market Y to o to Y Growth Comparison by Applications, 2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Applications
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

