Home Sleep Apnea Testing Market
The Home Sleep Apnea Testing Market is Segmented by Product (Wearable Sleep Screening Devices, Non-Wearable Sleep Screening Devices, Smart Sleep Equipment, Home Polysomnography Devices, Others), Indication (Obstructive Sleep Apnea, Central Sleep Apnea), and Distribution Channel (Offline Sales, Online Sales). Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Home Sleep Apnea Testing Market Size, Market Forecast and Outlook By FMI
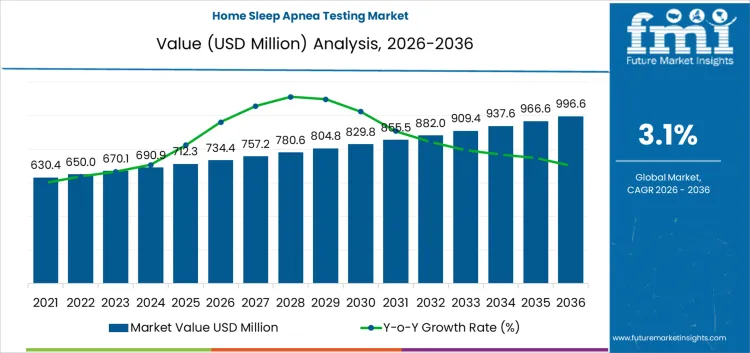
The home sleep apnea testing market is expected to expand from USD 734.4 million in 2026 to USD 996.6 million by 2036. The market is anticipated to register a 3.1% CAGR during the forecast period. Wearable sleep screening devices are likely to lead product with a 66.1% share, while obstructive sleep apnea is projected to account for 95.7% share of indication in 2026.
Summary of the Home Sleep Apnea Testing Market
- Demand and Growth Drivers
- Sleep laboratory capacity limits are expected to support wider use of home-based diagnostic pathways.
- Telehealth adoption is likely to increase demand for remote sleep screening and clinician review workflows.
- Patient convenience is anticipated to improve testing completion rates in eligible suspected apnea cases.
- Product and Segment View
- Wearable sleep screening devices are expected to lead product demand due to simplified overnight monitoring.
- Obstructive sleep apnea is likely to remain the dominant indication because home testing fits established screening protocols.
- Online sales are anticipated to gain use as diagnostic providers expand remote device distribution and follow-up models.
- Geography and Competitive Outlook
- India is expected to lead country-level growth due to limited sleep lab access and rising urban diagnosis needs.
- China is likely to record steady expansion as hospitals use home testing to triage large patient volumes.
- Companies with stronger sensor reliability and secure data platforms are anticipated to improve supplier positioning.
- Analyst Opinion
- Sabyasachi Ghosh, Principal consultant for Healthcare at FMI, suggests, “The home sleep apnea testing market is expected to favor suppliers that can improve diagnostic access without weakening clinical confidence. Providers are likely to prefer devices with reliable sensors, simple patient setup and secure data transfer into review workflows.”
- Home Sleep Apnea Testing Market Value Analysis
- The home sleep apnea testing market is moving from lab-dependent diagnosis toward outpatient screening support.
- Demand is likely to rise as clinicians use portable testing to shorten wait times for eligible patients.
- Adoption is expected to increase for wearable screening devices because they reduce setup complexity at home.
- Spending is anticipated to be supported by telehealth pathways and payer interest in cost-controlled diagnostics.
Home Sleep Apnea Testing Market Definition
Home sleep apnea testing covers portable diagnostic devices and related workflows used outside formal sleep laboratories for suspected sleep-disordered breathing. Scope includes systems used for airflow review, oxygen saturation tracking, respiratory effort capture, and clinician interpretation across home-based overnight testing.
Home Sleep Apnea Testing Market Inclusions
Market scope includes wearable sleep screening devices, non-wearable sleep screening devices, smart sleep equipment, and home polysomnography PSG devices. Coverage includes obstructive sleep apnea and central sleep apnea indications, along with offline and online sales channels used across home diagnostic deployment.
Home Sleep Apnea Testing Market Exclusions
Scope excludes revenue from in-laboratory polysomnography performed inside staffed sleep units and hospital-based overnight diagnostic facilities. General wellness sleep products and therapy-focused apnea devices are outside this market boundary unless they serve direct diagnostic use.
Home Sleep Apnea Testing Market Research Methodology
- Primary research covered sleep diagnostics stakeholders, outpatient testing channels, and clinician workflow patterns across leading healthcare services markets.
- Desk research covered published segment data, market structure notes, and country-level demand observations connected with home-based diagnostic use.
- Market sizing and forecasting aligned the 2026 base value, the stated CAGR, and segment-level demand concentration across product and indication groups.
- Data validation used cross-checking against forecast disclosures, qualitative market direction, and country pacing patterns across key national markets.
Why is the Home Sleep Apnea Testing Market Growing?
- Home testing is expanding its role in apnea screening as providers seek faster diagnosis outside crowded sleep laboratories.
- Portable devices support easier overnight assessment and reduce setup burden across clinically selected patients in outpatient care pathways.
- Digital review tools are improving workflow efficiency and helping clinicians manage higher screening volumes with less facility pressure.
Healthcare systems are moving first-line apnea evaluation away from overloaded laboratories and into portable home pathways. Use of sleep apnea diagnostic systems and sleep screening devices supports this change across clinician-guided screening programs. Portable testing helps sleep clinics and primary care networks reduce patient wait periods and keep overnight facility demand controlled. Home testing use is high in suspected obstructive cases with clear screening protocols and practical home suitability. This foundation supports steady demand as providers seek diagnostic access with manageable operating costs and controlled testing burden.
Device acceptance is rising through improved sensors and stable digital review platforms across remote diagnostic models. Expansion in wearable medical device and pulse oximeter adoption supports compact form factors used in overnight respiratory monitoring. Providers are using home pathways for timley patient sorting and efficient use of in-lab polysomnography capacity. Secure data handling and validation requirements keep supplier costs under pressure across this field. The model retains practical value across networks focused on access relief and timely clinician review.
Market Segmentation Analysis
- Wearable sleep screening devices are expected to account for 66.1% share in 2026 and lead product demand across unattended overnight testing use.
- Obstructive sleep apnea is projected to contribute 95.7% of total indication share in 2026 across home-based screening activity.
- Offline sales are estimated to lead distribution in 2026, reflecting clinician-led device supply and supervised onboarding across diagnostic programs.
- Sleep clinics are estimated to hold the leading care setting position in 2026 across organized home testing deployment.
- Initial risk stratification is estimated to command the larger workflow share in 2026 across patient selection and first-pass apnea screening.
The home sleep apnea testing market is divided into segments based on product type, indication, distribution channel, and region. Under product type, the market covers wearable sleep screening devices and non-wearable sleep screening devices. Product scope also extends to smart sleep equipment and home polysomnography PSG devices, along with other testing formats. By indication, the market is split into obstructive sleep apnea and central sleep apnea. Distribution channel coverage includes offline sales and online sales across clinician-related and direct ordering pathways. Regional analysis covers North America and Europe, along with Asia Pacific and other international markets included in the study.
Insights into the Wearable Sleep Screening Devices Segment
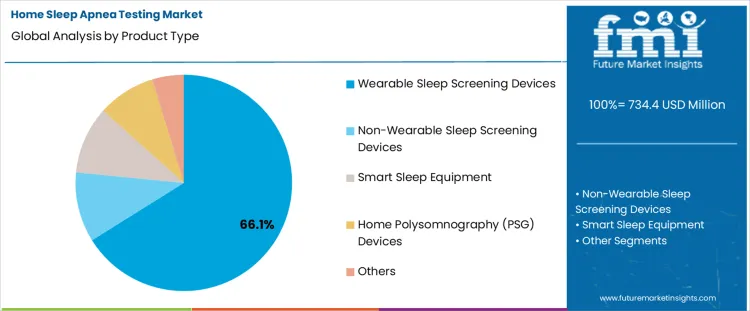
- Wearable sleep screening devices are expected to account for 66.1% share in 2026, because compact body-worn formats and steady overnight patient adherence keep this segment important.
- Product demand stays strong across this segment as simple setup reduces user error and supports timley clinician review after home test completion.
Insights into the Obstructive Sleep Apnea Segment
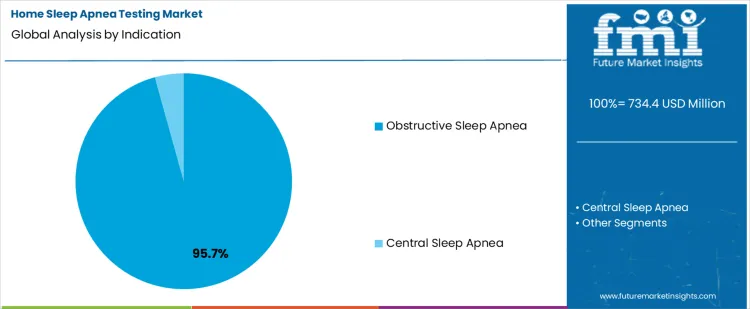
- Obstructive sleep apnea is projected to contribute 95.7% share in 2026, which keeps test volume concentrated inside one clinical indication.
- Home assessment aligns well with established screening parameters for this condition. This supports efficient patient sorting before detailed laboratory evaluation.
Insights into the Offline Sales Segment
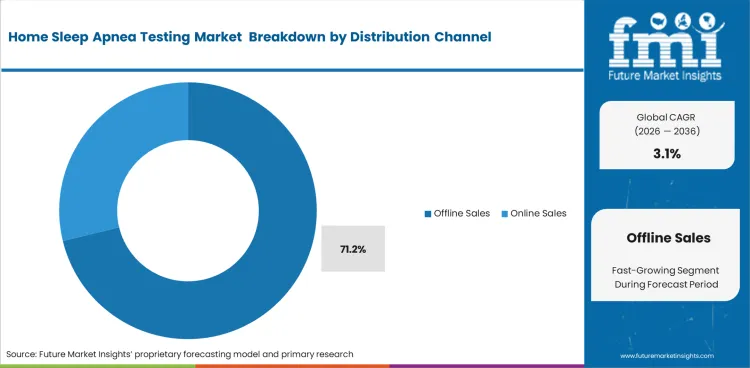
- Offline sales are estimated to represent 71.2% share in 2026, due to provider control across device delivery and patient onboarding.
- Clinic supply models keep this channel important because supervised setup and direct clinician guidance support complete data capture in unattended home studies.
Insights into the Sleep Clinics Segment
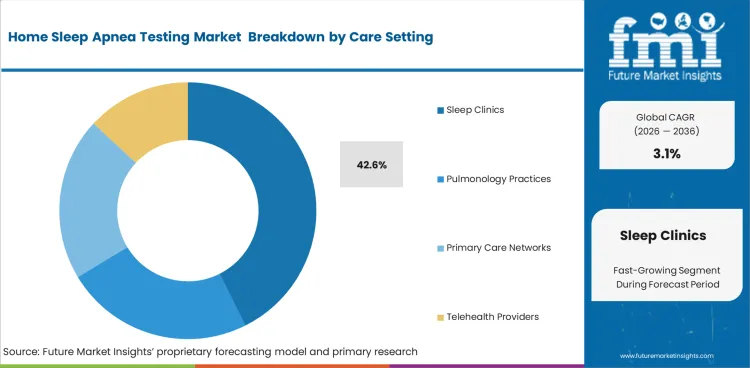
- Sleep clinics are poised to account for 42.6% share in 2026, reflecting their central position in referral intake and test interpretation.
- Organized diagnostic workflows support this segment as sleep clinics use home testing to relieve laboratory bottlenecks and accelerate early patient screening.
Insights into the Initial Risk Stratification Segment
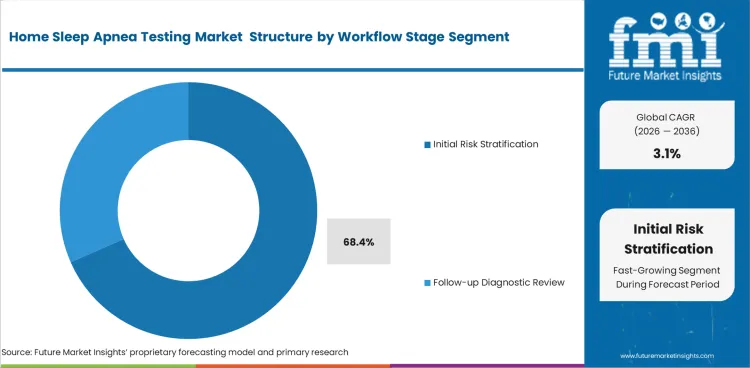
- Initial risk stratification is estimated to command 68.4% share in 2026, supported by primary use of home testing in first-pass clinical assessment.
- Revenue concentration remains in this workflow stage as comprehensive disorder evaluation usually takes place in staffed laboratories for selected patient groups.
Home Sleep Apnea Testing Market Drivers, Restraints, and Opportunities
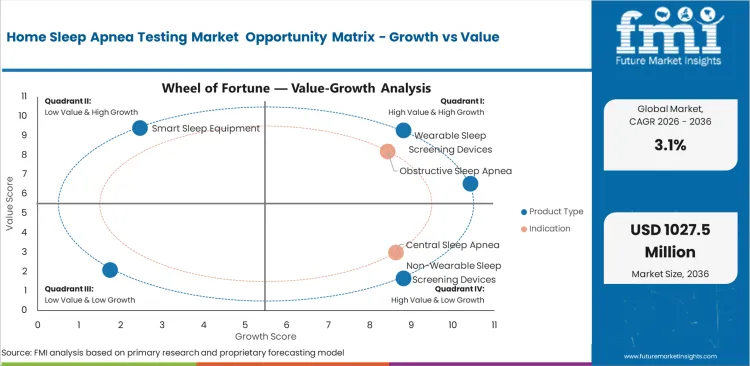
- Diagnostic access outside sleep laboratories is supporting demand for home testing across outpatient care and clinician-guided screening pathways.
- Limited physiological parameter capture and reimbursement scrutiny are restricting broader adoption across more complex diagnostic use cases.
- Digital review tools and telehealth workflows are opening fresh room for home testing across decentralized sleep care programs.
Home sleep apnea testing gains demand from healthcare systems seeking diagnostic access outside crowded laboratory environments. Portable testing supports timley evaluation for suspected breathing disorders and reduces pressure on overnight facility capacity. Product use is high across clinically selected obstructive sleep apnea cases with practical home testing suitability.
Diagnostic Access and Sleep Laboratory Relief
Sleep laboratories face capacity pressure across many care systems, which is supporting broader use of home-based testing pathways. Portable devices help providers start patient evaluation on time and reduce the burden associated with facility-based overnight studies. Primary care networks and sleep clinics use home testing to reduce delays across referral based screening programs. This driver keeps demand active across care environments seeking timley diagnostic throughput with controlled operating cost.
Limited Parameter Capture and Reimbursement Controls
Home testing devices capture fewer physiological signals than full polysomnography, which limits their role across more complex patient presentations. Physician caution stays high in cases needing detailed clinical evaluation or clear diagnostic certainty across mixed symptom profiles. Reimbursement control adds pressure to provider investment decisions and limits the patient pool suited for unattended testing. This restraint keeps adoption selective across established healthcare systems with laboratory infrastructure.
Analysis of Home Sleep Apnea Testing Market By Key Countries
.webp)
| Country | CAGR |
|---|---|
| India | 4.1% |
| China | 3.8% |
| UK | 2.6% |
| Spain | 1.9% |
| USA | 1.3% |
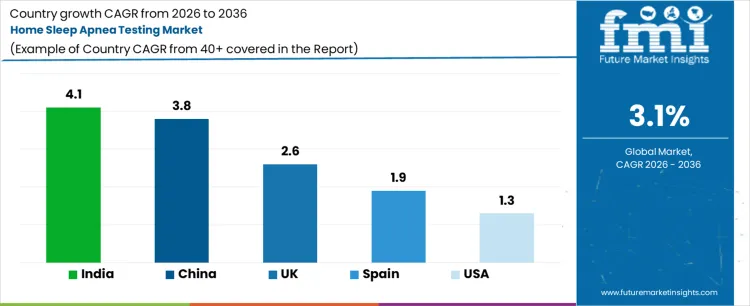
Analysis of Home Sleep Apnea Testing Market By Key Countries
- India leads the country outlook with a 4.1% CAGR, supported by rising diagnosis activity and limited accredited sleep laboratory access across major urban centers.
- China follows at 3.8% CAGR as outpatient diagnostic expansion and hospital triage use support a larger role for home-based testing pathways.
- United Kingdom records 2.6% CAGR and Spain posts 1.9%, reflecting selective public healthcare adoption and careful budget management across sleep diagnostic services.
- United States stands at 1.3% CAGR as mature sleep laboratory infrastructure and tighter reimbursement review limit faster expansion in home testing volumes.
Global home sleep apnea testing market is expected to expand at a 3.1% CAGR from 2026 to 2036, and the study covers over 40 countries with key markets listed below.
Demand Outlook for Home Sleep Apnea Testing Market in the United States
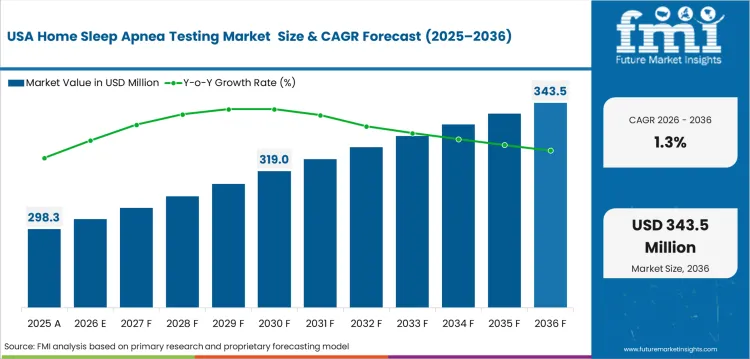
Home sleep apnea testing industry in the US is estimated to grow at a CAGR of 1.3% through 2036. Established diagnostic infrastructure and sleep laboratory access keep home testing focused on selected patient groups. Reimbursement review limits volume expansion across provider networks using payer-controlled diagnostic pathways. ResMed and Philips Respironics hold important positions across this national market.
- Managed care programs support demand for portable testing across cost-controlled diagnostic pathways.
- Replacement cycles carry a larger role across demand as first-time adoption stays limited.
- Clinician use centers on workflow efficiency and patient selection across established sleep care systems.
Future Outlook for Home Sleep Apnea Testing Market in the United Kingdom
The UK home sleep apnea testing domain is predicted to advance at a CAGR of 2.6% from 2026 to 2036. Public healthcare pathways favor evidence based diagnostic use and careful cost control across sleep services. Home testing is used to reduce wait periods. Centralized purchasing slows rollout across provider networks.
- National Health Service pathways support selective use across patients suited for portable assessment.
- Centralized procurement affects adoption speed across sleep clinics and related diagnostic programs.
- Specialist referral patterns keep patient selection narrow across many provider settings.
Opportunity Analysis of Home Sleep Apnea Testing Market in Spain
Spain home sleep apnea testing market is poised to grow at a CAGR of 1.9% over the forecast years. Hospital sleep units hold an important position across routine diagnostic practice in many regions. Budget control limits scale for portable testing, and regional variation affects deployment consistency across public health systems.
- Public healthcare budget discipline limits broader use across portable sleep diagnostic programs.
- Hospital-led workflows keep laboratory confirmation important across more complex patient cases.
- Regional variation affects uptake patterns across provider networks and local sleep service capacity.
In-depth Analysis of Home Sleep Apnea Testing Market in China
The home sleep apnea testing industry in China is estimated to advance at a CAGR of 3.8% during the forecast period. Outpatient diagnostic expansion and tertiary hospital triage use support a growing role for home testing. Digital health distribution and remote interpretation improve operating ease across urban care networks.
- Large patient volumes support early sorting through portable testing across major hospital systems.
- Hospital capacity pressure supports home screening use before more detailed in-lab evaluation.
- Urban diagnostic infrastructure improves clinician access to connected home testing platforms.
Sales Analysis of Home Sleep Apnea Testing Market in India
India home sleep apnea testing industry is projected to expand at a CAGR of 4.1% through 2036. Rising diagnosis activity and limited accredited sleep laboratory access support portable testing across this market. Private hospitals and diagnostic centers use home testing systems across cost-sensitive patient pathways in major cities.
- Limited sleep laboratory access supports portable testing across urban diagnostic networks.
- Cost-sensitive care pathways favor simpler home assessment across selected apnea screening programs.
- Physician awareness is stronger in major cities and supports organized referral-led adoption.
Competitive Landscape and Strategic Positioning
- Competitive positioning centers on device accuracy and review workflow quality across home diagnostics serving sleep clinics and outpatient providers.
- Supplier strength rises with dependable signal capture and secure data transfer, which support clinician confidence across unattended overnight testing.
- Market competition stays moderate as established brands combine portable hardware with software support for provider diagnostic use.
Competitive positioning in this market is built around signal reliability and patient comfort.
Secure clinician review also shapes competitive positioning across decentralized diagnostic environments. Product comparison often overlaps with remote patient monitoring devices and vital signs monitoring devices since oxygen saturation review and connected monitoring hold importance in supplier selection. Larger suppliers benefit from established distribution and stronger software-related interpretation tools across hospital and clinic accounts. Smaller participants can compete in focused diagnostic segments while broad platform integration and service continuity remain harder to match. This competitive pattern keeps scale and dependable workflow support closely connected with account retention.
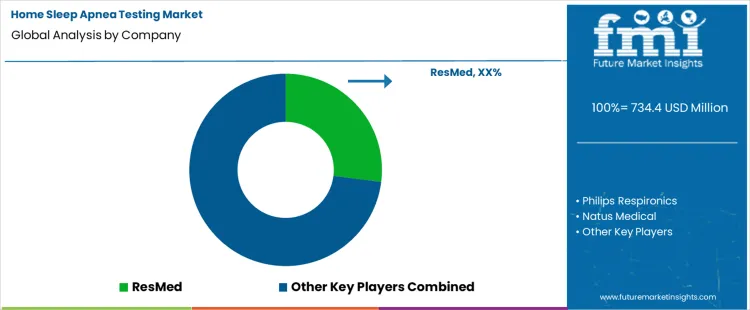
Home testing suppliers compete through portable device usability and through how smoothly results move into provider review systems. Use of home healthcare software and homecare medical devices is reinforcing interest in connected home diagnostics across care delivery models. ResMed holds a visible position through ApneaLink systems and remote monitoring support. Philips Respironics and Natus Medical remain relevant through portable hardware and clinical interpretation capability. Itamar Medical and Embla Systems remain relevant through portable hardware supported diagnostic interpretation. Market concentration is moderate since leading names hold recognition, and account choice continues to depend on workflow performance and clinician confidence.
Key Companies in the Home Sleep Apnea Testing Market
ResMed leads visible supplier positioning across home sleep testing with ApneaLink systems and broad remote monitoring support. Philips Respironics, Natus Medical, Itamar Medical, and Embla Systems hold active positions across portable hardware and clinic review workflows.
Competitive Benchmarking: Home Sleep Apnea Testing Market
| Company | Portable Test Device Breadth | Clinician Review Platform Depth | Sleep Clinic Access | Geographic Footprint |
|---|---|---|---|---|
| ResMed | High | Strong | Strong | Global |
| Philips Respironics | High | Strong | Strong | Global |
| Natus Medical | Medium | Strong | Moderate | Global |
| Itamar Medical | Medium | High | Moderate | North America, Europe |
| Embla Systems | Medium | Medium | Moderate | Europe, North America |
Source: Future Market Insights, 2026.
Key Developments in Home Sleep Apnea Testing Market
- In April 2025 Resmed announced USA-wide availability of NightOwl as an FDA-cleared home sleep apnea test supporting at-home obstructive sleep apnea diagnosis for patients nationwide.
- In August 2024 Huxley Medical received FDA 510(k) clearance for SANSA as a chest-worn diagnostic patch for adult sleep-related breathing disorder evaluation and suspected sleep apnea.
Key Players in the Home Sleep Apnea Testing Market
Major Global Players
- ResMed
- Philips Respironics
- Natus Medical
- Itamar Medical
- Embla Systems
Report Scope and Coverage
| Parameter | Details |
|---|---|
| Quantitative Units | USD 734.4 million to USD 996.6 million, at a CAGR of 3.1% |
| Market Definition | The home sleep apnea testing market encompasses portable diagnostic devices and related home-based testing workflows used for suspected sleep-disordered breathing, including wearable sleep screening devices, non-wearable sleep screening devices, smart sleep equipment, and home polysomnography PSG devices used across clinician-reviewed diagnostic pathways. |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, China, UK, Spain, USA, and 40+ countries |
| Key Companies Profiled | ResMed, Philips Respironics, Natus Medical, Itamar Medical, Embla Systems |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down methodology starting with home-based diagnostic demand patterns, estimating product and channel adoption across portable sleep testing formats, and projecting regional uptake across clinician-guided sleep disorder screening pathways. |
Source: Future Market Insights, 2026.
Home Sleep Apnea Testing Market by Segment
Home Sleep Apnea Testing Market Segmented by Product Type:
- Wearable Sleep Screening Devices
- Non-Wearable Sleep Screening Devices
- Smart Sleep Equipment
- Home Polysomnography PSG Devices
- Others
Home Sleep Apnea Testing Market Segmented by Indication:
- Obstructive Sleep Apnea
- Central Sleep Apnea
Home Sleep Apnea Testing Market Segmented by Distribution Channel:
- Offline Sales
- Online Sales
Home Sleep Apnea Testing Market Segmented by Care Setting:
- Sleep Clinics
- Pulmonology Practices
- Primary Care Networks
- Telehealth Providers
Home Sleep Apnea Testing Market Segmented by Workflow Stage:
- Initial Risk Stratification
- Follow-up Diagnostic Review
Home Sleep Apnea Testing Market by Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Research Sources and Bibliography
- Resmed. (2025, April 3). Resmed announces small, easy to use home sleep apnea test NightOwl now available across the USA
- Huxley Medical, Inc. (2024, August 7). USA Food and Drug Administration gives Huxley Medical 510(k) clearance for SANSA Home Sleep Apnea Test.
- Hussein, O., Alkhader, A., Gohar, A., & Bhat, A. (2024). Home sleep apnea testing for obstructive sleep apnea. Missouri Medicine, 121(1), 60–65.
- Go, B. C., & Thaler, E. R. (2024, June). Home sleep testing versus traditional polysomnography: Pros and cons. Otolaryngologic Clinics of North America, 57(3), 363–369.
- Ellender, C. M., Ruehland, W. R., Duce, B., Joyce, R., Worsnop, C., Mercer, J., Naughton, M., Hukins, C. A., Wheatley, J., & Cunnington, D. (2024, May 9). Australasian Sleep Association 2024 guidelines for sleep studies in adults. Sleep, 47(10), zsae107.
-
This bibliography is provided for reader reference.
This Report Answers
- What is the calculated 2025 market size for home sleep apnea testing based on the stated 2026 base value and forecast CAGR?
- How large can home sleep apnea testing market become by 2036 under the stated forecast path?
- How fast is home sleep apnea testing market expected to expand from 2026 to 2036?
- Which product type is expected to lead demand by 2026 across portable home diagnostic use?
- Which indication holds the largest share in 2026 across home sleep apnea testing demand?
- What factors are expanding diagnostic use outside traditional sleep laboratories across major healthcare systems?
- Which countries are expected to record the fastest pace through 2036 across named national markets?
- Which companies hold visible positions across portable testing hardware and connected clinician review workflows?
- How is home sleep apnea testing market defined within the diagnostic services and portable device landscape?
- How is the market forecast estimated and checked across values, segments, and country patterns?
Frequently Asked Questions
What is the global market demand for Home Sleep Apnea Testing in 2026?
In 2026, the global market for home sleep apnea testing is expected to be worth USD 734.4 million.
How big will the market for Home Sleep Apnea Testing be in 2036?
By 2036, the market for home sleep apnea testing is expected to be worth USD 996.6 million.
How much is demand for Home Sleep Apnea Testing expected to expand between 2026 and 2036?
Between 2026 and 2036, the demand for home sleep apnea testing is expected to expand at a 3.1% CAGR.
Which indication segment is likely to lead the market by 2026?
Obstructive sleep apnea is expected to account for 95.7% share in 2026 across the global home sleep apnea testing market.
Which product type is expected to lead the market by 2026?
Wearable sleep screening devices are projected to hold 66.1% share in 2026 across global product demand.
What is driving market expansion in China?
China is estimated to expand at a 3.8% CAGR through 2036 across outpatient diagnostic expansion and hospital triage use.
What does Home Sleep Apnea Testing Market mean in this report?
Home sleep apnea testing includes portable diagnostic devices and related workflows used outside sleep laboratories for suspected sleep-disordered breathing.
How does FMI estimate and validate the Home Sleep Apnea Testing forecast?
Forecasting uses the stated market values, the 2026 to 2036 CAGR, and cross-checking across segment structure and country demand patterns.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Wearable Sleep Screening Devices
- Non-Wearable Sleep Screening Devices
- Smart Sleep Equipment
- Home Polysomnography PSG Devices
- Others
- Wearable Sleep Screening Devices
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Indication
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Indication, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Indication, 2026 to 2036
- Obstructive Sleep Apnea
- Central Sleep Apnea
- Obstructive Sleep Apnea
- Y to o to Y Growth Trend Analysis By Indication, 2021 to 2025
- Absolute $ Opportunity Analysis By Indication, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Offline Sales
- Online Sales
- Offline Sales
- Y to o to Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Care Setting
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Care Setting, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Care Setting, 2026 to 2036
- Sleep Clinics
- Pulmonology Practices
- Primary Care Networks
- Telehealth Providers
- Sleep Clinics
- Y to o to Y Growth Trend Analysis By Care Setting, 2021 to 2025
- Absolute $ Opportunity Analysis By Care Setting, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Workflow Stage
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Workflow Stage, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Workflow Stage, 2026 to 2036
- Initial Risk Stratification
- Follow-up Diagnostic Review
- Initial Risk Stratification
- Y to o to Y Growth Trend Analysis By Workflow Stage, 2021 to 2025
- Absolute $ Opportunity Analysis By Workflow Stage, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Indication
- By Distribution Channel
- By Care Setting
- By Workflow Stage
- Competition Analysis
- Competition Deep Dive
- ResMed
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Philips Respironics
- Natus Medical
- Itamar Medical
- Embla Systems
- ResMed
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Care Setting, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Care Setting, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Care Setting, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Care Setting, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Care Setting, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Care Setting, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Care Setting, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Care Setting, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Indication
- Figure 9: Global Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Distribution Channel
- Figure 12: Global Market Value Share and BPS Analysis by Care Setting, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Care Setting, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Care Setting
- Figure 15: Global Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Workflow Stage
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Product Type
- Figure 32: North America Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Indication
- Figure 35: North America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Distribution Channel
- Figure 38: North America Market Value Share and BPS Analysis by Care Setting, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Care Setting, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Care Setting
- Figure 41: North America Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Workflow Stage
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Product Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Indication
- Figure 51: Latin America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Distribution Channel
- Figure 54: Latin America Market Value Share and BPS Analysis by Care Setting, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Care Setting, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Care Setting
- Figure 57: Latin America Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Workflow Stage
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Product Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Indication
- Figure 67: Western Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Distribution Channel
- Figure 70: Western Europe Market Value Share and BPS Analysis by Care Setting, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Care Setting, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Care Setting
- Figure 73: Western Europe Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Workflow Stage
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Indication
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Distribution Channel
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Care Setting, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Care Setting, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Care Setting
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Workflow Stage
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Product Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Indication
- Figure 99: East Asia Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Distribution Channel
- Figure 102: East Asia Market Value Share and BPS Analysis by Care Setting, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Care Setting, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by Care Setting
- Figure 105: East Asia Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Workflow Stage
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Indication
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Distribution Channel
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Care Setting, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Care Setting, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Care Setting
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Workflow Stage
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Indication
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Distribution Channel
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Care Setting, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Care Setting, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Care Setting
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Workflow Stage
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

