The demand for thromboprophylactic drugs in the USA is expected to grow from USD 1.7 billion in 2025 to USD 3.3 billion by 2035, reflecting a compound annual growth rate (CAGR) of 6.6%. Thromboprophylactic drugs are used to prevent blood clots, particularly in patients at risk of venous thromboembolism (VTE), often due to surgery, immobility, or underlying health conditions.
As awareness of thrombotic risks increases, particularly in aging populations and high-risk patients, the demand for thromboprophylaxis is expected to grow steadily over the forecast period, driven by advancements in anticoagulant therapies and an increasing number of procedures that necessitate clot prevention.
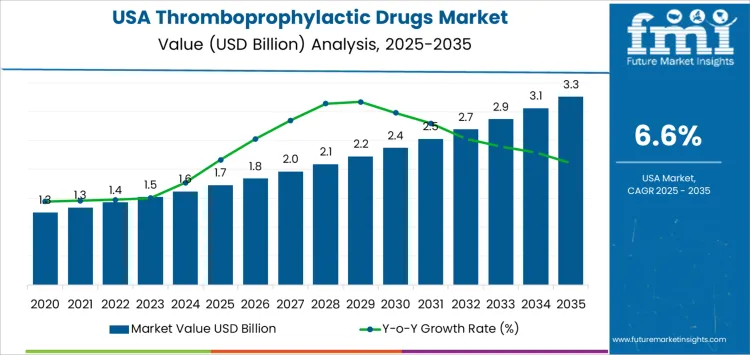
The market will experience gradual growth, starting at USD 1.7 billion in 2025 and increasing to USD 1.8 billion in 2026, USD 2.0 billion in 2027, and USD 2.1 billion in 2028. By 2029, the demand for thromboprophylactic drugs will rise to USD 2.2 billion, continuing its upward trajectory through the 2030s. By 2035, the market is expected to reach USD 3.3 billion, driven by continued advances in drug development, increased adoption of preventive therapies, and growing recognition of the benefits of thromboprophylaxis in various patient groups.
The contribution of volume vs price growth in the thromboprophylactic drugs market indicates that volume growth will play a more significant role in the overall market expansion, particularly in the early years of the forecast period (2025-2029). As the awareness of thrombotic risks increases, more patients will require thromboprophylaxis, contributing to a steady rise in volume demand. This volume-driven growth will be primarily supported by the expanding use of these drugs in preventive care for high-risk populations and surgical patients.
On the other hand, price growth will contribute to the overall market increase in the latter part of the forecast period (2030-2035), driven by the introduction of new, more effective, and specialized thromboprophylactic drugs. These may include novel anticoagulants or extended-release formulations that command higher prices due to their enhanced efficacy, convenience, or reduced side effects. While price increases will contribute to overall market growth, volume expansion remains the key driver, particularly as the adoption of thromboprophylactic therapies becomes more widespread in both the inpatient and outpatient settings. The combined effect of volume growth from increased patient awareness and price growth from new drug innovations will drive the overall market increase.
| Metric | Value |
|---|---|
| Industry Sales Value (2025) | USD 1.7 billion |
| Industry Forecast Value (2035) | USD 3.3 billion |
| Industry Forecast CAGR (2025-2035) | 6.6% |
Demand for thromboprophylactic drugs in the USA is rising due to increasing incidence of conditions that predispose to clot formation such as venous thromboembolism (VTE), deep vein thrombosis (DVT), and pulmonary embolism (PE). Each year hundreds of thousands of Americans suffer from these disorders, and many more face elevated risk because of age, sedentary lifestyles, surgeries, obesity, or cardiovascular disease.
Medical practitioners recognise thromboprophylaxis as a critical preventive measure against clot-related morbidity and mortality. This recognition, coupled with rising disease burden, expands the use of anticoagulants, antiplatelet agents, and other clot-prevention therapies among hospitalised patients, post-surgery patients, and individuals with chronic risk factors.
At the same time advances in drug development and improvements in patient care protocols support greater uptake. Newer oral anticoagulants offer more convenience than older therapies because they require less frequent monitoring, have fewer dietary and drug interactions, and tend to improve patient adherence. Meanwhile evolving clinical guidelines recommend prophylactic use in a broader set of at-risk populations, including those undergoing major surgery, requiring long-term immobility, or living with chronic conditions.
Growing awareness among clinicians and patients about the benefits of preventing clot formation before onset rather than only treating after episodes further enlarges the addressable population. As population ages, lifestyle-related risk factors increase, and medical standards emphasise prevention, demand for thromboprophylactic drugs in the USA is likely to grow steadily in coming years.
The demand for thromboprophylactic drugs in the USA is primarily driven by drug class and end user. The leading drug class is Direct Oral Anticoagulants (DOACs), which accounts for 39% of the market share, while hospitals dominate the end-user segment, capturing 61.8% of the demand. Thromboprophylactic drugs are essential for preventing blood clots in patients at risk for thrombosis, such as those undergoing surgery or suffering from conditions like deep vein thrombosis (DVT) or pulmonary embolism (PE). As the prevalence of clotting disorders and the need for preventive measures grow, the demand for these drugs continues to rise in the USA.
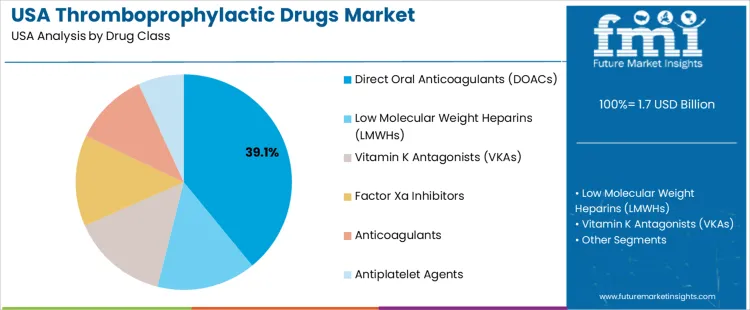
Direct Oral Anticoagulants (DOACs) lead the demand for thromboprophylactic drugs in the USA, holding 39% of the market share. DOACs, including apixaban, rivaroxaban, and dabigatran, have become the preferred choice for thromboprophylaxis due to their ease of use, predictable dosing, and fewer dietary restrictions compared to traditional anticoagulants. DOACs offer significant advantages in terms of patient compliance, as they do not require routine monitoring of blood levels, unlike vitamin K antagonists (VKAs) or low molecular weight heparins (LMWHs).
The growing adoption of DOACs is driven by their superior safety profile, especially their lower risk of major bleeding events and the convenience of oral administration. As the population ages and the incidence of conditions requiring thromboprophylaxis, such as atrial fibrillation, increases, DOACs are becoming the go-to option for healthcare providers. With continued advancements in DOAC formulations and the increasing focus on preventing thrombotic events in high-risk patients, the demand for DOACs is expected to remain strong in the thromboprophylactic drug market in the USA.
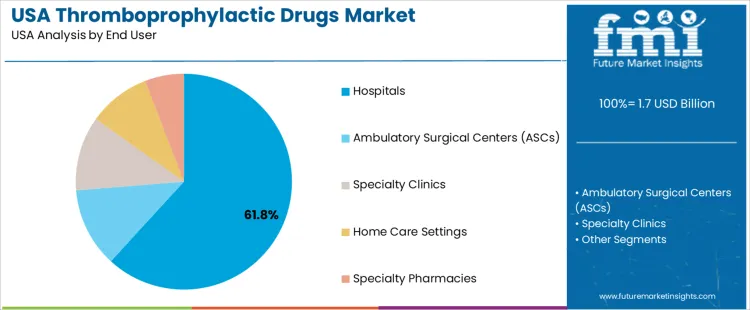
Hospitals lead the end-user demand for thromboprophylactic drugs in the USA, capturing 61.8% of the market share. Hospitals play a central role in prescribing and administering thromboprophylactic drugs, particularly for patients undergoing major surgeries or those at high risk of thromboembolic events. In hospital settings, thromboprophylaxis is essential in preventing complications such as deep vein thrombosis (DVT) and pulmonary embolism (PE) in postoperative patients, particularly after orthopedic surgeries, abdominal surgeries, or cancer treatments.
The demand from hospitals is driven by the high volume of patients requiring thromboprophylaxis, as well as the healthcare facility's capacity to monitor and adjust treatment protocols for complex cases. Hospitals also play a key role in administering newer anticoagulants like DOACs, which are often preferred for their ease of use and lower risk of adverse events compared to older therapies. With the ongoing efforts to improve patient outcomes and reduce complications in high-risk populations, hospitals will continue to be the dominant end-user segment for thromboprophylactic drugs in the USA.
Demand for thromboprophylactic drugs in the USA is increasing consistently as the burden of cardiovascular and thromboembolic diseases rises. Many patients are at risk of venous thromboembolism (VTE), deep-vein thrombosis (DVT), pulmonary embolism (PE), atrial fibrillation and other clot-related conditions. As population ages and risk factors such as obesity, sedentary lifestyle, and chronic diseases grow, more individuals require preventive treatment or long-term anticoagulation therapy. This has elevated demand for anticoagulant and antithrombotic medications across hospitals, clinics, outpatient settings and long-term care.
One major driver is the rising prevalence of cardiovascular conditions, atrial fibrillation, and venous thromboembolic disorders in the population, which increases the need for clot-prevention therapy. Another driver is growing awareness and medical guidelines recommending prophylactic anticoagulation in high-risk groups - for example post-surgical patients, immobile or hospitalized individuals, and older adults with comorbidities.
The increased use of modern oral anticoagulants with predictable dosing, fewer dietary restrictions and reduced need for regular blood monitoring has made prophylaxis more convenient and acceptable for patients and physicians. Advances in drug development and approval of new anticoagulants have expanded options and widened the eligible patient base. In addition, expanding use of outpatient care and home-based management of chronic conditions encourages long-term thromboprophylactic treatment outside hospital settings.
Despite growing need, some factors restrain demand. The risk of side effects - especially bleeding - remains a concern for both patients and physicians, which may limit use in certain groups. Cost of newer anticoagulants may be higher than older therapies, making adherence or long-term use difficult for some patients. Some individuals at risk may not be properly identified due to under-diagnosis or lack of screening, which reduces the treated population.
In addition, patient compliance to long-term medication regimens can be challenging, especially when frequent monitoring or lifestyle adjustments are required. Finally, medical or regulatory caution in prophylactic drug use - especially for lower-risk populations - may limit over-prescription, reducing overall demand growth.
A key trend is the growing shift toward use of direct oral anticoagulants (DOACs) and newer anticoagulants rather than traditional therapies, due to their ease of use, predictable effects and improved patient compliance. There is also increasing adoption of prophylactic anticoagulation in outpatient care, post-surgical care (for example after joint replacements), and long-term management of atrial fibrillation or chronic conditions - expanding the pool of patients receiving thromboprophylaxis.
Advancements in diagnostics and risk-assessment protocols help in identifying at-risk individuals earlier, which promotes preventive treatment. In addition, as the population ages and chronic disease burden rises, more people qualify for thromboprophylactic therapy. Finally, growing emphasis on preventive healthcare and long-term disease management supports steady growth of the market for antithrombotic and thromboprophylactic drugs.
The demand for thromboprophylactic drugs in the USA is expected to increase across all major regions over the coming years. The West shows the highest projected compound annual growth rate (CAGR) at 7.5%. The South follows at 6.8%, the Northeast at 6.0%, and the Midwest at 5.2%. These regional differences reflect variation in population age profiles, incidence of cardiovascular and thrombotic disorders, healthcare infrastructure, and adoption of preventive anticoagulant therapies. Rising prevalence of conditions such as deep vein thrombosis (DVT), atrial fibrillation, and post-surgical prophylaxis supports growth in demand for these medications.
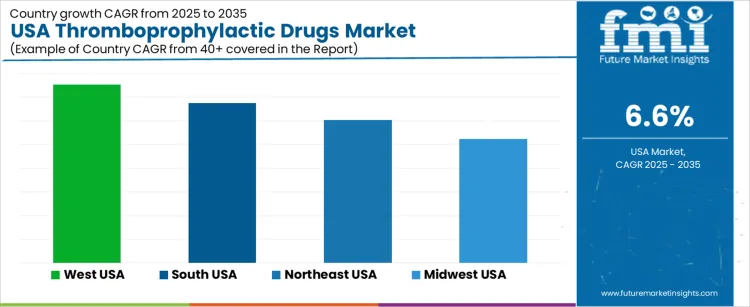
| Region | CAGR (%) |
|---|---|
| West USA | 7.5 |
| South USA | 6.8 |
| Northeast USA | 6.0 |
| Midwest USA | 5.2 |
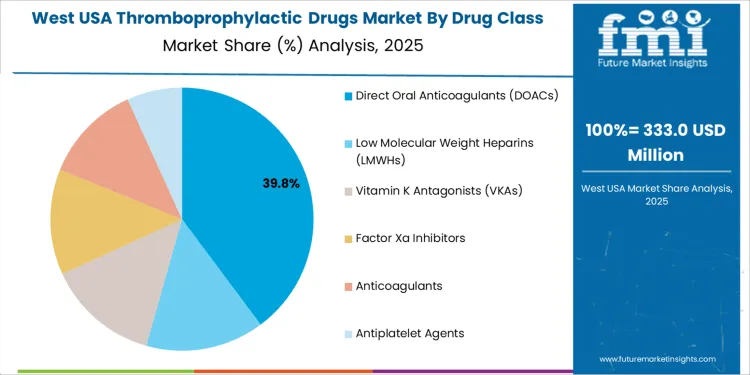
In the West USA, the projected 7.5% annual growth reflects multiple converging factors that elevate demand. The region hosts a mix of urban centers and aging populations. As age advances, conditions such as atrial fibrillation, venous thromboembolism (VTE), and other risk factors for clot formation become more common. Widespread use of direct oral anticoagulants and improved access to healthcare services encourage uptake of preventive therapies.
The growth of surgical procedures and hospital-based interventions that require post-operative prophylaxis also contributes to rising demand. As more patients are identified as high-risk, physicians increasingly prescribe thromboprophylactic drugs to prevent complications like deep vein thrombosis and pulmonary embolism. Combined with heightened awareness of clot-related risks and easier availability of modern anticoagulants, this supports a relatively high growth rate for the West region.
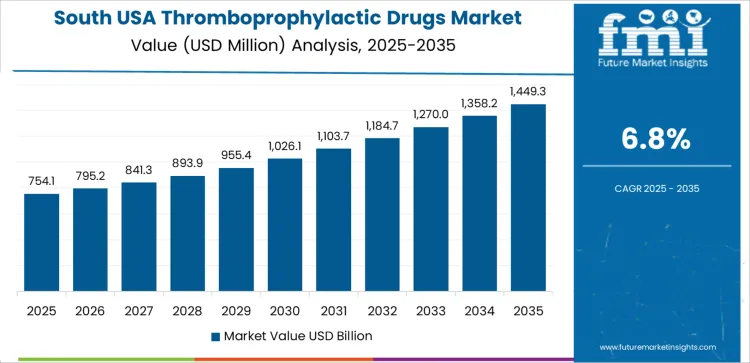
In the South USA, a 6.8% projected CAGR reflects growing recognition of thrombotic disease burden and expanding healthcare access. The region has a substantial population with risk factors such as obesity, diabetes, and sedentary lifestyle that raise the likelihood of clotting events. As medical care and preventive cardiology expand, more patients receive diagnosis and follow-up for conditions requiring long-term anticoagulation or prophylaxis. Post-operative thromboprophylaxis after surgeries and interventions remains a consistent driver. Adoption of newer anticoagulants that offer more convenient dosing and lower monitoring requirements supports wider use. Over time, increasing awareness among physicians and patients about the benefits of preventive therapy contributes to a steady rise in demand across the South.
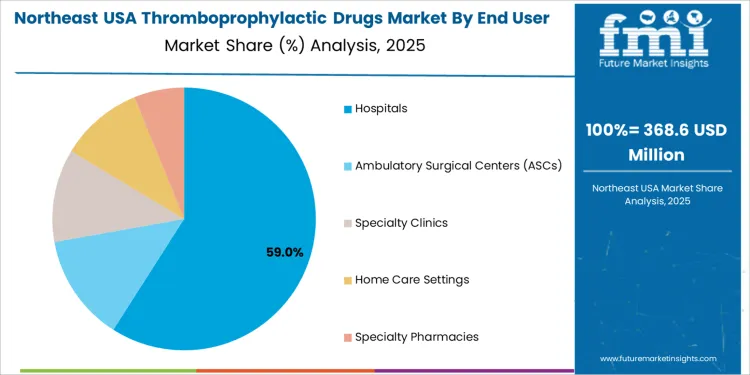
In the Northeast USA, the projected 6.0% CAGR reflects a stable but growing demand for thromboprophylactic drugs. The region is characterized by a significant older population and high rates of cardiovascular disease, atrial fibrillation, and related complications. Hospitals and clinics in this region frequently employ anticoagulant therapy for patients with risk factors for stroke, deep vein thrombosis, or pulmonary embolism.
As diagnostic capabilities improve and guidelines evolve to favour preventive anticoagulation in high-risk individuals, more patients begin long-term management. In addition, growing use of direct oral anticoagulants simplifies treatment regimens and improves patient adherence. Ongoing awareness of thrombosis risk in conditions such as cancer, immobility, and surgical recovery supports demand, leading to steady growth in this region.
In the Midwest USA, the projected 5.2% CAGR indicates moderate but consistent growth in demand for thromboprophylactic drugs. The region’s demographic mix includes both urban and rural populations, with a significant portion of older adults and patients with chronic conditions that increase thrombotic risk. Healthcare infrastructure expansion as well as rising incidence of risk factors such as obesity, diabetes, and sedentary lifestyle contribute to greater need for preventive anticoagulation. Post-surgical prophylaxis, management of atrial fibrillation, and treatment of venous thromboembolism remain key applications. As newer oral anticoagulants become more widely available and cost-effective, adoption rises among physicians and patients, sustaining gradual demand growth in the Midwest.
The demand for thromboprophylactic drugs in the USA is rising, driven by increasing prevalence of cardiovascular disease, aging population, and heightened focus on preventing clot-related disorders such as deep vein thrombosis, pulmonary embolism, atrial fibrillation, and post-surgical thrombosis. Growth in hospitalizations, surgical procedures, chronic disease burden, and long-term care needs supports steady use of anticoagulants and prophylactic therapies across hospitals, clinics, and outpatient settings. The broader anticoagulant and antithrombotic market in North America remains among the largest globally, reflecting robust adoption of clot-prevention regimens. This underpins growing demand for prophylactic drugs rather than reactive treatment alone.
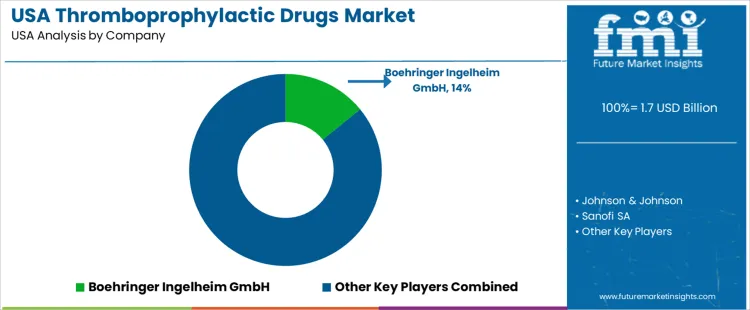
In this competitive environment several established firms hold leading positions. According to your data, Boehringer Ingelheim GmbH controls about 14.1% of the thromboprophylactic market share in the USA. Other significant players include Johnson & Johnson, Sanofi SA, Pfizer Inc., and Bayer AG. These firms supply a range of anticoagulant and antithrombotic agents-direct oral anticoagulants (DOACs), low-molecular-weight heparins, factor Xa inhibitors, and older agents for various prophylactic uses such as surgery and atrial fibrillation.
Competition among these firms focuses on drug efficacy, safety profiles, ease of use, dosing convenience, regulatory approvals, and product availability. Some companies prioritise newer oral agents with fewer monitoring requirements and improved tolerability compared with legacy therapies. Others maintain portfolios with well-established medications widely used in hospitals and outpatient care. Firms also compete to expand distribution across hospitals, ambulatory surgical centers, and retail or specialty pharmacies.
Product brochures and clinical information highlight benefits such as reduced risk of venous thromboembolism, ease of adherence, reduced monitoring needs, and suitability for different patient populations. As cardiovascular disease prevalence remains high and preventive healthcare awareness strengthens, the market for thromboprophylactic drugs in the USA is likely to remain competitive and steadily grow.
| Items | Details |
|---|---|
| Quantitative Units | USD Billion |
| Regions Covered | USA |
| Drug Class | Direct Oral Anticoagulants (DOACs), Low Molecular Weight Heparins (LMWHs), Vitamin K Antagonists (VKAs), Factor Xa Inhibitors, Anticoagulants, Antiplatelet Agents |
| End-User | Hospitals, Ambulatory Surgical Centers (ASCs), Specialty Clinics, Home Care Settings, Specialty Pharmacies |
| Key Companies Profiled | Boehringer Ingelheim GmbH (14.1% market share), Johnson & Johnson, Sanofi SA, Pfizer Inc., Bayer AG |
| Additional Attributes | Dollar sales by drug class and end-user show strong demand for Direct Oral Anticoagulants (DOACs), particularly in hospitals and specialty pharmacies. Hospitals and ambulatory surgical centers lead the demand, while specialty clinics and home care settings also contribute. Companies like Boehringer Ingelheim and Pfizer are key players in the market, offering a range of anticoagulants and antiplatelet agents. The market is expected to grow due to the increasing need for thromboprophylaxis in surgeries, chronic conditions, and other medical procedures, with DOACs driving a significant portion of the growth. |
How big is the demand for thromboprophylactic drugs in USA in 2025?
The demand for thromboprophylactic drugs in USA is estimated to be valued at USD 1.7 billion in 2025.
What will be the size of thromboprophylactic drugs in USA in 2035?
The market size for the thromboprophylactic drugs in USA is projected to reach USD 3.3 billion by 2035.
How much will be the demand for thromboprophylactic drugs in USA growth between 2025 and 2035?
The demand for thromboprophylactic drugs in USA is expected to grow at a 6.6% CAGR between 2025 and 2035.
What are the key product types in the thromboprophylactic drugs in USA?
The key product types in thromboprophylactic drugs in USA are direct oral anticoagulants (doacs), low molecular weight heparins (lmwhs), vitamin k antagonists (vkas), factor xa inhibitors, anticoagulants and antiplatelet agents.
Which end user segment is expected to contribute significant share in the thromboprophylactic drugs in USA in 2025?
In terms of end user, hospitals segment is expected to command 61.8% share in the thromboprophylactic drugs in USA in 2025.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.