Cold Chain Packaging Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
The Cold Chain Packaging Market is Segmented by Product Type (Insulated Shippers, Insulated Containers, Refrigerants, and Others), Application (Pharmaceuticals and Healthcare, Food and Beverage, Chemicals, and Others), Temperature Range (Frozen, Chilled, Ambient Controlled, and Cryogenic), Material Type (Expanded Polystyrene, Polyurethane, Vacuum Insulated Panels, and Others), and End Use Industry (Pharmaceuticals, Biotechnology, Clinical Trials, Food Processing, and Others). Forecast for 2026 to 2036.
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Cold Chain Packaging Market Size, Market Forecast and Outlook By FMI
The cold chain packaging market is expected to expand from USD 20.0 billion in 2026 to USD 39.1 billion by 2036. The market is forecast to register a 6.9% CAGR during the forecast period. Insulated shippers are likely to lead product type with a 52.5% share, while pharmaceuticals and healthcare are anticipated to account for 46.5% share of application demand in 2026.
Summary of the Cold Chain Packaging Market
- Demand and Growth Drivers
- Biologics and vaccine shipments are expected to support higher use of validated temperature-controlled packaging.
- Frozen food and meal-kit delivery are likely to increase the need for reliable thermal protection across longer routes.
- Digital temperature tracking is anticipated to improve compliance visibility for pharmaceutical and food logistics providers.
- Product and Segment View
- Insulated shippers are expected to lead the product type segment because they protect sensitive products during multi-day transport.
- Pharmaceuticals and healthcare are likely to remain the main application area due to strict storage requirements for biologics and vaccines.
- Vacuum insulated panels and phase change materials are anticipated to gain wider use as companies seek lighter and higher-performing systems.
- Geography and Competitive Outlook
- India is expected to lead country-level growth as pharmaceutical manufacturing and vaccine exports require stronger cold chain systems.
- China is likely to record strong expansion as biopharmaceutical output and regulated drug distribution increase.
- Suppliers with validated shipper fleets and digital monitoring systems are anticipated to gain preference in healthcare logistics.
- Analyst Opinion
- Ismail Sutaria, Principal consultant for Packaging at FMI, suggests, “The cold chain packaging market is moving toward validated systems that combine thermal reliability with lower waste and better shipment visibility. Suppliers with reusable assets and proven pharmaceutical compliance support are likely to gain advantage as biologics and specialty medicines expand across global routes.”
- Cold Chain Packaging Market Value Analysis
- The cold chain packaging market is shifting from basic insulated transport into a compliance-led logistics packaging category.
- Spending is likely to rise as pharmaceutical companies move more biologics and vaccines through global supply chains.
- Adoption is expected to improve as reusable shipper systems help reduce waste and long-term logistics cost.
- Value expansion is anticipated to be supported by IoT monitoring and advanced insulation materials that reduce temperature excursion risk.
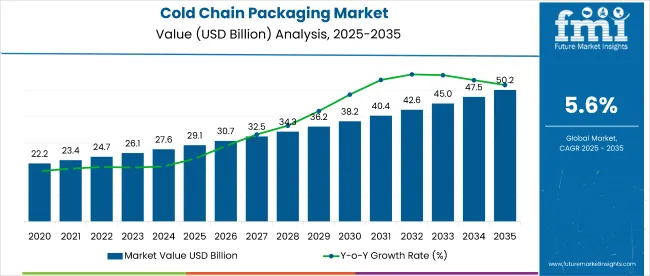
Cold Chain Packaging Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 20.0 billion |
| Market Forecast Value (2036) | USD 39.1 billion |
| Forecast CAGR (2026 to 2036) | 6.9% |
How Is the Cold Chain Packaging Market Segmented?
The Cold Chain Packaging Market is segmented by product type, application, temperature range, material type, end use industry, and region to support the safe movement of temperature sensitive products across global supply chains. By product type, the industry includes insulated shippers, insulated containers, refrigerants, and supporting packaging formats designed to preserve thermal integrity. By application, demand is generated from pharmaceuticals and healthcare, food and beverage, chemicals, and other regulated industries.
By temperature range, solutions are designed for frozen, chilled, ambient controlled, and cryogenic transport. By material type, packaging systems use expanded polystyrene, polyurethane, vacuum insulated panels, and hybrid materials. By end use industry, the industry serves pharmaceuticals, biotechnology, clinical trials, and food processing. Regionally, it spans North America, Europe, East Asia, South Asia Pacific, Latin America, and Middle East & Africa.
Why Do Insulated Shippers Dominate the Cold Chain Packaging Market?
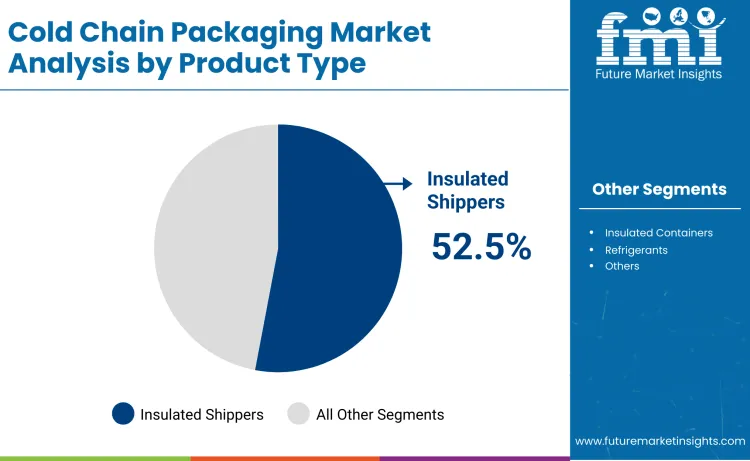
Insulated shippers hold about 52.5% of the Cold Chain Packaging Market because they provide the most flexible and reliable temperature control for pharmaceutical and biologic shipments. These systems combine high performance insulation with phase change materials that maintain precise temperature ranges over long transit durations. Insulated shippers are widely used for vaccines, cell therapies, and clinical trial materials because they support regulatory validated thermal performance.
Their compatibility with real time temperature monitoring sensors also enables pharmaceutical companies to meet strict compliance and audit requirements. Manufacturers continue to improve insulation efficiency and payload capacity, making these shippers lighter, more durable, and more cost effective. As global distribution of temperature sensitive therapies increases, insulated shippers remain the core packaging solution supporting safe and compliant cold chain logistics worldwide.
Why Do Pharmaceuticals and Healthcare Lead Cold Chain Packaging Demand?
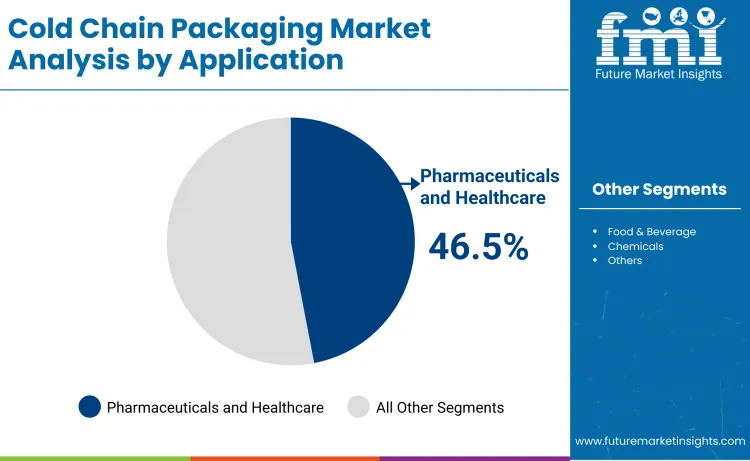
Pharmaceuticals and healthcare account for around 46.5% of cold chain packaging demand because modern medicines increasingly rely on strict temperature control to preserve safety and efficacy. Biologics, vaccines, and gene and cell therapies are highly sensitive to thermal fluctuations and require validated packaging to maintain stability throughout transportation and storage. Cold chain packaging protects these high value therapies from degradation while supporting compliance with global regulatory frameworks. Pharmaceutical companies also use temperature controlled packaging to distribute clinical trial materials across multiple countries. As personalized medicine and advanced biologics expand, the healthcare sector continues to drive the majority of demand for cold chain packaging, making it the most commercially important application segment in the global market.
What Are the Key Drivers, Trends, and Restraints in the Cold Chain Packaging Market?
The Cold Chain Packaging Market is driven by rapid growth in biologics, vaccines, and temperature sensitive pharmaceuticals that require reliable thermal protection during global distribution. Expansion of clinical trials, personalized medicine, and cell and gene therapies increases the volume of shipments needing validated packaging. Food safety regulations and the growth of international trade in perishable foods further support demand.
Major trends include the adoption of IoT enabled packaging with real time temperature tracking, the shift toward reusable insulated shippers to reduce waste and logistics costs, and the use of vacuum insulated panels and advanced thermal materials to improve performance and payload efficiency. However, the industry faces restraints from high initial investment costs, complex thermal validation requirements, and limited cold chain infrastructure in emerging markets. Compliance costs, specialized handling needs, and regulatory complexity also limit rapid adoption in some regions, particularly where logistics networks remain underdeveloped.
How Is Cold Chain Packaging Becoming Essential to Global Healthcare Logistics?
Cold chain packaging has moved from a support function to a strategic requirement in pharmaceutical and life science supply chains. From 2026 to 2036, the rapid rise of biologics, vaccines, injectable therapies, and personalized medicines is pushing demand for packaging that can maintain precise temperature conditions over long transport distances. Cold chain solutions now include insulated shippers, gel packs, phase change materials, and monitoring devices that ensure drug integrity.
Governments are also tightening rules on temperature control, making validated packaging mandatory for distribution. Emerging markets are investing heavily in refrigerated logistics and packaging infrastructure, while developed countries are upgrading to reusable and digitally tracked systems. These shifts are transforming cold chain packaging into a core enabler of global drug availability, with India, China, Brazil, the United States, Germany, Japan, and France shaping market momentum.
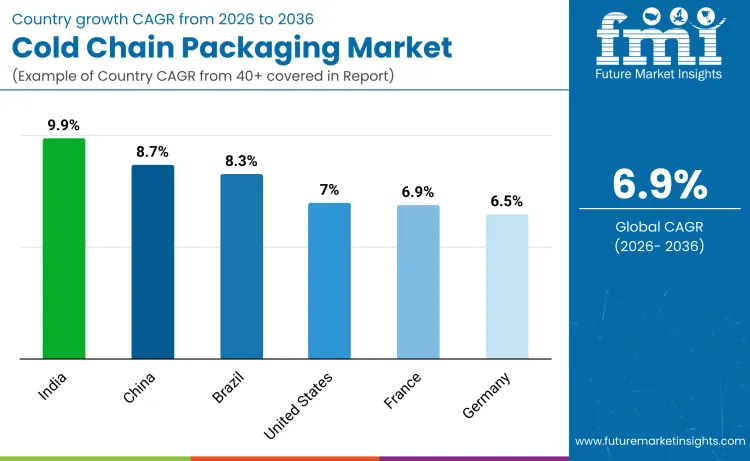
| Country | CAGR |
|---|---|
| India | 9.9% |
| China | 8.7% |
| Brazil | 8.3% |
| United States | 7.0% |
| France | 7.4% |
| Germany | 6.5% |
How Is India Becoming a Global Engine for Cold Chain Packaging Growth?
India’s cold chain packaging market is expected to expand at a CAGR of 9.9% from 2026 to 2036 as pharmaceutical manufacturing and vaccine exports continue to surge. India supplies a large share of the world’s vaccines and generic injectables, which must be transported under strict temperature control. Pharmaceutical hubs such as Hyderabad, Pune, and Visakhapatnam are investing in modern packaging, insulated containers, and temperature monitoring tools. Government programs supporting pharma parks and logistics corridors are improving cold chain reach. As Indian companies move further into biologics and specialty drugs, they require more advanced thermal packaging. This positions India as one of the most dynamic markets for cold chain packaging worldwide.
How Is China Driving Scale in Cold Chain Packaging Demand?
China’s cold chain packaging market is forecast to grow at a CAGR of 8.7% from 2026 to 2036, supported by rising biopharmaceutical production and healthcare consumption. Vaccine manufacturing, hospital drug distribution, and export shipments all depend on temperature controlled packaging. Cities such as Shanghai, Beijing, and Shenzhen are building advanced cold chain networks that require high quality insulated boxes and reusable containers. Regulatory focus on drug safety is also pushing companies to adopt validated packaging solutions. As China continues to expand its pharmaceutical footprint globally, cold chain packaging becomes a key investment area to protect product quality and ensure regulatory compliance.
How Is the United States Advancing High Value Cold Chain Packaging?
The United States cold chain packaging market is projected to grow at a CAGR of 7.0% from 2026 to 2036, driven by strong demand for biologics, cell therapies, and specialty drugs. These products often require ultra low temperature control, making advanced packaging critical. Pharmaceutical firms and logistics providers are investing in reusable shippers, smart sensors, and data logging systems to improve shipment reliability. Major hubs in California, New Jersey, and North Carolina handle large volumes of temperature sensitive drugs. Growth in clinical trials and direct-to-patient delivery models also increases packaging needs. This makes the United States a leading market for premium cold chain packaging solutions.
How Is Brazil Expanding Cold Chain Packaging for Public Health?
Brazil’s cold chain packaging market is expected to grow at a CAGR of 8.3% from 2026 to 2036 as vaccine distribution and pharmaceutical imports rise. The country’s large population and widespread healthcare system depend on reliable temperature controlled transport. Public immunization campaigns and expanding hospital networks drive steady demand for insulated packaging. Brazilian logistics providers are upgrading storage and transport systems to reduce product loss and improve reach into rural areas. As domestic biopharmaceutical production increases, the need for validated cold chain packaging becomes even stronger, supporting long term market expansion.
How Is Germany Maintaining Its Leadership in Quality Cold Chain Packaging?
Germany’s cold chain packaging market is forecast to grow at a CAGR of 6.5% from 2026 to 2036, supported by its strong pharmaceutical and biotechnology industries. German companies specialize in high performance packaging systems used for biologics, clinical trials, and global drug shipments. Advanced insulation, strict validation, and reusable container systems are widely used. Export oriented pharmaceutical manufacturers rely on these solutions to meet international shipping standards. With Europe’s growing demand for advanced medicines, Germany continues to play a central role in supplying reliable cold chain packaging.
How Is France Strengthening Its Role in Cold Chain Packaging?
France’s cold chain packaging market is projected to grow at a CAGR of 7.4% from 2026 to 2036, driven by its strong biopharmaceutical research base and pharmaceutical distribution network. Research hospitals, biotech firms, and vaccine producers depend on precise temperature control for sensitive products. France also serves as a key logistics hub for European drug distribution, increasing the need for validated packaging. As clinical trials and biologic exports increase, cold chain packaging remains a critical investment area for French healthcare and life science companies.
What Is the Competitive Landscape of the Cold Chain Packaging Market?
The cold chain packaging market operates as a mission critical segment of pharmaceutical and life science logistics where performance, compliance, and reliability determine supplier selection. A relatively small group of global providers captures most industry revenue because pharmaceutical companies prefer long term partners that can guarantee temperature control, regulatory documentation, and consistent delivery across international shipping lanes. Purchasing decisions in this market are driven by risk mitigation rather than packaging cost.
Market leaders such as Sonoco Products Company, Pelican BioThermal, and Cold Chain Technologies compete by offering large fleets of qualified shippers that support multi day temperature stability across ambient, refrigerated, and frozen ranges. These firms provide full service models that include packaging selection, route validation, asset management, and technical support, making them deeply embedded in the distribution networks of vaccine makers, biologics producers, and specialty logistics providers.
Advanced technology suppliers including va-Q-tec AG, Softbox Systems, and CSafe Global focus on high performance insulation systems, phase change materials, and active temperature controlled containers. Their solutions are widely used in long distance and intercontinental shipments where temperature excursions carry high financial and regulatory risk. Integration with tracking and data logging platforms further strengthens their value to pharmaceutical supply chains.
Companies such as Cryopak, Intelsius, and Envirotainer play an important role in clinical trials, last mile delivery, and regional distribution. Their competitive edge comes from flexible packaging configurations, fast response times, and the ability to support smaller, time sensitive shipments for emerging therapies and decentralized clinical research.
As the industry expands with the growth of cell therapies, gene therapies, and temperature sensitive vaccines, competitive advantage increasingly belongs to suppliers that can combine validated thermal protection, global service reach, and digital visibility. Firms that deliver predictable performance across every shipment stage will continue to strengthen their position in the evolving cold chain packaging market.
Key Players in the Cold Chain Packaging Market
- Sonoco Products Company
- Pelican BioThermal
- Cold Chain Technologies
- va-Q-tec AG
- Softbox Systems
- Cryopak
- CSafe Global
- Intelsius
- Envirotainer
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Market Size Reference | Market size in value terms |
| Market Parameter | Revenue in USD billion |
| Product Type | Insulated shippers, insulated containers, refrigerants, others |
| Application | Pharmaceuticals and healthcare, food and beverage, chemicals, others |
| Temperature Range | Frozen minus 20 C to minus 80 C, chilled 2 C to 8 C, ambient controlled 15 C to 25 C, cryogenic below minus 80 C |
| Material Type | Expanded polystyrene EPS, polyurethane PU, vacuum insulated panels VIP, others |
| End Use Industry | Pharmaceuticals, biotechnology, clinical trials, food processing, others |
| Regions Covered | North America, Europe, East Asia, South Asia and Pacific, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, United Kingdom, Germany, France, China, Japan, South Korea, India, Brazil, Australia and 40+ countries |
| Key Companies Profiled | Sonoco Products Company, Pelican BioThermal, Cold Chain Technologies, va Q tec AG, Softbox Systems, Cryopak, CSafe Global, Intelsius, Envirotainer |
| Additional Attributes | Dollar sales by product type and application category, regional demand trends, competitive landscape, technological advancements in thermal insulation systems, phase change material development, IoT monitoring innovation, and pharmaceutical distribution integration |
Cold Chain Packaging Market by Segments
By Product Type:
- Insulated Shippers
- Insulated Containers
- Refrigerants
- Others
By Application:
- Pharmaceuticals & Healthcare
- Food & Beverage
- Chemicals
- Others
By Temperature Range:
- Frozen (-20°C to -80°C)
- Chilled (2°C to 8°C)
- Ambient Controlled (15°C to 25°C)
- Cryogenic (Below -80°C)
By Material Type:
- Expanded Polystyrene (EPS)
- Polyurethane (PU)
- Vacuum Insulated Panels (VIP)
- Others
By End-Use Industry:
- Pharmaceuticals
- Biotechnology
- Clinical Trials
- Food Processing
- Others
By Region:
- North America
- United States
- Canada
- Mexico
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Switzerland
- Nordic
- BENELUX
- Rest of Europe
- East Asia
- China
- Japan
- South Korea
- Taiwan
- South Asia & Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia & Pacific
- Latin America
- Brazil
- Chile
- Argentina
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- World Health Organization. (2024). Temperature controlled supply chains for vaccines, biologics, and essential medicines. World Health Organization.
- United States Food and Drug Administration. (2023). Guidance for industry on good distribution practices for temperature sensitive pharmaceutical products. United States Food and Drug Administration.
- International Air Transport Association. (2024). Temperature control regulations and packaging standards for air cargo. International Air Transport Association.
- International Organization for Standardization. (2023). Cold chain packaging and temperature controlled transport standards for pharmaceuticals and food products. International Organization for Standardization.
- European Medicines Agency. (2024). Good distribution practice guidelines for medicinal products requiring cold chain logistics. European Medicines Agency.
Frequently Asked Questions
How big is the cold chain packaging market in 2026?
The global cold chain packaging market is valued at USD 20.0 billion in 2026.
What will be the size of the cold chain packaging market in 2036?
The cold chain packaging market is projected to reach USD 39.1 billion by 2036.
How much will the cold chain packaging market grow between 2026 and 2036?
The cold chain packaging market is expected to grow at a CAGR of 6.9% between 2026 and 2036.
What are the key product types in the cold chain packaging market?
The main product types include insulated shippers, insulated containers, refrigerants, and other temperature controlled packaging solutions.
Which application segment contributes the largest share in the cold chain packaging market in 2026?
Pharmaceuticals and healthcare lead with about 46.5% share in 2026, driven by strong demand for vaccine, biologics, and clinical trial shipments.
Table of Content
- Executive Summary
- Global Cold Chain Packaging Market Outlook
- Demand Side Trends
- Supply Side Trends
- Technology and Digital Logistics Roadmap
- Strategic Analysis and Recommendations
- Market Overview
- Market Coverage and Taxonomy
- Market Definition, Scope, and Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunities
- Trends
- Scenario Forecast Analysis
- Optimistic Scenario
- Likely Scenario
- Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter Five Forces Analysis
- Regulatory and Cold Chain Standards Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Cold Chain Packaging Market Analysis 2021 to 2025 and Forecast 2026 to 2036
- Historical Market Size Value Analysis 2021 to 2025
- Current and Future Market Size Projections 2026 to 2036
- Year to Year Growth Trend Analysis
- Absolute Dollar Opportunity Analysis
- Global Cold Chain Packaging Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Cold Chain Packaging Market Analysis by Product Type
- Insulated Shippers
- Insulated Containers
- Refrigerants
- Others
- Global Cold Chain Packaging Market Analysis by Application
- Pharmaceuticals & Healthcare
- Food & Beverage
- Chemicals
- Others
- Global Cold Chain Packaging Market Analysis by Temperature Range
- Frozen (−20°C to −80°C)
- Chilled (2°C to 8°C)
- Ambient Controlled (15°C to 25°C)
- Cryogenic (Below −80°C)
- Global Cold Chain Packaging Market Analysis by Material Type
- Expanded Polystyrene (EPS)
- Polyurethane (PU)
- Vacuum Insulated Panels (VIP)
- Others
- Global Cold Chain Packaging Market Analysis by End-Use Industry
- Pharmaceuticals
- Biotechnology
- Clinical Trials
- Food Processing
- Others
- Global Cold Chain Packaging Market Analysis by Region and Country
- Market Structure Analysis
- Competition Dashboard
- Market Share Analysis
- Company Benchmarking
- Competitive Landscape
- Sonoco Products Company
- Pelican BioThermal
- Cold Chain Technologies
- va-Q-tec AG
- Softbox Systems
- Cryopak
- CSafe Global
- Intelsius
- Envirotainer
- Assumptions and Acronyms
- Research Methodology
List of Tables
- Table 1 Global Cold Chain Packaging Market Value USD Billion and Volume Forecast by Region 2021 to 2036
- Table 2 Global Cold Chain Packaging Market Value and Volume by Product Type 2021 to 2036
- Table 3 Global Cold Chain Packaging Market Value and Volume by Application 2021 to 2036
- Table 4 Global Cold Chain Packaging Market Value and Volume by Temperature Range 2021 to 2036
- Table 5 Global Cold Chain Packaging Market Value and Volume by Material Type 2021 to 2036
- Table 6 Global Cold Chain Packaging Market Value and Volume by End-Use Industry 2021 to 2036
- Table 7 North America Market Value and Volume by Country 2021 to 2036
- Table 8 North America Market Value and Volume by Product Type 2021 to 2036
- Table 9 North America Market Value and Volume by Application 2021 to 2036
- Table 10 North America Market Value and Volume by Temperature Range 2021 to 2036
- Table 11 Europe Market Value and Volume by Country 2021 to 2036
- Table 12 Europe Market Value and Volume by Product Type 2021 to 2036
- Table 13 Europe Market Value and Volume by Application 2021 to 2036
- Table 14 Europe Market Value and Volume by Temperature Range 2021 to 2036
- Table 15 East Asia Market Value and Volume by Country 2021 to 2036
- Table 16 East Asia Market Value and Volume by Product Type 2021 to 2036
- Table 17 East Asia Market Value and Volume by Application 2021 to 2036
- Table 18 East Asia Market Value and Volume by Temperature Range 2021 to 2036
- Table 19 South Asia & Pacific Market Value and Volume by Country 2021 to 2036
- Table 20 Latin America Market Value and Volume by Country 2021 to 2036
- Table 21 Middle East & Africa Market Value and Volume by Country 2021 to 2036
List of Figures
- Figure 1 Global Market Share Growth Rate and Value by Product Type 2021 to 2036
- Figure 2 Global Market Share Growth Rate and Value by Application 2021 to 2036
- Figure 3 Global Market Share Growth Rate and Value by Temperature Range 2021 to 2036
- Figure 4 Global Market Share Growth Rate and Value by Material Type 2021 to 2036
- Figure 5 Global Market Share Growth Rate and Value by End-Use Industry 2021 to 2036
- Figure 6 North America Market Share and Growth by Product Type 2021 to 2036
- Figure 7 North America Market Share and Growth by Application 2021 to 2036
- Figure 8 North America Market Share and Growth by Temperature Range 2021 to 2036
- Figure 9 Europe Market Share and Growth by Product Type 2021 to 2036
- Figure 10 Europe Market Share and Growth by Application 2021 to 2036
- Figure 11 Europe Market Share and Growth by Temperature Range 2021 to 2036
- Figure 12 East Asia Market Share and Growth by Product Type 2021 to 2036
- Figure 13 East Asia Market Share and Growth by Application 2021 to 2036
- Figure 14 East Asia Market Share and Growth by Temperature Range 2021 to 2036

