The sleep apnea implants market is valued at USD 480.6 million in 2026 and is projected to reach USD 697.8 million by 2036, reflecting a CAGR of 3.8%. Growth is driven by increasing prevalence of obstructive and central sleep apnea and rising adoption of implantable neurostimulation therapies. Product types include phrenic nerve stimulators, hypoglossal neurostimulation devices, bone screw implant systems, and palatal implants, with hypoglossal neurostimulation devices representing approximately 60% of offerings. Cost structures are shaped by device development, regulatory approval, and clinical validation, while margin concentration favors companies providing clinically validated, multi-indication implants rather than volume-focused production.
Between 2026 and 2036, adoption varies by patient population, healthcare infrastructure, and physician familiarity with implantable therapies. Indication focus spans obstructive sleep apnea, which accounts for the majority of procedures, and central sleep apnea. Leading companies including Inspire Medical Systems, Respicardia, Medtronic Plc, Pillar Palatal LLC, Siesta Medical, LivaNova/ImThera, Nyxoah SA, and LinguaFlex LLC capture value through proprietary device design, regulatory compliance, and clinical support programs. Fragmentation persists in smaller regional markets with niche providers, whereas established players maintain pricing power, high margin concentration, and long-term revenue visibility through validated implant solutions.
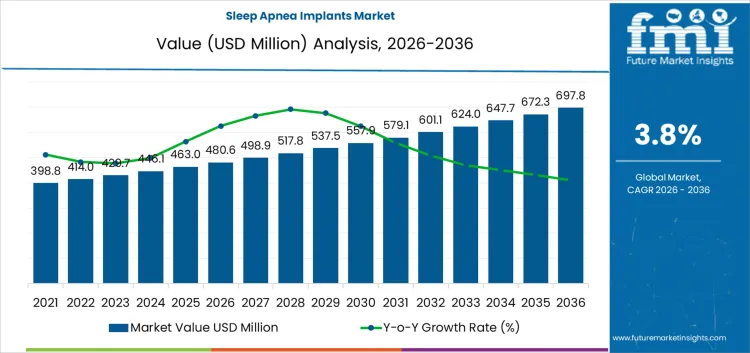
Between 2026 and 2031, the sleep apnea implants market is projected to grow from USD 480.6 million to USD 558.1 million, generating an absolute increase of USD 77.5 million and reflecting a CAGR of 3.8%. Growth during this phase is driven by rising prevalence of obstructive sleep apnea, increased adoption of implantable devices, and awareness among healthcare providers and patients. Volume expansion contributes approximately 68% of growth, supported by procedural uptake and hospital integration, while price growth accounts for 32%, driven by device enhancements, premium offerings, and clinical validation. Value capture favors manufacturers with regulatory approvals and strong hospital networks.
From 2031 to 2036, the market is expected to expand from USD 558.1 million to USD 697.8 million, adding USD 139.7 million. Volume continues to dominate growth, representing roughly 70% of the increase due to expanding patient base, broader adoption in emerging markets, and additional clinical indications. Price growth accounts for 30%, supported by innovative device designs and enhanced clinical support services. Competitive advantage shifts toward firms offering validated, reliable implants, comprehensive training programs, and strong clinical partnerships, while late entrants focus on cost efficiency and niche device innovations to capture incremental market share.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 480.6 million |
| Forecast Value (2036) | USD 697.8 million |
| Forecast CAGR 2026 to 2036 | 3.8% |
The sleep apnea implants market is expanding as minimally invasive therapies gain traction among patients with obstructive sleep apnea (OSA) who struggle with traditional CPAP or oral appliances. Historically, treatment relied heavily on external devices, which faced compliance challenges and limited long-term effectiveness. Modern implants, including hypoglossal nerve stimulators and palatal implants, mechanically support airway patency during sleep while reducing dependence on external devices. Hospitals, sleep clinics, and otolaryngology centers focus on implantation safety, device reliability, and patient comfort. Early adoption was restricted to severe OSA cases or patients intolerant to CPAP, while current uptake extends to broader patient segments seeking long-term, low-maintenance interventions with proven efficacy.
Future demand is driven by increasing OSA prevalence, greater diagnostic awareness, and expanding insurance coverage rather than incremental procedural innovation alone. Compared with initial devices, contemporary implants feature optimized stimulation algorithms, longer battery life, and improved MRI safety. Cost considerations include device production, surgical implantation, and follow-up monitoring, concentrating margins among manufacturers with validated clinical outcomes and regulatory approvals. Healthcare providers adopt these implants to enhance patient adherence, reduce secondary complications, and integrate device-based therapy into comprehensive OSA management. By 2036, sleep apnea implants are expected to become a standard clinical solution, complementing conventional therapies and supporting improved patient quality of life.
The demand for sleep apnea implants is segmented by product type and indication. Product types include phrenic nerve stimulators, hypoglossal neurostimulation devices, bone screw implant systems, and palatal implants or pillar procedures. Indications cover central sleep apnea (CSA) and obstructive sleep apnea (OSA). Segment adoption is influenced by severity, patient eligibility, clinical efficacy, and regulatory approval. Uptake is driven by the need for minimally invasive interventions, improved patient outcomes, and long-term symptom management rather than marketing considerations. Selection of product and indication depends on anatomical compatibility, therapeutic effectiveness, and integration with patient-specific treatment plans.
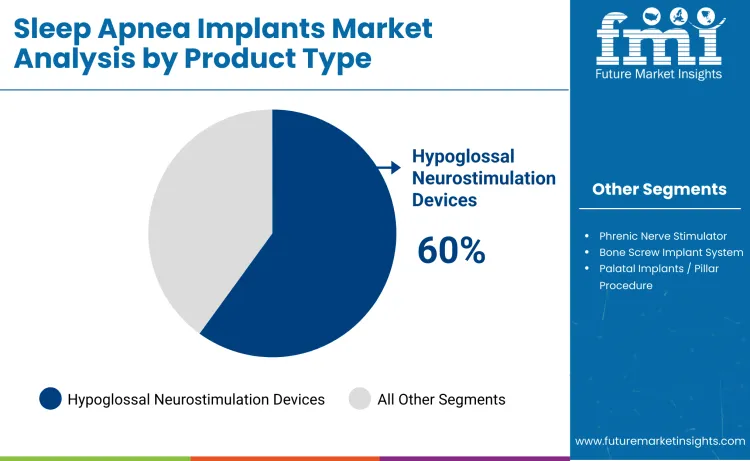
Hypoglossal neurostimulation devices account for approximately 60% of total product demand, making them the leading category. This dominance reflects their clinical effectiveness in treating obstructive sleep apnea by activating the tongue muscles to maintain airway patency during sleep. Clinicians favor these devices due to minimally invasive implantation, predictable therapy outcomes, and long-term symptom reduction. Adoption is reinforced by high patient adherence and regulatory approvals across major healthcare markets. The devices are compatible with diagnostic titration and remote monitoring, supporting tailored therapy based on individual airway dynamics.
Demand for hypoglossal neurostimulation devices is further shaped by evidence of efficacy and safety. Clinical studies demonstrate significant reductions in apnea-hypopnea index, improved oxygen saturation, and enhanced sleep quality. Hospitals and specialized sleep centers prioritize devices that reduce adverse events and ensure consistent therapeutic benefit. The segment leads because hypoglossal neurostimulation provides targeted, effective, and minimally invasive treatment for OSA patients, establishing it as the primary product driving growth in the sleep apnea implants market.
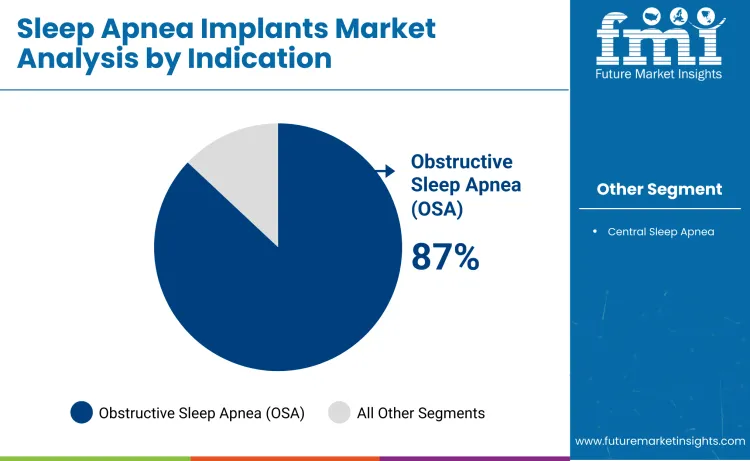
Obstructive sleep apnea (OSA) accounts for approximately 87% of total indication demand, making it the largest segment. OSA involves repeated airway obstruction during sleep, leading to disrupted breathing, hypoxia, and cardiovascular complications. Implantable devices, such as hypoglossal neurostimulation systems, offer mechanical and neuromuscular support to maintain airway patency and reduce apneic events. Adoption is driven by the high prevalence of OSA, clinical guideline recommendations, and patient demand for effective minimally invasive therapies.
Demand in OSA is shaped by clinical urgency, patient quality of life, and therapy adherence. Sleep centers and hospitals prioritize implants that deliver reliable symptom reduction, improve oxygenation, and support long-term compliance. Device selection is guided by anatomical assessment, therapeutic response, and ease of implantation. The segment leads because OSA represents the majority of treatable sleep apnea cases, combining high patient volume with clear clinical benefit, establishing it as the primary driver of growth in the sleep apnea implants market.
Sleep apnea implants are increasingly used as a long-term alternative to CPAP devices, targeting patients with moderate to severe obstructive sleep apnea who struggle with conventional therapies. Adoption is strongest in regions with specialized sleep clinics, advanced surgical infrastructure, and trained ENT surgeons. Devices are selected based on airway support effectiveness, minimally invasive implantation, and patient comfort. Growth is driven by clinical outcomes and regulatory approvals rather than volume of devices sold. Hospitals and clinics prioritize implants that reduce apnea-hypopnea episodes, improve oxygen saturation, and demonstrate predictable efficacy. Procedural reliability and regulatory compliance are critical factors for adoption.
Demand is shaped by local healthcare policies, clinical protocols, and insurance reimbursement schemes that support surgical interventions for sleep apnea. Awareness campaigns and patient education in certain regions increase the number of candidates seeking implantable solutions. Hospitals and surgeons prioritize devices with proven long-term efficacy and minimal complications to ensure patient adherence and satisfaction. The driver is the combination of regulatory endorsement, clinical validation, and patient-centered care rather than cost advantages. Facilities capable of integrating implant procedures into existing sleep disorder programs achieve higher adoption rates and improved patient outcomes.
Adoption is constrained by high device costs, limited access to trained surgeons, and procedural complexity. Pre-operative assessment and post-operative monitoring require significant clinical resources. Some patients are not suitable candidates due to anatomical or health considerations. Regulatory approval processes and regional certification requirements can delay market entry. Smaller hospitals or clinics may lack the infrastructure or patient volume to justify investment. These challenges concentrate adoption in specialized centers with expertise in sleep medicine and surgical intervention, slowing broader penetration despite clinical benefits.
Innovations focus on miniaturized, bio-compatible implants, improved surgical tools, and enhanced patient-specific customization. Partnerships between manufacturers, surgeons, and clinical researchers support procedural training, long-term outcome studies, and iterative product development. Pilot programs are increasingly used to validate safety and efficacy before full-scale deployment. Data tracking and digital monitoring improve post-operative care and regulatory reporting. Focus is on patient safety, procedural reproducibility, and regulatory compliance rather than production volume or cost. Collaborative efforts accelerate adoption in specialized centers, enhancing treatment options for sleep apnea patients across regional markets.
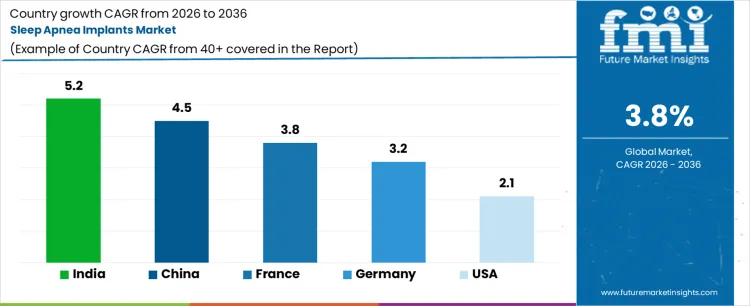
| Country | CAGR (%) |
|---|---|
| USA | 2.1% |
| Germany | 3.2% |
| France | 3.8% |
| China | 4.5% |
| India | 5.2% |
Demand for sleep apnea implants is increasing as awareness of obstructive sleep apnea (OSA) grows and minimally invasive implantable therapies gain acceptance. India leads with a 5.2% CAGR, supported by rising prevalence of OSA, growing healthcare infrastructure, and adoption of cost-effective implantable solutions. China follows at 4.5%, driven by increasing patient diagnosis rates and hospital adoption of implant-based therapies. France records 3.8% growth, shaped by advanced medical facilities and reimbursement support. Germany grows at 3.2%, reflecting steady adoption in specialized ENT centers. The USA shows 2.1% CAGR, influenced by moderate growth in surgical interventions and established non-invasive therapies limiting rapid expansion of implant adoption.
United States sleep apnea implants market is expanding at a CAGR of 2.1%, supported by increasing prevalence of obstructive sleep apnea and growing interest in minimally invasive treatments. Hospitals and specialized clinics are implementing implantable airway stimulation devices to enhance patient outcomes and reduce reliance on CPAP therapy. Demand is concentrated among tertiary care centers, sleep clinics, and specialized ENT hospitals. Investments prioritize device reliability, procedural training, and regulatory compliance rather than rapid facility expansion. Growth reflects steady adoption of innovative therapies alongside ongoing patient awareness campaigns and clinical adoption of advanced treatment protocols.
Germany is growing through increased adoption of minimally invasive sleep apnea therapies, with a market CAGR of 3.2%. Hospitals and ENT centers are deploying implantable devices to enhance treatment efficacy, reduce patient discomfort, and improve surgical outcomes. Demand is concentrated in urban hospitals, university medical centers, and specialized sleep clinics. Investments focus on device performance, clinician training, and procedural standardization rather than expanding hospital networks. Growth reflects patient preference for advanced therapies and alignment with national healthcare standards for minimally invasive sleep disorder interventions.
France is expanding in the sleep apnea implants sector at a CAGR of 3.8%, driven by adoption of upper airway stimulation devices and increasing patient awareness of minimally invasive therapies. Hospitals and clinics are utilizing implants to improve airway function and reduce dependence on CPAP. Demand is concentrated among specialized sleep centers, tertiary hospitals, and university medical facilities. Investments prioritize procedural training, device reliability, and regulatory alignment rather than large-scale infrastructure expansion. Growth reflects steady uptake of innovative solutions and evolving patient preference for less intrusive treatment options.
China is witnessing growth in sleep apnea implants at a CAGR of 4.5%, fueled by rising prevalence of obstructive sleep apnea and increasing awareness of implantable treatment options. Hospitals and specialized ENT clinics are deploying airway stimulation devices to improve patient outcomes and treatment compliance. Demand is concentrated in urban healthcare centers and tertiary hospitals. Investments focus on device performance, procedural standardization, and regulatory compliance rather than rapid network expansion. Growth reflects increasing diagnosis rates, clinical adoption of minimally invasive procedures, and patient demand for innovative therapies.
India is experiencing rapid growth in sleep apnea implants with a CAGR of 5.2%, supported by rising prevalence of sleep disorders, expanding hospital infrastructure, and increasing adoption of implantable airway stimulation devices. Hospitals and specialized clinics are implementing devices to enhance therapy adherence and improve patient quality of life. Demand is concentrated among urban tertiary care hospitals and ENT specialty centers. Investments prioritize device reliability, clinician training, and regulatory compliance rather than rapid facility expansion. Growth reflects both increasing patient awareness and rising preference for minimally invasive treatment options.
Competition in the sleep apnea implants market is shaped by differences in technology approach, mechanism of action, and clinical support for obstructive sleep apnea (OSA) patients who are intolerant of continuous positive airway pressure therapy. Inspire Medical Systems leads with a hypoglossal nerve stimulation system that uses implantable neurostimulation to maintain airway patency during sleep, supported by a growing clinical evidence base and defined patient selection protocols. Respicardia, Inc. developed an implantable transvenous phrenic nerve stimulation system targeting central sleep apnea, differentiating by addressing a distinct OSA subtype and offering programmable therapy synchronized with patient breathing patterns. Medtronic Plc participates through broader neurostimulation expertise, exploring implantable approaches that integrate with its existing portfolio of implantable devices and clinician support systems.
Smaller and emerging competitors focus on niche technologies or device design innovations. Pillar Palatal LLC and Siesta Medical, Inc. explore implants aimed at supporting soft tissue structures in the upper airway to reduce collapse. LivaNova/ImThera and Nyxoah SA develop alternative stimulation strategies with varying implant sites and stimulation algorithms. LinguaFlex LLC focuses on flexible, minimally invasive implant concepts to address tongue base obstruction. Others in the market contribute emerging technologies in implant geometry, energy delivery, and integration with diagnostic systems. Competitive differentiation arises from therapy mechanism, ease of implantation, patient selection criteria, long term clinical outcomes, and integration with sleep medicine care pathways, with practitioners choosing systems based on evidence, patient anatomy, and device support infrastructure.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD million |
| Product | Phrenic Nerve Stimulator, Hypoglossal Neurostimulation Devices, Bone Screw Implant System, Palatal Implants / Pillar Procedure |
| Indication | Central Sleep Apnea, Obstructive Sleep Apnea (OSA) |
| End User | Hospitals, Sleep Clinics, ENT Specialty Centers, Tertiary Care Centers |
| Region | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Key Countries Covered | USA, Germany, France, China, India, Japan, South Korea, United Kingdom, Italy, Spain, Nordic, BENELUX, Australia & New Zealand, ASEAN, Rest of Asia Pacific, Rest of Europe, Canada, Mexico, Brazil, Chile, Kingdom of Saudi Arabia, Other GCC Countries, Turkey, South Africa, Other African Union, Rest of Middle East & Africa |
| Key Companies Profiled | Inspire Medical Systems, Respicardia, Inc., Medtronic Plc, Pillar Palatal LLC, Siesta Medical, Inc., LivaNova/ImThera, Nyxoah SA, LinguaFlex LLC, Others |
| Additional Attributes | Dollar sales by product and indication; adoption by patient population and healthcare infrastructure; procedural volume vs. price contribution; margin concentration among clinically validated implants; competitive differentiation through device efficacy, regulatory approval, clinical support programs, and training; regional adoption influenced by healthcare awareness, insurance coverage, and clinical guidelines |
How big is the sleep apnea implants market in 2026?
The global sleep apnea implants market is estimated to be valued at USD 480.6 million in 2026.
What will be the size of sleep apnea implants market in 2036?
The market size for the sleep apnea implants market is projected to reach USD 697.8 million by 2036.
How much will be the sleep apnea implants market growth between 2026 and 2036?
The sleep apnea implants market is expected to grow at a 3.8% CAGR between 2026 and 2036.
What are the key product types in the sleep apnea implants market?
The key product types in sleep apnea implants market are phrenic nerve stimulator, hypoglossal neurostimulation devices, bone screw implant system and palatal implants / pillar procedure.
Which indication segment to contribute significant share in the sleep apnea implants market in 2026?
Obstructive sleep apnea (OSA) will dominate the indication segment with 87% share, while central sleep apnea accounts for the remaining share
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.