Injectable Drug Market
Injectable Drug Market Size and Share Forecast Outlook 2026 to 2036
Injectable Drug Market Forecast and Outlook 2026 to 2036
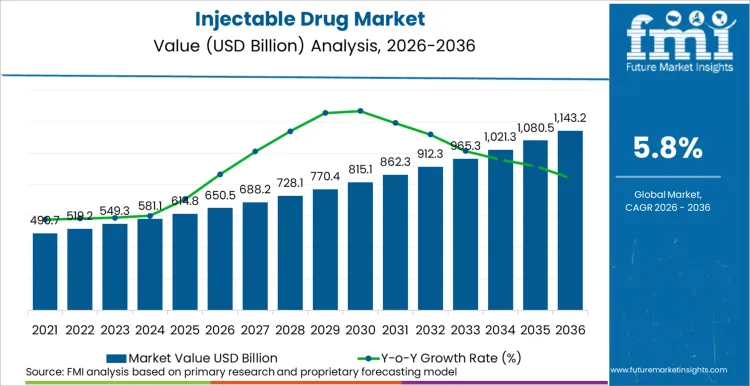
The Injectable Drug market valuation is poised to ascend from USD 650.5 Billion in 2026 to USD 1,100.0 Billion by 2036, registering a CAGR of 5.8%. growth is driven by the rise of biologic therapies and high-value oncology treatments, especially antibody-drug conjugates (ADCs) and bispecific antibodies. In March 2025, CDER (FDA) disclosed that oncology medications made up 28% of all new approvals in 2024, underscoring the prominent role of parenteral administration in critical care.
“Importantly, [we’re] building four new sites in to‑be‑determined locations in the USA This is the largest single company action ever taken, and will, in total, be $50 billion in nine new factories.” states David Ricks, CEO of Eli Lilly. This suggests that while demand accelerates, manufacturers must prioritize capacity expansion and 24/7 operational resilience to prevent shortages.
Quick Stats for Injectable Drug Market
- Injectable Drug Market Value (2026): USD 650.5 Billion
- Injectable Drug Market Value (2036): USD 1,100.0 Billion
- Injectable Drug Market Forecast CAGR: 5.8%
- Leading Component: Monoclonal Antibodies (25.2%)
- Leading End User: Hospitals
- Leading Application: Oncology (33%)
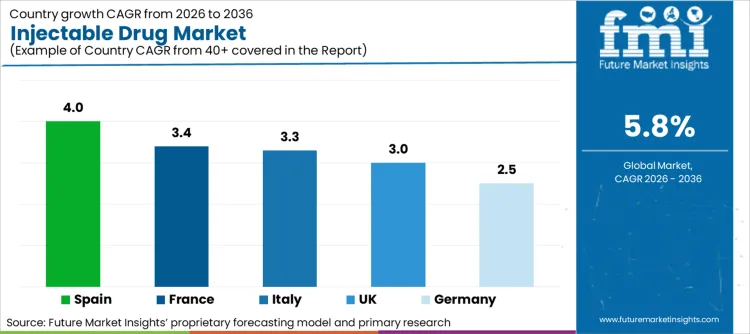
- Key Growth Regions: Germany, United Kingdom, Italy, France, Spain
- Key Players: Pfizer Inc., Teva Pharmaceutical Industries Ltd., Sanofi S.A., Sun Pharmaceutical Industries Ltd., AstraZeneca Plc, Merck & Co., Inc., Viatris Inc., Cipla Inc., Dr. Reddy's Laboratories Ltd., Samsung Biologics
Companies are actively diversifying into self-administered devices and biosimilars to meet cost-containment demands. A report from the ASHP in October 2024 highlighted that 84% of healthcare respondents reported critical impacts from sterile fluid shortages, underscoring the urgent need for supply chain redundancy. Such efficiency gains drive adoption among institutional investors seeking yield optimization in the pharmaceutical sector. FMI projects monoclonal antibody segments will continue dominating global demand, supported by robust R&D pipelines targeting autoimmune disorders and cancer.
Injectable Drug Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 650.5 Billion |
| Industry Value (2036) | USD 1,100.0 Billion |
| CAGR (2026-2036) | 5.8% |
Source: Future Market Insights (FMI) analysis.
What Drivers Are Fueling Injectable Drug Adoption?
Clinical preferences are shifting towards high-potency biologics, necessitating sterile parenteral delivery routes. Scaling operations requires robust cold chain logistics to ensure the efficacy of temperature-sensitive therapies like insulin and vaccines. 'Looking ahead, 2026 will be a milestone-rich year... together, these pipeline assets represent a potential of over $10 billion,' notes Richard Francis, CEO of Teva Pharmaceutical Industries. This shift drives demand for generic injectables and small molecule injectables portfolios. Findings from Gavi, the Vaccine Alliance in September 2025 highlight that immunization programs saved a record 1.7 million lives in 2024, validating the critical role of injectable vaccines. Such consolidation mandates injectable drug delivery innovation to manage complex formulations efficiently at scale.
How is Injectable Drug Market Segmentation Evolving?
Market segmentation within the Injectable Drug sector is undergoing a fundamental pivot, moving away from simple vials toward user-friendly combination products. Stakeholders are increasingly prioritizing solutions that offer convenience, such as pre-filled syringes and auto-injectors. This evolution is accelerating as chronic disease management shifts home, driving the adoption of self-administered parenteral strategies. FMI analysis reveals that capital is disproportionately flowing into oncology and immunology sectors, supported by emerging interest in drug-device combination products for precise dosing. As patient adherence remains a critical driver, the market is favoring vendors who offer connected delivery systems, reshaping competitive boundaries and driving consolidation.
Why do Monoclonal Antibodies Dominate Product Types?
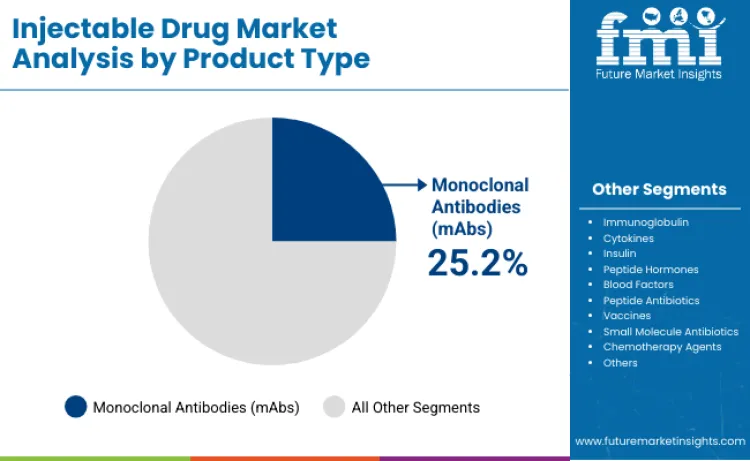
Monoclonal Antibodies (mAbs) are expected to hold a dominant 25.2% share of the product segment, primarily because they offer targeted mechanisms of action for treating complex diseases like cancer. Unlike small molecules, mAbs require parenteral administration to avoid gastric degradation, ensuring high bioavailability. This dominance is reinforced by the integration of advanced pharmaceutical manufacturing equipment, which enables the sterile production of these large molecules. The preference for mAbs is driven by efficacy; usage statistics indicate that the multi-drug combination injectable sector heavily relies on these biologics for first-line therapies. By consolidating multiple functions-such as immune system engagement and tumor targeting-into one therapy, mAbs significantly improve clinical outcomes.
Why is Intravenous (IV) the Leading Route?
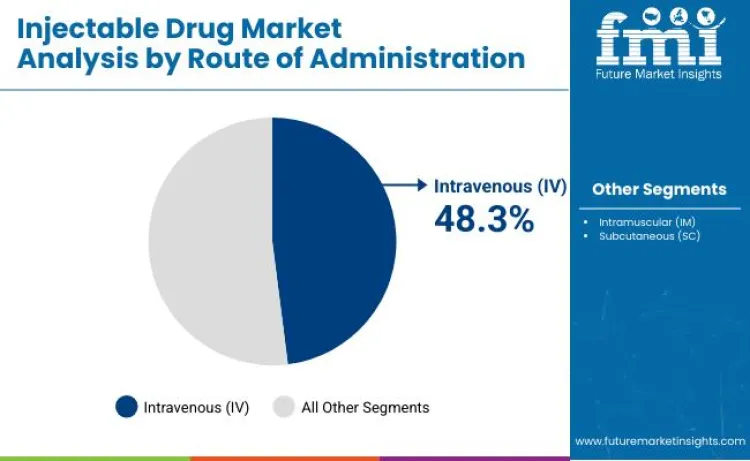
Intravenous (IV) administration is projected to lead the market with a 48.3% share, fueled by the need for rapid onset of action in acute care settings. This segment's growth is underpinned by hospitals that are actively utilizing parenteral compounding services to prepare customized infusions. Data published by the IQVIA Institute in July 2025 highlights that sterile injectables account for the majority of active drug shortages, driving demand for robust IV fluid supply chains. This concentration of demand necessitates robust pharmaceutical grade sodium chloride sourcing to maintain hospital operations. As clinicians continue to seek immediate therapeutic effects, the demand for IV delivery in emergency and inpatient scenarios will continue to outpace subcutaneous methods.
What Trends Are Shaping the Injectable Drug Landscape?
- GLP-1 Supply Chain Expansion: Obesity therapies are driving manufacturing upgrades. In February 2025, Eli Lilly announced a $27 billion investment to enhance manufacturing for Zepbound, addressing overwhelming demand. This pushes demand for syringes and injectable drugs packaging and high-volume cartridge production.
- Biosimilar Market Penetration: Competition is lowering biologic costs. In February 2025, Amgen launched biosimilars for Eylea and Soliris, expanding patient access to complex therapies. This trend boosts drug formulation and rare disease treatment adoption.
- Connected Delivery Devices: Digital health is merging with injectables. Companies are integrating smart sensors into pens and auto-injectors. This aligns with the growth in the connected drug delivery devices to monitor adherence in chronic conditions.
How Will Injectable Drug Expansion Unfold Across Key Global Regions?
Global markets are bifurcating into mature innovator hubs and rapid-growth biosimilar economies. Germany focuses on high-tech packaging and manufacturing, while the UK prioritizes cost-effective biosimilar adoption within the NHS. Lyophilized injectable and subcutaneous drug delivery sectors are witnessing varied adoption rates. Developed nations leverage drug delivery technology to enhance patient convenience, whereas emerging regions utilize robust generic pipelines to expand essential medicine access.
|
Country |
CAGR (2026 to 2036) |
|
Germany |
2.5% |
|
United Kingdom |
3.0% |
|
Italy |
3.3% |
|
France |
3.4% |
|
Spain |
4.0% |
How is Germany driving pharmaceutical packaging innovation?
Sales of Injectable Drugs in Germany are set to rise at 2.5% CAGR. Germany remains a global hub for high-quality glass and polymer packaging essential for biologics. Sector is witnessing a surge in demand for pre-fillable syringes tailored for GLP-1 drugs. Data released by Gerresheimer AG in May 2025 indicated that revenues from biologic solutions are increasing, driving profitability despite slower general market growth. “Together, we have delivered industry‑leading innovations, achieved remarkable growth, and improved the health of many people in a sustainable way.” Andreas Reisse, CEO of SCHOTT Pharma This implies that German manufacturers are pivoting toward premium, high-margin containment solutions.
What factors are accelerating adoption in the United Kingdom?
Demand for Injectable Drugs in UK is anticipated to grow at 3.0% CAGR. The UK market is driven by NHS initiatives to adopt biosimilars and reduce long-term healthcare costs. The market is witnessing a surge in domestic manufacturing capacity for sterile injectables. Statistics from Hikma Pharmaceuticals in August 2025 projected that new launches would provide a tailwind, with the company seeing opportunities to compete in high-value segments. 'We remain excited for the full year Injectables performance,' states Riad Mishlawi, CEO of Hikma Pharmaceuticals. This suggests a robust environment for generic and biosimilar market penetration.
Why is Italy maintaining manufacturing excellence?
The Injectable Drug industry in Italy is projected to expand at 3.3% CAGR. Italy's CDMO (Contract Development and Manufacturing Organization) sector is a critical pillar, supplying sterile products to global pharma giants. The market is pivoting towards complex biologics and vaccine production. Investments in sterile fill-finish capacity are driving export growth to North American and Asian markets. Research confirms that Italy's pharmaceutical production reached €56.1 billion in 2024, an 18% value added increase since 2022, solidifying its position as a top EU manufacturer. This indicates that Italy will remain a strategic manufacturing node for the global supply chain.
How does France lead in vaccine development?
Injectable Drug market in France is poised to register a 3.4% CAGR. Growth is characterized by a strong focus on immunology and preventative medicine. The country is seeing massive investments in mRNA and traditional vaccine platforms. In October 2025, Sanofi reported positive results for its investigational flu vaccine, reinforcing its dominance in the respiratory immunization space. 'Our strong sales growth was driven by our biopharma launches and our vaccines portfolio,' states Paul Hudson, CEO of Sanofi. This confirms that France is leveraging its R&D heritage to lead in next-generation injectable vaccines.
What role does Spain play in plasma therapies?
Sales of Injectable Drugs in Spain are set to rise at 4.0% CAGR. Spain is becoming a key player in plasma-derived therapies and hospital-administered injectables. The industry is witnessing a surge in demand for immunoglobulin and blood factors. Local players are expanding capacity to meet both domestic and international demand for critical care products. A study in 2024 highlight that pharmaceutical manufacturing reached €23 billion, a 40% increase over three years, with 174 production plants operating in the country. This confirms Spain's growing role as a reliable production hub for essential biological medicines.
What Strategic Moves Are Defining Competitive Landscape?
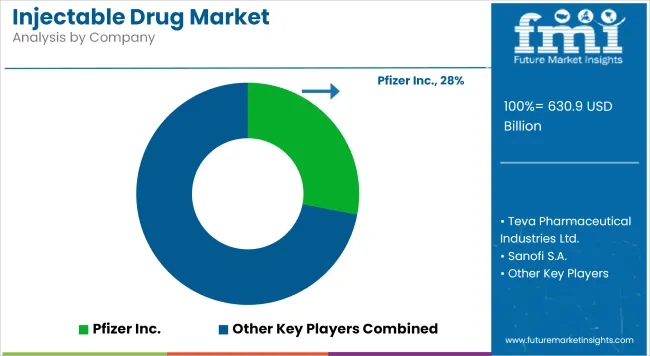
The market structure is undergoing significant consolidation as dominant players seek to establish "Bio-Pharma" ecosystems. Leading firms are moving beyond simple molecule production to offer comprehensive patient support programs and device integration. FMI analysis indicates a strategic shift toward "Pipeline-in-a-Product" models where single assets are developed for multiple indications. This evolution is driving aggressive M&A activity, where established giants acquire biotech innovators to enhance their immunology portfolios and secure a competitive moat against emerging biosimilar disruptors.
Strategies are increasingly focused on operational efficiency and R&D prioritization. 'Pfizer is implementing cost-cutting measures and refocusing its research and development efforts,' states Albert Bourla, CEO of Pfizer, highlighting the industry's discipline. This suggests that market leaders use strategic capital allocation to fund high-impact projects. In 2025, Samsung Biologics confirmed its strategy to expand its manufacturing footprint, securing a KRW 1.1 trillion agreement with a European pharma company and planning a US facility acquisition.
Key Players
- Pfizer Inc.
- Teva Pharmaceutical Industries Ltd.
- Sanofi S.A.
- Sun Pharmaceutical Industries Ltd.
- AstraZeneca Plc
- Merck & Co., Inc.
- Viatris Inc.
- Cipla Inc.
- Dr. Reddy's Laboratories Ltd.
- Samsung Biologics
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units | USD Billion |
| Product Type | Monoclonal Antibodies, Immunoglobulin, Cytokines, Insulin, Peptide Hormones, Others |
| Route of Administration | Intravenous (IV), Intramuscular (IM), Subcutaneous (SC) |
| Application | Oncology, Infectious Diseases, Diabetes, Blood Disorders, Hormonal Disorders, Musculoskeletal Disorders, CNS Diseases, Pain Management, Cardiovascular Diseases |
| Regions | North America, Latin America, Europe, East Asia, South Asia & Pacific, Middle East & Africa |
Injectable Drug Market by Segments
Product Type
- Monoclonal Antibodies
- Immunoglobulin
- Cytokines
- Insulin
- Peptide Hormones
- Blood Factors
- Peptide Antibiotics
- Vaccines
- Small Molecule Antibiotics
- Chemotherapy Agents
- Others
Route of Administration
- Intravenous (IV)
- Intramuscular (IM)
- Subcutaneous (SC)
Application
- Oncology
- Infectious Diseases
- Diabetes
- Blood Disorders
- Hormonal Disorders
- Musculoskeletal Disorders
- CNS Diseases
- Pain Management
- Cardiovascular Diseases
Bibliography
- USA Food and Drug Administration, Center for Drug Evaluation and Research. (2025, March). New drugs at FDA: CDER’s new molecular entities and new therapeutic biological products. USA Department of Health and Human Services.
- American Society of Health-System Pharmacists. (2024, October). Sterile fluid shortage survey. ASHP.
- IQVIA Institute for Human Data Science. (2025, July). Trends in drug shortages and ANDA approvals. IQVIA Institute.
- Gavi, the Vaccine Alliance. (2025, September). Annual impact report. Gavi.
- CNN. (2025, February). Eli Lilly manufacturing investment expands USA capacity. CNN.
- Centers for Disease Control and Prevention. (2026, January). Flu vaccination coverage estimates, 2024-2025 season. USA Department of Health and Human Services.
- Hikma Pharmaceuticals PLC. (2025, August). Interim results presentation. Hikma Pharmaceuticals.
Frequently Asked Questions
What is the current global market size for the Injectable Drug category?
The Injectable Drug Market valuation is estimated at USD 650.5 Billion in 2026, driven by biologics adoption.
What is the projected Compound Annual Growth Rate (CAGR) for the Injectable Drug market over the next 10 years?
The sector is expected to grow at a CAGR of 5.8% through 2036.
Which high-growth therapeutic areas are the primary drivers of demand?
Oncology applications are the primary drivers, accounting for 33% of the market share due to high-value antibody therapies.
What are the primary market risks, supply chain constraints, or regulatory hurdles affecting the Injectable Drug market globally?
Sterile manufacturing shortages and complex regulatory approval pathways for biosimilars are significant challenges.
Who are the top 3-5 leading suppliers, and what is the basis of their market leadership?
Leading players include Pfizer, Sanofi, and Teva, defined by their extensive biologic pipelines and global manufacturing footprint.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Roots Internal Knowledge Base and Historical Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended A8oach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Monoclonal Antibodies
- Immunoglobulin
- Cytokines
- Insulin
- Peptide Hormones
- Blood Factors
- Peptide Antibiotics
- Vaccines
- Small Molecule Antibiotics
- Chemotherapy Agents
- Others
- Monoclonal Antibodies
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Route of Administration
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Route of Administration, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Route of Administration, 2026 to 2036
- Intravenous (IV)
- Intramuscular (IM)
- Subcutaneous (SC)
- Intravenous (IV)
- Y to o to Y Growth Trend Analysis By Route of Administration, 2021 to 2025
- Absolute $ Opportunity Analysis By Route of Administration, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Oncology
- Infectious Diseases
- Diabetes
- Blood Disorders
- Hormonal Disorders
- Musculoskeletal Disorders
- CNS Diseases
- Pain Management
- Cardiovascular Diseases
- Oncology
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Route of Administration
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Route of Administration
- By Application
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Route of Administration
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Route of Administration
- By Application
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Route of Administration
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Route of Administration
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Route of Administration
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Route of Administration
- By Application
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Route of Administration
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Route of Administration
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Route of Administration
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Route of Administration
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Route of Administration
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Route of Administration
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Route of Administration
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Route of Administration
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Route of Administration
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Route of Administration
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Route of Administration
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Route of Administration
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Route of Administration
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Route of Administration
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Route of Administration
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Route of Administration
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Route of Administration
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Route of Administration
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Route of Administration
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Route of Administration
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Route of Administration
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Route of Administration
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Route of Administration
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Route of Administration
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Route of Administration
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Route of Administration
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Route of Administration
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Route of Administration
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Route of Administration
- By Application
- Competition Analysis
- Competition Deep Dive
- Pfizer Inc.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Teva Pharmaceutical Industries Ltd.
- Sanofi S.A.
- Sun Pharmaceutical Industries Ltd.
- AstraZeneca Plc
- Merck & Co., Inc.
- Viatris Inc.
- Cipla Inc.
- Dr. Reddy's Laboratories Ltd.
- Samsung Biologics
- Abbott Laboratories
- Pfizer Inc.
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Route of Administration
- Figure 9: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Application
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Product Type
- Figure 26: North America Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Route of Administration
- Figure 29: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Application
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Product Type
- Figure 36: Latin America Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by Route of Administration
- Figure 39: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Application
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Product Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by Route of Administration
- Figure 49: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by Application
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Route of Administration
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by Application
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Product Type
- Figure 66: East Asia Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by Route of Administration
- Figure 69: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by Application
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Route of Administration
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Route of Administration
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

