Fill Finish Manufacturing Market
The Fill Finish Manufacturing Market is segmented by Product Type (Prefilled Syringes, Vials, Cartridges, Ampoules, and Bottles), Application (Vaccines, Biologics, Small Molecules, and Cell & Gene Therapies), End Use (Pharmaceutical Companies, Contract Manufacturing Organizations, Biotechnology Companies, and Others) and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2015 to 2023 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2026 to 2035
Fill Finish Manufacturing Market Size, Market Forecast and Outlook By FMI
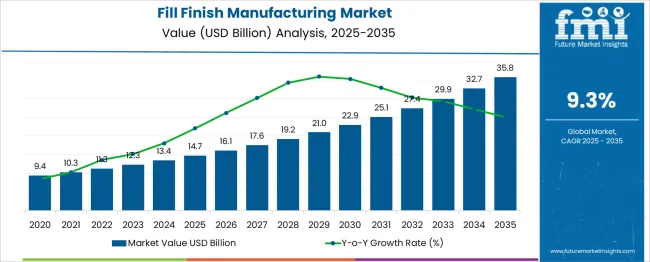
In 2025, the fill finish manufacturing market surpassed USD 14.7 billion. Based on Future Market Insights' analysis, demand for fill finish manufacturing services is estimated to grow to USD 16.2 billion in 2026 and USD 38.9 billion by 2036. FMI projects a CAGR of 9.1% during the forecast period.
Absolute dollar growth of USD 22.7 billion over the decade signals transformational opportunity rather than incremental expansion. Despite manufacturing complexity and stringent sterile processing requirements, major fill finish manufacturers maintain robust growth trajectories through facility automation and production capacity optimization.
Summary of Fill Finish Manufacturing Market
- Fill Finish Manufacturing Market Definition
- The industry covers sterile drug product manufacturing services including filling, sealing, inspection, and packaging operations for pharmaceutical and biotechnology applications.
- Demand Drivers in the Market
- Biologics market expansion drives specialized aseptic processing demand.
- Regulatory compliance requirements accelerate facility modernization and automation adoption.
- Contract manufacturing consolidation enables production scale optimization and cost reduction.
- Key Segments Analyzed in the FMI Report
- Product Type: Prefilled syringes (42%) versus traditional vials and cartridges.
- Application: Biologics and vaccines (55-60% of total value) versus small molecule drugs.
- Geography: High-capacity North America versus cost-competitive Asia-Pacific manufacturing.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Consultant for Healthcare at Future Market Insights, opines, "In the updated version of the Fill Finish Manufacturing Market Report for 2026 to 2036, executives will find insights into automation technology adoption that drives production efficiency gains. My findings point at sterile processing capabilities directly impacting pharmaceutical supply chain reliability and cold chain distribution requirements could influence regional manufacturing strategies."
- Strategic Implications/Executive Takeaways
- Shift focus from manual operations to automated production platforms.
- Treat manufacturing capacity as competitive advantage rather than operational function.
- Prioritize sterile processing expertise to capture biologics manufacturing opportunities.
- Methodology
- Validated through manufacturing capacity data and commercial production benchmarks.
- Zero reliance on speculative technology adoption timelines.
- Based on verifiable facility inspections and proven manufacturing capabilities.
In February 2026, West Pharma released its full-year 2025 results. "Our strong finish to 2025 was a result of the team's relentless execution of our growth strategy," said Eric M. Green, West's President, Chief Executive Officer and Chair of the Board. "Our performance in the quarter was led by our High-Value Product Components business in our Proprietary Products Segment, enabling us to deliver double-digit adjusted earnings per share growth. We expect positive momentum to continue in 2026, which will be a key year of execution and operational excellence to drive sustainable growth and create value for our patients, customers and shareholders." [1]
United States (9.8% CAGR) and Germany (9.3% CAGR) drive market expansion through advanced manufacturing infrastructure and regulatory framework maturation. Japan (8.9% CAGR) contributes significantly via biologics manufacturing capacity and automated production line integration. Emerging fill finish markets such as China (8.7%), South Korea (8.2%), and India (7.8%) generate growth through contract manufacturing expansion and pharmaceutical production capacity development, while volume acceleration occurs through biosimilar manufacturing and generic drug production.
Fill Finish Manufacturing Market Definition
Fill finish manufacturing serves as the final pharmaceutical production stage where sterile drug products are filled into primary containers, sealed, and prepared for distribution through aseptic processing techniques and quality control validation. The fill finish manufacturing market comprises global contract manufacturing services, equipment supply, and technology platforms for sterile drug product finishing across pharmaceutical applications.
Fill Finish Manufacturing Market Inclusions
The report includes a comprehensive analysis of market dynamics, featuring Global and Regional Market Sizes (Volume and Value) and a 10-year Forecast (2026-2036). It covers segmental breakdowns by product type (Prefilled Syringes, Vials, Cartridges), applications (Vaccines, Biologics, Small Molecules), and end-users (Pharmaceutical Companies, Contract Organizations, Biotech Companies).
Fill Finish Manufacturing Market Exclusions
The scope excludes upstream drug substance manufacturing or downstream pharmaceutical packaging operations unless integrated into aseptic processing workflows. It also omits laboratory-scale filling equipment or primary container manufacturing, focusing strictly on commercial-scale sterile processing and immediate quality control systems. Report does not account for investigational drug manufacturing or pilot-scale operations with limited commercial potential.
Fill Finish Manufacturing Market Research Methodology
- Primary Research: Interviews were conducted with contract manufacturing executives, pharmaceutical production managers, regulatory compliance specialists, facility engineering teams, and procurement departments across major regions. Manufacturing technology vendors clarified equipment capabilities and production capacity dynamics.
- Desk Research: FDA inspection databases, EMA manufacturing guidelines, ICH quality standards, and pharmaceutical industry associations supported manufacturing benchmarking and regulatory requirement analysis.
- Market-Sizing and Forecasting: A hybrid top-down and bottom-up model was developed. Demand was reconstructed from pharmaceutical production volumes, manufacturing facility capacity, and contract service pricing, then validated against equipment installation and technology adoption rates.
- Data Validation and Update Cycle: Outputs undergo manufacturing capacity verification, variance checks across production and financial datasets, and structured expert review prior to release.
Segmental Analysis
Fill Finish Manufacturing Market Analysis by Product Type
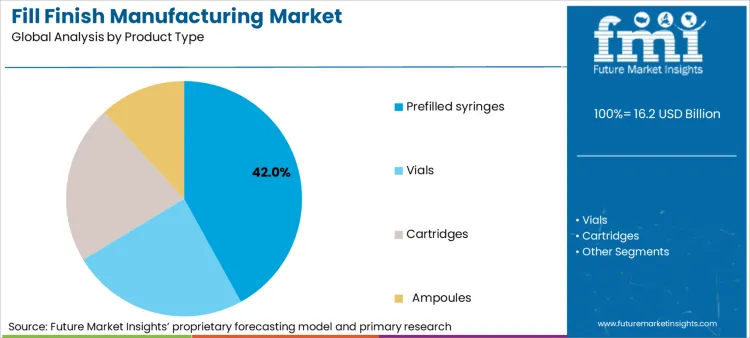
Based on FMI's fill finish manufacturing market report, demand for prefilled syringes is estimated to hold 42% share in 2026. Growth in prefilled syringe manufacturing comes from pharmaceutical companies leveraging patient convenience benefits and dosing accuracy advantages through automated assembly line production and integrated quality systems. The advancements in drug delivery technology has furthered strengthened market position.
- Manufacturing Scale: Companies are expanding production capabilities through automated filling systems and robotics integration to achieve higher throughput rates while maintaining sterile processing standards across multiple pharmaceutical applications.
- Quality Standardization: Companies are leveraging automated inspection systems to integrate real-time quality control across production batches, enabling regulatory submissions that meet FDA sterile processing requirements while maintaining product consistency across multiple therapeutic areas.
- Technology Integration: The industry shift toward fully automated production lines enables pharmaceutical manufacturers to execute high-volume sterile processing, decoupling manual operations from production capacity to optimize manufacturing economics typically constrained by labor availability and training requirements.
Fill Finish Manufacturing Market Analysis by Application
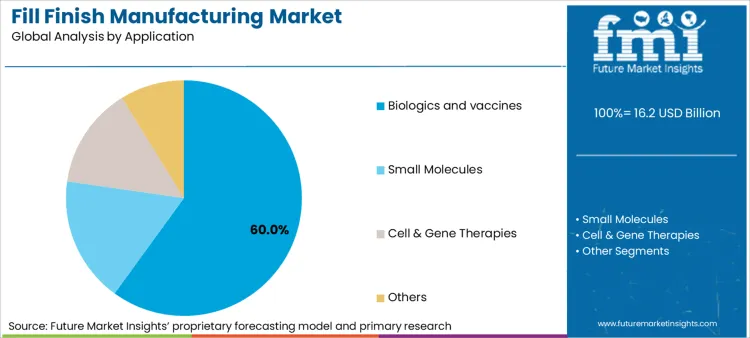
Biologics manufacturing applications hold a 32.8% market share in 2026, driven by their role as the primary growth driver for specialized aseptic processing and temperature-controlled production protocols. This concentration creates market opportunities that expand during pharmaceutical innovation cycles. The segment's integration with advanced therapy development and protein manufacturing transforms production facilities into comprehensive manufacturing platforms, while regulatory validation pathways offer structured development timelines for capacity expansion and technology upgrades.
- Technology Integration: Pharmaceutical companies are investing in specialized biologics manufacturing infrastructure that incorporates cold chain management systems and advanced monitoring technologies to maintain product stability throughout the filling process.
- Production Protocol Development: Manufacturing organizations are developing standardized protocols for complex biologics processing that integrate upstream production with downstream filling operations, enabling seamless technology transfer and scale-up capabilities across multiple facility locations.
Fill Finish Manufacturing Market Drivers, Restraints, and Opportunities
Future Market Insights analysis indicates historical patterns point to an emerging, high-value global industry where fill finish manufacturing services remain the critical bottleneck for pharmaceutical product commercialization and supply chain optimization. Estimated valuation in 2026 represents the immediate "capacity expansion phase" as manufacturers invest in automated production facilities and sterile processing technology to meet pharmaceutical demand growth.
While demand for complex biologics manufacturing faces technical challenges (production complexity), the specialized nature of sterile processing commands premium service pricing (revenue acceleration), resulting in robust percentage growth rates. The forecast accounts for the market reaching operational maturity where manufacturing standards are established and production scales support global pharmaceutical supply chains.
- Technology Differentiation: Growth shifts from manual operations toward automated production systems, using real-time monitoring and validated sterile processing to maintain competitive advantages against traditional manufacturing approaches.
- Regulatory Pathway Maturation: Standards like FDA 21 CFR Part 211 and ICH Q7 guidelines establish baseline requirements rather than experimental protocols, forcing transition from conventional filling to advanced aseptic manufacturing.
- Manufacturing Capacity Requirements: To maintain cost competitiveness with integrated pharmaceutical production, contract manufacturers aggressively optimize facility utilization to achieve per-unit economics that support pharmaceutical industry adoption and supply chain integration.
Regional Analysis
Based on regional analysis, the fill finish manufacturing market is segmented into North America, Latin America, Europe, East Asia, South Asia, Oceania and Middle East & Africa across 40+ countries. The full report also offers market attractiveness analysis based on regional trends.
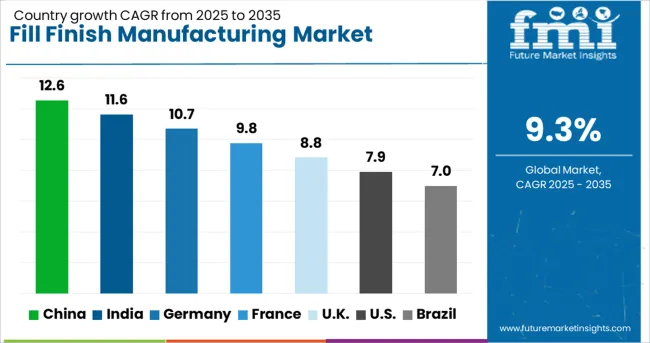
| Country | CAGR (2026 to 2036) |
|---|---|
| United States | 9.8% |
| Germany | 9.3% |
| Japan | 8.9% |
| China | 8.7% |
| South Korea | 8.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Fill Finish Manufacturing Market Analysis
North America is characterized by "manufacturing leadership," where established FDA validation pathways combine with advanced production infrastructure to drive commercial adoption of automated filling systems and sterile processing technology. Key players in North America include Catalent, which leads in contract manufacturing services, West Pharmaceutical Services and Gerresheimer specialize in prefilled syringe systems and container manufacturing.
- United States: Demand for fill finish manufacturing services in the United States is projected to rise at 9.8% CAGR through 2036. Growth is driven by pharmaceutical production capacity expansion and biologics manufacturing investment under advanced therapy development programs.
FMI's report includes detailed analysis of growth in the North American region, along with country-wise assessment that includes the USA and Canada. Readers can find manufacturing trends, regulatory frameworks, and market development based on different product segments and pharmaceutical applications.
Asia Pacific Fill Finish Manufacturing Market Analysis
Asia Pacific represents the global contract manufacturing hub, leveraging cost-competitive production facilities and specialized technical capabilities to achieve manufacturing costs that enable pharmaceutical companies to optimize production economics compared to domestic facilities. Lonza and Samsung Biologics lead in large-scale manufacturing services, WuXi Biologics and AGC Biologics drive regional capacity expansion.
- China: Leading with an 8.7% CAGR, the market benefits from National Medical Products Administration manufacturing standards and government investment in pharmaceutical production infrastructure and biotechnology industrial parks.
- Japan: Domestic demand accelerates at 8.9% CAGR, supported by PMDA manufacturing guidelines and pharmaceutical industry collaboration with international contract manufacturers for supply chain optimization.
The full report analyzes the fill finish manufacturing market across East and South Asia from 2021-2036, covering production capacity, technology adoption, and regulatory frameworks in China, Japan, South Korea, India, Singapore, and Australia. The assessment highlights manufacturing segments that drive regional production growth.
Europe Fill Finish Manufacturing Market Analysis
Europe functions as the "regulatory standardization center," where market development is driven by EMA manufacturing guidelines and coordinated inspection networks across member states. Leading players include Recipharm, Vetter Pharma, and Boehringer Ingelheim driving contract manufacturing and specialized sterile processing development.
- Germany: Demand for fill finish manufacturing services in Germany rises at 9.3% CAGR through 2036, driven by pharmaceutical industry investment and manufacturing infrastructure that supports both domestic production and international pharmaceutical partnerships.
FMI's analysis of the European fill finish manufacturing market includes country-specific assessment covering Germany, France, Italy, UK, Spain, Netherlands, and Scandinavia. Readers can access detailed regulatory framework analysis and manufacturing development trends across the European region.
Competitive Landscape for Market Players
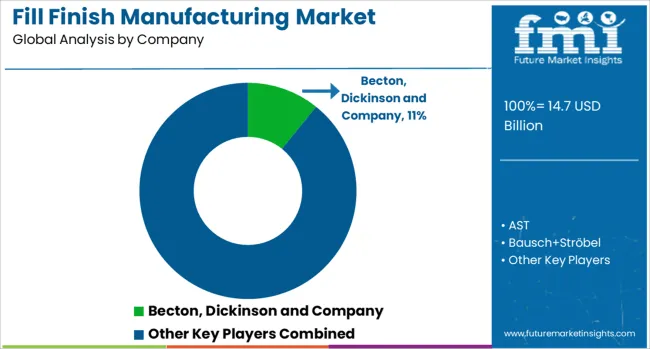
Market structure remains concentrated among established contract manufacturers with validated production capabilities and regulatory compliance records. Primary competitive variables include manufacturing capacity demonstration, production cost optimization, and facility validation timing rather than technology platform breadth.
Companies with integrated production facilities and established quality systems demonstrate superior ability to scale operations while maintaining sterile processing consistency. Strategic manufacturing partnerships provide market access for pharmaceutical companies while larger contract organizations leverage facility expansion strategies to access specialized production capabilities.
Regulatory validation status creates distinct competitive tiers. Companies with FDA-approved facilities command premium pricing and customer access, while pre-validation manufacturers compete primarily on capacity availability and technical capabilities. Samsung Biologics’ CEO and President John Rim said, "Despite industry challenges, Samsung Biologics is well positioned to kick-start a new phase of growth in 2025 with the opening of Bio Campus II and launch of antibody-drug conjugate (ADC) services." He added, "We’ll continue to invest in new modalities and technologies to better address client needs, maximizing satisfaction and ensuring the delivery of highest-quality products." [2]
Recent Developments
The report includes comprehensive coverage of key industry developments and competitive positioning:
- In December 2025, expanded USA manufacturing capabilities with strategic acquisition of human genome sciences from GSK. [3]
- In December 2025, Recipharm Advanced Bio, a segment of Recipharm and a leading Contract Development and Manufacturing Organisation (CDMO), and Infant Bacterial Therapeutics AB (IBT), announced the initiation of Process Performance Qualification (PPQ) for IBT's lead product, IBP-9414. The PPQ programme will be conducted at Recipharm's microbial manufacturing facility in Cuxhaven, Germany. [4]
- In October 2025, Lonza, one of the world’s largest contract development and manufacturing organizations (CDMOs), announced that it has signed an agreement to acquire Redberry SAS (Redberry), an innovative company specializing in rapid microbiology testing solutions using solid-phase cytometry (SPC) technology. [5]
- In February 2024, Samsung Biologics inked a deal with LegoChem Biosciences for ADC development and manufacturing. [6]
Key Players in Fill Finish Manufacturing Market
- Catalent Inc.
- West Pharmaceutical Services
- Gerresheimer AG
- Lonza Group AG
- Samsung Biologics
- WuXi Biologics
- Recipharm AB
- Vetter Pharma
- Boehringer Ingelheim
- AGC Biologics
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 16.2 billion (2026) to USD 38.9 billion (2036), at a CAGR of 9.1% |
| Market Definition | The fill finish manufacturing market comprises global contract manufacturing services, equipment supply, and technology platforms for sterile drug product finishing across pharmaceutical applications including filling, sealing, inspection, and packaging operations. |
| Product Type Segmentation | Prefilled Syringes, Vials, Cartridges, Ampoules, Bottles |
| Application Coverage | Vaccines, Biologics, Small Molecules, Cell & Gene Therapies |
| End Use Segmentation | Pharmaceutical Companies, Contract Manufacturing Organizations, Biotechnology Companies, Others |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Japan, China, South Korea, India, Australia and 40 plus countries |
| Key Companies Profiled | Catalent Inc., West Pharmaceutical Services, Gerresheimer AG, Lonza Group AG, Samsung Biologics, WuXi Biologics, Recipharm AB, Vetter Pharma, Boehringer Ingelheim, AGC Biologics |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top down and bottom up market modeling validated through manufacturing capacity databases and regulatory inspection tracking, supported by production facility verification and pharmaceutical industry benchmarking |
Fill Finish Manufacturing Market Analysis by Segments
Product Type:
- Prefilled Syringes
- Vials
- Cartridges
- Ampoules
- Bottles
Application:
- Vaccines
- Biologics
- Small Molecules
- Cell & Gene Therapies
- Others (veterinary, diagnostic applications)
End Use:
- Pharmaceutical Companies
- Contract Manufacturing Organizations
- Biotechnology Companies
- Others
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia
- Singapore
- Thailand
- Rest of Asia Pacific
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Netherlands
- Scandinavia
- Switzerland
- Rest of Europe
- North America
- United States
- Canada
- Latin America
- Brazil
- Mexico
- Argentina
- Rest of Latin America
- Middle East & Africa
- Saudi Arabia
- United Arab Emirates
- South Africa
- Israel
- Rest of Middle East & Africa
Bibliography
- [1] West Pharmaceutical Services, Inc. (2026, February 12). West reports fourth-quarter and full-year 2025 results [Press release].
- [2] Samsung Biologics Co., Ltd. (2025, January 6). Samsung Biologics to present business updates at 2025 [Press release].
- [3] Samsung Biologics Co., Ltd. (2025, December 22). Samsung Biologics expands USA manufacturing capabilities with strategic acquisition from GSK. Samsung Biologics Co., Ltd.
- [4] Infant Bacterial Therapeutics AB & Recipharm Advanced Bio. (2025, December 1). Infant Bacterial Therapeutics AB secures partnership with Recipharm Advanced Bio for commercial production of drug substance. Recipharm Advanced Bio.
- [5] Lonza Group AG. (2025, October 27). Lonza press advisory [Media advisory]. Lonza.
- [6] Samsung Biologics Co., Ltd. (2025, January 9). Samsung Biologics extends collaboration with LigaChem Biosciences for ADC development and manufacturing. Samsung Biologics Co., Ltd.
This Report Addresses
- Market intelligence to enable strategic decision making across established and emerging fill finish manufacturing markets
- Market size estimation and 10-year revenue forecasts from 2026 to 2036, supported by validated manufacturing capacity data and regulatory compliance benchmarks
- Growth opportunity mapping across product types and pharmaceutical applications with emphasis on production optimization
- Segment and regional revenue forecasts covering contract manufacturing, equipment supply, pharmaceutical production, and biotechnology applications
- Competition strategy assessment including manufacturing scale requirements, facility validation timelines, and regulatory compliance capabilities
- Manufacturing development tracking including FDA inspections, EMA guidelines, and international quality harmonization initiatives
- Market access analysis covering contract pricing frameworks, pharmaceutical industry adoption, and supply chain integration programs
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats for executive and operational use
Frequently Asked Questions
How large is the demand for Fill Finish Manufacturing in the global market in 2026?
Demand for Fill Finish Manufacturing in the global market is estimated to be valued at USD 16.2 billion in 2026.
What will be the market size of Fill Finish Manufacturing in the global market by 2036?
Market size for Fill Finish Manufacturing is projected to reach USD 38.9 billion by 2036.
What is the expected demand growth for Fill Finish Manufacturing in the global market between 2026 and 2036?
Demand for Fill Finish Manufacturing in the global market is expected to grow at a CAGR of 9.1% between 2026 and 2036.
Which Product Type is poised to lead global sales by 2026?
Prefilled Syringes are expected to be the dominant product type, capturing approximately 42.0% of global market share in 2026 due to patient convenience benefits and automated manufacturing capabilities.
How significant is the role of Pharmaceutical Companies in driving Fill Finish Manufacturing adoption in 2026?
Pharmaceutical companies represent the primary end-user segment, projected to hold approximately 58% share of the total market in 2026 as biologics production accelerates through capacity expansion.
What is Driving Fill Finish Manufacturing Demand in the United States?
Pharmaceutical production capacity expansion and biologics manufacturing investment under advanced therapy development programs are driving growth.
What Regulatory Standards are Referenced for the United States?
FDA 21 CFR Part 211 and current Good Manufacturing Practice (cGMP) guidelines are referenced as key regulatory frameworks.
What is the United States Growth Outlook in this Report?
The United States is projected to grow at a CAGR of 9.8% during 2026 to 2036.
Why is Europe described as Important for Regulatory Development in this Report?
Growth is driven by EMA manufacturing guidelines and coordinated inspection networks that establish international quality standards.
What Type of Development Dominates in Europe?
Contract manufacturing services and specialized sterile processing development for pharmaceutical applications dominate regional activity.
What is Germany Growth Outlook in this Report?
Germany is projected to expand at a CAGR of 9.3% during 2026 to 2036.
Does the Report Cover Japan in its Regional Analysis?
Japan is included within East Asia under the regional scope of analysis.
What are the Sources referred to for analyzing the Market in Japan?
PMDA manufacturing guidelines, pharmaceutical industry databases, and biotechnology manufacturing associations covering production capacity, facility inspections, and technology adoption are cited as primary reference sources.
What is the Main Development Theme Linked to Japan in Asia Coverage?
Asia Pacific development is associated with contract manufacturing expansion to enable cost-competitive pharmaceutical production.
Does the Report Cover South Korea in its Regional Analysis?
South Korea is included within East Asia under the regional coverage framework.
What is the Main Korea Related Development Theme in Asia Coverage?
Biotechnology manufacturing infrastructure investment and pharmaceutical production capacity building is emphasized.
Which Manufacturing Approaches are Strategically Important for Asia Pacific Supply Chains?
Cost-competitive production facilities and specialized technical capabilities are prioritized for pharmaceutical supply chain optimization across regional markets.
What is Fill Finish Manufacturing and What is It Mainly Used For?
Fill finish manufacturing involves sterile drug product processing primarily used for pharmaceutical applications including filling, sealing, inspection, and packaging of vaccines, biologics, and small molecule drugs.
What does Fill Finish Manufacturing Market Mean in this Report?
Fill finish manufacturing market refers to global contract manufacturing services, equipment supply, and technology platforms for sterile pharmaceutical production across various applications.
What is Included in the Scope of this Fill Finish Manufacturing Market Report?
Scope covers sterile processing services by product type, key applications such as vaccines and biologics manufacturing, and end-use facilities including pharmaceutical companies and contract manufacturers.
What is Excluded from the Scope of this Report?
Upstream drug substance manufacturing, downstream packaging operations, laboratory equipment, and pilot-scale operations are excluded unless part of integrated sterile processing platforms.
What does Market Forecast Mean on this Page?
Market forecast represents model-based projections built on manufacturing capacity expansion and pharmaceutical demand assumptions for strategic planning purposes.
How does FMI Build and Validate the Fill Finish Manufacturing Market Forecast?
Forecast is developed using hybrid modeling validated through manufacturing capacity databases, regulatory inspection tracking, and production facility verification.
What does Zero Reliance on Speculative Third Party Market Research Mean Here?
Market analysis is based on verifiable manufacturing capabilities, facility inspections, and commercial production data rather than speculative technology adoption timelines.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Prefilled syringes
- Vials
- Cartridges
- Ampoules
- Prefilled syringes
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Biologics and vaccines
- Small Molecules
- Cell & Gene Therapies
- Others
- Biologics and vaccines
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Application
- Competition Analysis
- Competition Deep Dive
- Catalent Inc.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- West Pharmaceutical Services
- Gerresheimer AG
- Lonza Group AG
- Samsung Biologics
- WuXi Biologics
- Recipharm AB
- Catalent Inc.
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Product Type
- Figure 23: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Application
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Product Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Application
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Product Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Application
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Application
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Product Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Application
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

