Breast Cancer Subtyping Kits Market
Breast Cancer Subtyping Kits Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Breast Cancer Subtyping Kits Market Forecast and Outlook 2026 to 2036
The breast cancer subtyping kits market is likely to be valued at USD 4.8 billion in 2026 and reach USD 12.0 billion by 2036, advancing at a CAGR of 9.6%. Growth is anchored in the structural role subtyping plays in modern breast cancer care, where treatment eligibility, sequencing, and prognosis depend on verified receptor and molecular status. Subtyping has become a compulsory diagnostic step rather than an optional refinement, directly influencing access to endocrine therapy, HER2-targeted regimens, and emerging immuno-oncology approaches. Testing demand is therefore tied to treatment initiation rates and therapy complexity rather than screening volumes alone. Hospitals and reference laboratories allocate spending toward kits that deliver consistent ER, PR, HER2, and proliferation readouts across high sample throughput, supporting predictable clinical decision timelines.
Utilization patterns are shaped by workflow efficiency and diagnostic standardization rather than incremental increases in case incidence. Pathology services prioritize kits that integrate into existing IHC and ISH platforms, minimizing disruption while maintaining scoring consistency across sites. Expansion of centralized laboratory models and automated staining systems further reinforces repeat kit consumption at scale. Clinical reliance on subtype confirmation at diagnosis and recurrence extends testing beyond initial biopsy, supporting recurring demand across disease stages. Reimbursement alignment and guideline enforcement sustain routine use, while quality assurance requirements limit substitution risk. Market durability reflects dependence of breast cancer treatment algorithms on validated subtyping outputs, positioning these kits as essential diagnostic infrastructure embedded within oncology care delivery rather than discretionary consumables.
Quick Stats for Breast Cancer Subtyping Kits Market
- Breast Cancer Subtyping Kits Market Value (2026): USD 4.8 billion
- Breast Cancer Subtyping Kits Market Forecast Value (2036): USD 12.0 billion
- Breast Cancer Subtyping Kits Market Forecast CAGR 2026 to 2036: 9.6%
- Leading Product Type by Utilization: IHC-based kits
- Fastest-Growing Countries: India, China, Brazil, USA, Germany
- Top Players in Global Demand: Roche Diagnostics, Agilent, Abbott, Bio-Rad, Thermo Fisher Scientific
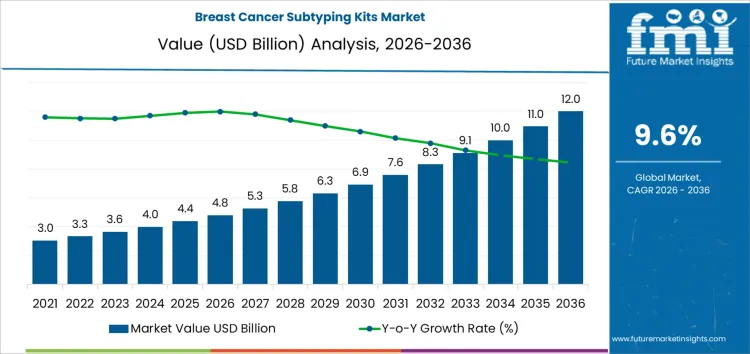
Breast Cancer Subtyping Kits Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 4.8 billion |
| Market Forecast Value (2036) | USD 12.0 billion |
| Forecast CAGR 2026 to 2036 | 9.6% |
Why is the Demand for Breast Cancer Subtyping Kits Growing?
Demand for breast cancer subtyping kits is increasing as oncology practices and diagnostic laboratories integrate precision medicine into treatment selection and prognostic planning. Subtyping kits enable classification of tumors into clinically relevant subgroups such as hormone receptor positive, HER2 enriched, and triple negative phenotypes, which directly influence therapeutic pathways including endocrine therapy, HER2 targeted agents, and chemotherapy selection. Pathology teams prioritize kits that deliver reliable receptor quantification, gene expression profiles, or multi-gene signatures because robust subtype assignment supports clinician decision making and alignment with evidence-based guidelines. Procurement decisions emphasize analytical accuracy, reproducibility across runs, and compatibility with existing laboratory platforms to support consistent diagnostic workflows without requiring extensive new instrumentation or retraining.
Growth in adoption of targeted therapies and immunotherapy in breast cancer management reinforces the need for precise subtyping to match patients with appropriate agents that improve disease control and survival outcomes. Health systems and payers are adjusting coverage frameworks to support early molecular characterization because subtype information can reduce utilization of ineffective therapies and limit unnecessary toxicity. Research advances that correlate specific subtypes with response phenotypes in real-world cohorts encourage broader use of subtyping tools in community and academic settings alike. Laboratories also focus on quality assurance and regulatory compliance for kit performance to ensure consistent results across diverse specimen types and processing conditions. These clinical and operational considerations are contributing to sustained demand growth in the breast cancer subtyping kits market.
How Is the Breast Cancer Subtyping Kits Market Segmented by Product Type, Application, and End User?
Demand for breast cancer subtyping kits is shaped by precision treatment protocols, mandatory biomarker testing, and standardized pathology workflows. Clinical practice requires accurate molecular and protein-level classification to guide therapy selection and prognostic assessment. Adoption reflects guideline-driven testing, reimbursement alignment, and expansion of companion diagnostics. Kit selection considers analytical reliability, turnaround time, and compatibility with existing laboratory infrastructure. Segmentation clarifies how assay format, clinical objective, and testing environment influence utilization patterns, purchasing priorities, and integration within breast cancer diagnostic pathways.
Which Product Type Concentrates Utilization in Breast Cancer Subtyping?

Utilization is highest for IHC-based kits at 46.0%, reflecting their role as frontline tools in routine breast cancer classification. These kits enable visualization of protein expression using established pathology workflows and widely available equipment. Pathologists rely on IHC for its interpretability, cost efficiency, and suitability for high sample volumes. FISH kits at 28.0% support confirmatory analysis, particularly in equivocal HER2 cases requiring gene amplification assessment. PCR-based assays at 16.0% provide molecular-level insights with higher sensitivity for specific targets. Product segmentation reflects reliance on methods balancing diagnostic confidence, workflow familiarity, and scalability.
Key Points:
- IHC kits integrate seamlessly into routine pathology workflows.
- FISH supports confirmatory testing in selected cases.
- PCR assays address targeted molecular questions.
How Do Clinical Subtyping Objectives Shape Application-Level Demand?

Application demand centers on HER2 subtyping at 38.0%, driven by its direct role in determining eligibility for targeted therapies. Accurate HER2 status influences first-line treatment decisions and prognosis. Hormone receptor status at 34.0% remains essential for endocrine therapy planning and disease stratification. Triple-negative identification at 28.0% guides chemotherapy selection and clinical trial consideration due to limited targeted options. Application segmentation highlights stronger utilization where biomarker results directly dictate therapeutic pathways and long-term management strategies.
Key Points:
- HER2 status directly informs targeted treatment decisions.
- Hormone receptor testing supports endocrine therapy planning.
- Triple-negative identification guides alternative treatment strategies.
Which End User Settings Drive Subtyping Kit Utilization?
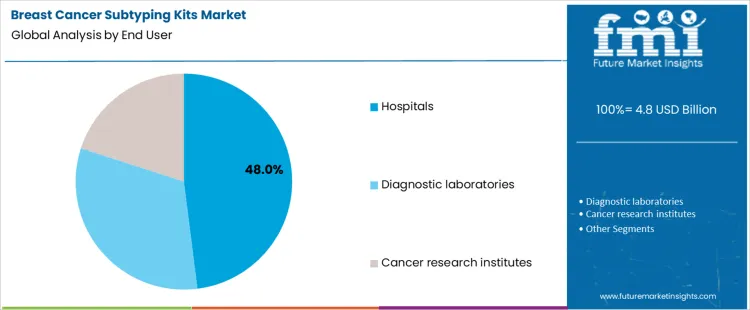
Hospitals account for 48.0% of subtyping kit utilization due to their role in diagnosis, surgical pathology, and treatment initiation. On-site laboratories support timely reporting aligned with clinical decision timelines. Diagnostic laboratories at 32.0% provide centralized testing services for referral networks and outpatient centers. Cancer research institutes at 20.0% use subtyping kits for translational studies and clinical trial enrollment. End-user segmentation reflects concentration where diagnostic authority, sample volume, and infrastructure capability determine testing intensity and procurement focus.
Key Points:
- Hospitals integrate subtyping into diagnostic workflows.
- Diagnostic laboratories serve multi-site testing demand.
- Research institutes apply kits in clinical and translational studies.
What are the Key Dynamics in the Breast Cancer Subtyping Kits Market?
Demand for breast cancer subtyping kits reflects diagnostic need to classify tumors based on molecular and protein expression profiles that guide treatment selection. Adoption concentrates in hospital pathology laboratories, diagnostic reference centers, and oncology-focused testing networks. Global scope aligns with standardized breast cancer care pathways and guideline-mandated testing. Usage centers on immunohistochemistry and molecular assay kits used to determine hormone receptor status, HER2 expression, and proliferation indices.
How do treatment decision dependency and diagnostic standardization shape adoption?
Breast cancer management depends on accurate tumor subtyping to determine suitability for endocrine therapy, targeted agents, or chemotherapy. Demand increases as treatment outcomes vary significantly across molecular subtypes. Subtyping kits enable reproducible assessment of estrogen receptor, progesterone receptor, HER2 status, and proliferation markers using standardized scoring systems. Pathology workflows rely on validated kits to ensure inter-laboratory consistency and regulatory compliance. Turnaround time remains critical to avoid treatment delays following surgery or biopsy. Centralized testing supports uniform quality in regions with limited pathology capacity. Adoption reflects clinical reliance on precise classification to avoid overtreatment or undertreatment across early and advanced disease stages.
How do assay variability, reimbursement pressure, and workflow constraints influence market scalability?
Subtyping accuracy depends on pre-analytical factors including tissue fixation, handling, and staining consistency. Demand sensitivity rises where variability affects result reliability. Interpretation requires trained pathologists, limiting scalability in resource-constrained settings. Reimbursement policies influence kit selection and testing breadth, particularly for additional prognostic markers. Automation compatibility affects laboratory throughput and capital investment requirements. Quality control and proficiency testing add operational cost. Updating kits to align with evolving guidelines increases validation burden. Scalability remains constrained by dependence on skilled interpretation, cost containment pressure, and need for consistent analytical performance across diverse laboratory environments.
How Is Demand for Breast Cancer Subtyping Kits Evolving Globally?
Demand for breast cancer subtyping kits is expanding globally as treatment selection increasingly depends on molecular and receptor-based classification. Clinical pathways emphasize ER, PR, HER2, and proliferation markers to guide targeted and endocrine therapies. Laboratory standardization, automation, and reimbursement alignment support routine testing. Adoption is reinforced by guideline mandates and therapy access expansion. Growth rates in India at 11.0%, China at 10.8%, Brazil at 10.4%, USA at 9.2%, and Germany at 9.1% indicate sustained expansion driven by precision oncology requirements, diagnostic capacity scaling, and protocol adherence rather than population-wide screening intensity.
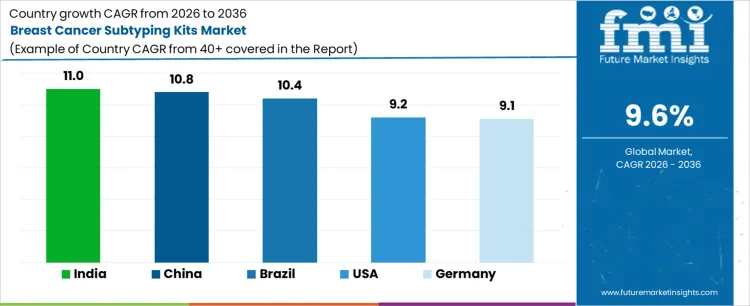
| Country | CAGR (%) |
|---|---|
| India | 11.0% |
| China | 10.8% |
| Brazil | 10.4% |
| USA | 9.2% |
| Germany | 9.1% |
What Is Driving Accelerated Adoption of Breast Cancer Subtyping Kits in India?
Breast cancer subtyping kit demand in India is growing at a CAGR of 11.0%, reflecting rapid integration of receptor testing into oncology workflows. Private cancer centers prioritize in-house IHC and ISH capabilities to shorten turnaround times. Rising early-stage diagnosis increases reliance on accurate subtype classification. Medical oncologists require standardized results before initiating targeted or endocrine therapy. Domestic kit manufacturing improves affordability and supply consistency. Expansion reflects therapy-driven diagnostic necessity and infrastructure build-out rather than shifts in screening participation.
- In-house testing expansion at private centers
- Therapy-mandated ER, PR, HER2 classification
- Earlier-stage diagnosis increasing test volumes
- Improved affordability via domestic production
Why Is China Showing Strong Momentum in Breast Cancer Subtyping Utilization?
Breast cancer subtyping demand in China is expanding at a CAGR of 10.8%, supported by standardized oncology protocols and high patient throughput. Tertiary hospitals embed subtyping within diagnostic algorithms. Reimbursement inclusion for essential markers improves access. Centralized laboratories process large volumes with consistent quality control. Alignment with domestic targeted therapy development reinforces routine testing. Growth reflects protocolized adoption at scale rather than experimental or discretionary use.
- Protocol-embedded subtyping in tertiary hospitals
- Reimbursement support for essential markers
- Centralized laboratory processing efficiency
- Alignment with domestic therapy pipelines
How Is Brazil Sustaining Growth in Breast Cancer Subtyping Kits?
Breast cancer subtyping kit demand in Brazil is growing at a CAGR of 10.4%, influenced by expansion of comprehensive oncology networks. Public and private providers adopt subtype-guided treatment pathways. Increased access to HER2-targeted therapies elevates testing frequency. Laboratory partnerships extend diagnostic reach beyond major cities. Legal access mechanisms occasionally broaden test availability. Growth reflects institutional adoption and therapy access expansion rather than broad screening growth.
- Network expansion of oncology centers
- HER2 therapy access increasing test frequency
- Laboratory partnerships improving regional reach
- Institutional adoption of subtype-guided pathways
What Factors Are Supporting Breast Cancer Subtyping Growth in United States?
Breast cancer subtyping kit demand in the United States is expanding at a CAGR of 9.2%, driven by guideline-led precision oncology. Payer coverage supports routine IHC and reflex ISH testing. High laboratory automation ensures throughput and consistency. Clinical trials and evolving therapy options sustain comprehensive profiling. Quality assurance programs reinforce standardization. Growth reflects protocol adherence and innovation pace rather than increases in disease incidence.
- Guideline-mandated routine subtyping
- Broad payer coverage for IHC and ISH
- Automated laboratories ensuring consistency
- Trial activity sustaining comprehensive profiling
Why Is Germany Experiencing Sustained Expansion in Breast Cancer Subtyping?
Breast cancer subtyping demand in Germany is growing at a CAGR of 9.1%, shaped by evidence-based oncology pathways. Certified breast centers require standardized subtype reporting. Reimbursement frameworks support validated assays. Central pathology networks ensure quality control and comparability. Emphasis on cost-effectiveness guides marker selection. Growth remains steady, reflecting structured adoption and system-wide standardization rather than rapid decentralization.
- Certified center requirements for standardized reporting
- Reimbursement for validated assay panels
- Central pathology network quality control
- Cost-effectiveness guiding marker selection
What is the competitive landscape of demand for the breast cancer subtyping kits market globally?
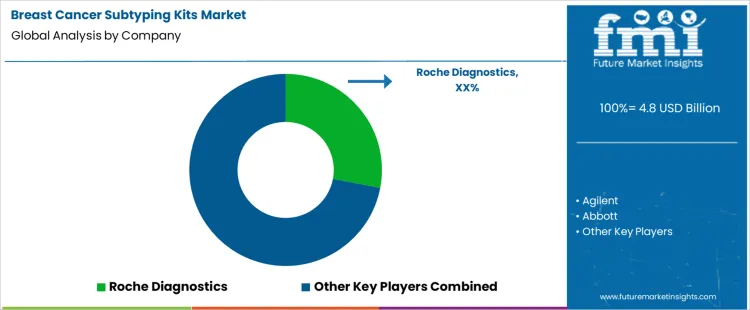
Demand for breast cancer subtyping kits is shaped by personalized treatment pathways and mandatory biomarker stratification at diagnosis. Pathology laboratories evaluate assay accuracy for hormone receptors and HER2 status, reproducibility across sites, turnaround time, and tissue compatibility. Buyer assessment includes regulatory clearance, automation readiness, staining consistency, and integration with digital pathology systems. Procurement behavior reflects centralized testing models, long-term reagent agreements, and preference for kits aligned with clinical guidelines. Purchasing decisions emphasize diagnostic confidence, workflow efficiency, and reimbursement acceptance. Trend in the breast cancer subtyping kits market shows sustained reliance on standardized immunohistochemistry and molecular assays supporting therapy selection.
Roche Diagnostics leads competitive positioning through widely adopted subtyping assays integrated with companion diagnostic frameworks. Agilent competes via high-quality reagents and platforms supporting precise receptor and gene expression analysis. Abbott supports demand with automated diagnostic systems delivering consistent results across high-volume laboratories. Bio-Rad maintains relevance through molecular subtyping tools used in research and clinical validation workflows. Thermo Fisher Scientific participates by supplying flexible assay solutions compatible with diverse laboratory infrastructures. Competitive differentiation centers on diagnostic accuracy, regulatory breadth, automation compatibility, and alignment with evolving breast cancer treatment algorithms.
Key Players in the Breast Cancer Subtyping Kits Market
- Roche Diagnostics
- Agilent
- Abbott
- Bio-Rad
- Thermo Fisher Scientific
- Others
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Product Type | IHC-based kits; FISH kits; PCR-based assays; Others |
| Application | HER2 subtyping; Hormone receptor status; Triple-negative identification |
| End User | Hospitals; Diagnostic laboratories; Cancer research institutes |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, China, Brazil, USA, Germany, and 40+ countries |
| Key Companies Profiled | Roche Diagnostics; Agilent; Abbott; Bio-Rad; Thermo Fisher Scientific; Others |
| Additional Attributes | Dollar sales by product type, application, end user, and sales channel; analytical accuracy and reproducibility of IHC, FISH, and PCR-based breast cancer subtyping assays; workflow efficiency and turnaround time in hospital and reference laboratories; role of biomarker stratification in therapy selection and prognosis assessment; regulatory compliance and companion diagnostic alignment; procurement dynamics shaped by oncology testing volumes, centralized laboratory networks, and research-driven demand. |
Breast Cancer Subtyping Kits Market by Segment
Product Type:
- IHC-based kits
- FISH kits
- PCR-based assays
- Others
Application:
- HER2 subtyping
- Hormone receptor status
- Triple-negative identification
End User:
- Hospitals
- Diagnostic laboratories
- Cancer research institutes
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- European Society for Medical Oncology. (2024). ESMO clinical practice guidelines for breast cancer: Early and metastatic disease. Annals of Oncology.
- European Medicines Agency. (2024). Companion diagnostics and biomarker-driven medicines in oncology. European Medicines Agency.
- Organisation for Economic Co-operation and Development. (2024). Precision oncology and molecular diagnostics in cancer care pathways. OECD Publishing.
- U.S. Food and Drug Administration, Center for Devices and Radiological Health. (2024). In vitro companion diagnostic devices: Regulatory framework and oncology applications. U.S. Food and Drug Administration.
- World Health Organization. (2024). WHO classification of tumours of the breast (5th edition): Molecular pathology and clinical relevance. World Health Organization.
Frequently Asked Questions
How big is the breast cancer subtyping kits market in 2026?
The global breast cancer subtyping kits market is estimated to be valued at USD 4.8 billion in 2026.
What will be the size of breast cancer subtyping kits market in 2036?
The market size for the breast cancer subtyping kits market is projected to reach USD 12.0 billion by 2036.
How much will be the breast cancer subtyping kits market growth between 2026 and 2036?
The breast cancer subtyping kits market is expected to grow at a 9.6% CAGR between 2026 and 2036.
What are the key product types in the breast cancer subtyping kits market?
The key product types in breast cancer subtyping kits market are ihc-based kits, fish kits, pcr-based assays and others.
Which application segment to contribute significant share in the breast cancer subtyping kits market in 2026?
In terms of application, her2 subtyping segment to command 38.0% share in the breast cancer subtyping kits market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- IHC-based kits
- FISH kits
- PCR-based assays
- Others
- IHC-based kits
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- HER2 subtyping
- Hormone receptor status
- Triple-negative identification
- HER2 subtyping
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospitals
- Diagnostic laboratories
- Cancer research institutes
- Hospitals
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Application
- By End User
- Competition Analysis
- Competition Deep Dive
- Roche Diagnostics
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Agilent
- Abbott
- Bio-Rad
- Thermo Fisher Scientific
- Others
- Roche Diagnostics
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by End User
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Product Type , 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Product Type
- Figure 26: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Application
- Figure 29: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by End User
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026 to 2036
- Figure 35: Latin America Market Attractiveness Analysis by Product Type
- Figure 36: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 38: Latin America Market Attractiveness Analysis by Application
- Figure 39: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by End User
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026 to 2036
- Figure 45: Western Europe Market Attractiveness Analysis by Product Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 48: Western Europe Market Attractiveness Analysis by Application
- Figure 49: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 51: Western Europe Market Attractiveness Analysis by End User
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026 to 2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Application
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End User
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026 to 2036
- Figure 65: East Asia Market Attractiveness Analysis by Product Type
- Figure 66: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 68: East Asia Market Attractiveness Analysis by Application
- Figure 69: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 71: East Asia Market Attractiveness Analysis by End User
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026 to 2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026 to 2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

