Temporary Pacing Leads Market
The Temporary Pacing Leads Market is Segmented by Product (Cardiac Pacing Leads/Catheter, Cardiac Pacing Wires), Application (Transcatheter Aortic Valve Replacement, Cardiothoracic Surgeries, Acute Myocardial Infarction, Bradycardia, Heart Blocks, Sick Sinus Syndrome, Congestive Heart Failure), Age Group (Adult, Pediatric), Technique (Epicardial Pacing, Transvenous Pacing, Transcutaneous Pacing), End User (Hospitals, Independent Catheterization Labs), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Temporary Pacing Leads Market Size, Market Forecast and Outlook By FMI
The temporary pacing leads market was valued at USD 379.03 million in 2025 and is expected to expand from USD 396.09 million in 2026 to USD 558.73 million by 2036. The market is anticipated to register a 3.5% CAGR during the forecast period. Cardiac pacing leads/catheter are likely to lead product demand with a 95.3% share, while transcatheter aortic valve replacement is projected to account for 24.6% share of the application segment in 2026.
Summary of the Temporary Pacing Leads Market
- Demand and Growth Drivers
- Transcatheter aortic valve replacement procedure growth is expected to support recurring demand for temporary pacing leads.
- Cardiac catheterization laboratory expansion is likely to improve access to complex cardiac interventions in emerging regions.
- Reimbursement support is anticipated to strengthen adoption across hospitals and independent catheterization labs.
- Product and Segment View
- Cardiac pacing leads/catheter are expected to lead the product segment due to broad use across cardiac procedure settings.
- Transcatheter aortic valve replacement is likely to remain the leading application because procedures need temporary ventricular pacing support.
- Adult patients are anticipated to account for stronger use as age-linked cardiac rhythm disorders remain more common.
- Geography and Competitive Outlook
- India is expected to lead country-level growth due to catheterization lab expansion and wider cardiac procedure reimbursement.
- China is likely to record strong expansion as reimbursement access for cardiac intervention consumables improves.
- Companies with stronger regulatory clearance, hospital supply agreements and clinical support are anticipated to improve procurement positions.
- Analyst Opinion
- Sabyasachi Ghosh, Principal Consultant for Healthcare at FMI, suggests, “The temporary pacing leads market is likely to move toward certified lead configurations that support procedural reliability and safer cardiac rhythm management. Suppliers with validated products and stronger hospital contracting support are expected to gain advantage as cardiac intervention volumes increase.”
- Temporary Pacing Leads Market Value Analysis
- The temporary pacing leads market is moving from emergency rhythm support into a procedure-linked consumable category.
- Demand is likely to rise as TAVR and cardiothoracic surgeries require reliable temporary pacing support.
- Adoption is expected to improve through cardiac pacing leads/catheter used across acute and interventional cardiac care.
- Spending is anticipated to be supported by cardiac procedure expansion and stronger reimbursement for structural heart interventions.
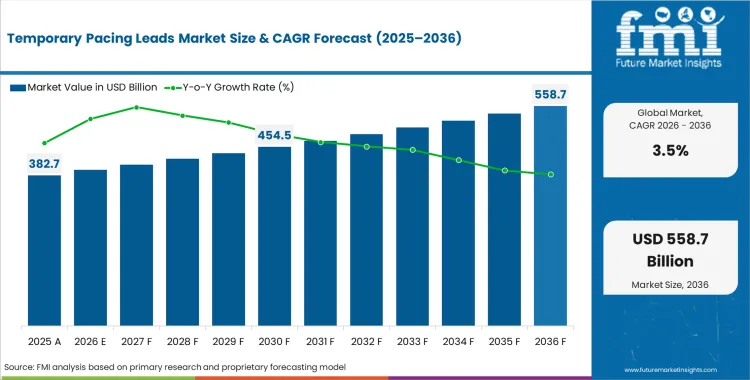
Temporary Pacing Leads Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 396.09 Million |
| Industry Value (2036) | USD 558.73 Million |
| CAGR (2026-2036) | 3.5% |
| Source | Future Market Insights, 2026 |
Source: Future Market Insights, 2026
The incremental opportunity of USD 162.64 million over the forecast decade reflects the global expansion of structural heart disease intervention programmes driven by ageing population demographics increasing the prevalence of aortic stenosis and conduction system disorders requiring temporary cardiac pacing support. Bipolar temporary pacing lead configurations hold the dominant product share as bipolar sensing and pacing capability reduces the risk of far-field sensing artefacts in post-operative cardiac monitoring environments. Manufacturers that achieve ISO 13485 quality management system certification and CE Mark or FDA 510(k) clearance for bipolar and quadripolar temporary pacing lead configurations are capturing multi-year hospital group supply agreements that provide predictable volume commitments aligned with cardiac catheterisation laboratory procedure plan targets.
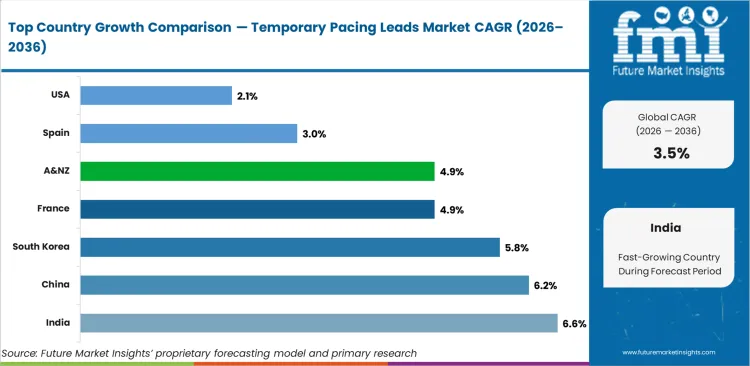
India leads at a 6.6% CAGR through 2036, driven by rapid cardiac catheterisation laboratory expansion under PMJAY scheme reimbursement for structural heart procedures and the ongoing commissioning of tertiary cardiac centres in tier-2 cities. China advances at 6.2%, supported by National Healthcare Security Administration reimbursement expansion for TAVI procedures at designated cardiac surgery centres. South Korea registers 5.8% growth driven by National Health Insurance coverage expansion for complex electrophysiology procedures. France at 4.9% and Australia and New Zealand at 4.9% reflect established cardiac programme growth, while Spain at 3.0% and the USA at 2.1% represent mature markets with stable per-procedure consumption underpinned by large installed cardiac procedure volumes.
Temporary Pacing Leads Market Definition
The temporary pacing leads market encompasses single-use electrode catheters and pacing wires designed for transient electrical stimulation of the cardiac myocardium during acute cardiac events, cardiothoracic surgical procedures, and interventional cardiology interventions including transcatheter valve replacement. The market covers temporary unipolar, bipolar, and quadripolar pacing lead and catheter configurations, floating balloon-tipped pacing catheters, and epicardial pacing wires placed intraoperatively during cardiac surgery for post-operative rhythm management.
Temporary Pacing Leads Market Inclusions
The scope covers global and regional revenue from single-use temporary pacing lead catheters, epicardial pacing wire sets, and associated introducer sheath and connection cable accessories. Forecast includes all pacing configuration types across TAVI, cardiac surgery, acute myocardial infarction, bradycardia, heart block, sick sinus syndrome, and congestive heart failure application segments through 2036.
Temporary Pacing Leads Market Exclusions
The scope excludes permanent implantable cardiac pacemaker leads used for long-term pacing therapy, implantable cardioverter defibrillator leads, cardiac resynchronisation therapy device leads, and leadless pacemaker systems. External temporary pacemaker pulse generator units are also excluded from the lead and catheter market valuation.
Temporary Pacing Leads Market Research Methodology
Primary Research
Analysts engaged with procurement leads, quality managers, and technical specialists across the temporary pacing leads value chain to identify adoption triggers, specification requirements, and contract structures governing purchasing decisions.
Desk Research
Data collection phases aggregated regulatory publications, government programme announcements, standards body guidelines, company investor disclosures, and trade association reports to establish the policy and commercial context framing market demand.
Market-Sizing and Forecasting
Baseline values derive from bottom-up aggregation of segment-level deployment volumes calibrated against publicly disclosed procurement and shipment data, with region-specific adoption velocity curves applied to project demand through 2036.
Data Validation and Update Cycle
Projections are cross-validated against quarterly publicly reported revenue guidance from leading market participants and retested against regulatory milestones that structurally alter adoption timelines within each geographic segment.
Key Industry Highlights
Growing Number of New Product Launches as well as Improved Reimbursement Policies the Market for Temporary Pacing Leads is Continue to Grow in Upcoming Years
The reimbursement policies properly address the TPL-related costs and the procedure costs, they are more willing to carry out the implementation without financial worries. This increases application in many clinical environments such as ICUs, cardiac catheterization laboratories, and operating theaters where there is a great role of TPLs in the management of acute cardiac rhythm disturbances after surgery or during critical care.
Apart from it favorable reimbursement policies make it encouraging for the healthcare professional to consider TPLs as a standard of care in those who are going to need temporary cardiac pacing. This acceptance of TPLs is important, especially in emergent situations like post-heart surgeries or those with serious arrhythmias, where timely interventions with TPLs will save lives.
The continuous innovation and competitive pressures in cardiac care have raised the competition graph to introduce competitively priced products into the market. The competitive climate, in turn, will motivate manufacturers to invest in research and development, further driving technological advancement and improving overall quality.
Also most of the manufacturers, in a bid to meet the changing clinical needs and preferences of healthcare, have their focus on launching new products in the market, offering health providers an array of options that best suit the varied population.
For instance, in January 2021, Teleflex Incorporated, a global provider of medical technologies, received USA FDA clearance for its WattsonTM temporary pacing guidewire. This bipolar temporary pacing guidewire is specifically designed for use during transcatheter aortic valve replacement (TAVR) and balloon aortic valvuloplasty (BAV).
Hence the market for owing to the favorable reimbursement policies as well as new product launches the temporary pacing leads sales is anticipated.
Expanding Use in Complex Cardiac Procedures is Among the Key Trend in Temporary Pacing Leads Market
In recent year the temporary pacing leads being increasingly used in these complex and specialized cardiac procedures, such as structural heart interventions and electrophysiology studies.
In structural heart interventions, the procedures include, transcatheter aortic valve replacements and mitral valve repairs, temporary pacing leads are playing important role during and immediately following the procedure in order to manage the rhythms of the heart.
These leads are responsible for maintaining the heart rate and rhythm steady, offsetting the disturbing effect brought about by the intervention.
Similarly, in electrophysiology studies that diagnose and treat arrhythmias, temporary pacing leads allow for precise mapping and manipulative maneuvers of electrical pathways of the heart. They provide necessary control over heart rate and rhythm, enabling cardiologists to perform intricate diagnostic and therapeutic maneuvers safely.
This can be attributed to the ever-expanding use of temporary pacing leads, primarily based on key trends in market growth. With the continuously rising prevalence of complex cardiac conditions and greater adoption of advanced minimally invasive procedures, demands for continuous and reliable pacing solutions continue to rise.
Finally, technological advances have enhanced functionality and usability in temporary pacing leads, making them even more appealing for healthcare professionals. The trend broadens the market scope, underlining the role of temporary pacing leads as indispensable to contemporary cardiac care, consequently fostering deeper market penetration and continued growth.
The Complexities Associated with Insertion of Temporary Pacing Leads Could Lower its Adoption Rate
Temporary pacing leads (TPLs) are important tool for managing acute cardiac rhythm disturbances but pose procedural complexities. Inserting the leads requires precise placement within or near the heart, demanding skilled practitioners to navigate anatomical variations and ensure effective pacing.
Risks associated with TPLs include vascular injury, cardiac perforation, and pneumothorax, particularly with subclavian vein access. These risks necessitate meticulous technique, real-time imaging, and vigilant monitoring to mitigate complications.
Furthermore, serious complications such as cardiac perforation (up to 4%) and dislodgment (10% to 37%) with loss of pace capture can occur. These intraprocedural risks significantly impact post-procedural use, confining patients to bed rest and contributing to costly lengths of stay (LOS) in the intensive care unit.
The presence of electromagnetic interference (EMI) from nearby medical equipment can disrupt pacing signals, requiring shielding techniques and careful device management. Moreover, the placement of pacing leads carries a potential risk of infection, necessitating strict aseptic practices and post-procedure surveillance.
Therefore, while TPLs are essential for managing acute cardiac conditions, their use during procedures involves technical challenges and risks of complications. This complexity can create hesitation among healthcare providers regarding adoption and may limit their market growth.
Emphasis on Strategic Acquisitions for Expanding Geographical Presence is Creating Opportunities in the Temporary Pacing Leads Market
This expansion in new emerging markets opens up growth within healthcare infrastructures and thereby boosts the demand for advanced medical technologies. Such a strategic expansion opens one's services to an expanded end user base, including health facilities and providers that require temporary pacing solutions to assist with the management of cardiac conditions.
Geographical expansion reduces dependence on particular markets, disperses sources of revenue, and reduce risk because of economic fluctuation or changes in regulatory policies in established regions.
Besides, geographic expansion by a manufacturer allows the company to use regional differences in health needs, preferences, and regulatory environments to the full advantage of developing and marketing product offerings that are tailored to specific needs.
With a local presence, the market is more penetrated because access is improved, customer service at the local level will more readily be available, and responsiveness to changes in the market will be accentuated.
The company has closer relationships with health professionals and with distribution partners-developing trust and loyalty. In this manner, strategic partnerships with local distributors and healthcare networks improve market access and enable more efficient distribution channels.
For instance, in February 2023 Abbott signed a definitive agreement for the acquisition of Cardiovascular Systems, Inc., a medical device company specializing in innovative atherectomy systems used in the treatment of peripheral and coronary artery disease.
Hence such expansion of key players in untapped market is creating opportunities of temporary pacing leads market growth.
2021 to 2025 Global Temporary Pacing Leads Sales Outlook Compared to Demand Forecast from 2026 to 2036
Historically, temporary pacing leads were first utilized in the management of bradycardia and heart block, particularly during emergencies or in a perioperative care context. These served as important devices that helped in the stabilization of patients' heart rhythms when their natural peacemaking was compromised and acted as a bridge to more permanent measures, such as the implantation of a permanent pacemaker.
The indications of such leads were initially limited to these few emergency and perioperative scenarios. However, with the evolution of cardiac care, their indications expanded considerably. With the advancement of cardiac surgery, especially minimally invasive surgery and electrophysiology studies, the indications of temporary pacing leads went beyond conventional ones.
The future opportunity looks much promising for temporary pacing leads, owing to an uptick in cardiovascular diseases, aging of the population, and complex cardiac interventions. The growth prospects could be further helped by increased adoption for specialized procedures including structural heart interventions and electrophysiology applications.
Further, continuous development with respect to the design of leads and their integration with digital health will go further in opening new pathways for market growth; it is here that temporary pacing leads could assume a very important role as part of modern cardiac care.
Market Concentration
Tier 1 companies comprise market leaders with a market revenue of above USD 209.98 million capturing significant market share of 58.4% in global market. These companies are targeting further market exposure through new geographical regions and diversified product offerings.
This includes targeting emerging markets within Asia-Pacific that are recording growth in disposable incomes and improving healthcare infrastructure. Prominent companies in tier 1 include Medtronic plc, Abbott and BD.
Tier 2 companies include mid-size players with revenue of USD 50 to 100 million having presence in specific regions and highly influencing the local market and holds around 25.0% market share. They typically pursue partnerships with academic institutions and research organizations to leverage emerging technologies and expedite product development.
These companies often emphasize agility and adaptability, allowing them to quickly bring new treatments to market, sometimes targeting specific types of cancer or rare conditions. Additionally, they focus on cost-effective production methods to offer competitive pricing. Prominent companies in tier 2 include B. Braun SE, Zimmer Biomet and Teleflex Incorporated.
Finally, Tier 3 companies, such as Oscor Inc, BioTrace Medical (Merit Medical Systems Inc.), Edwards Lifesciences Services GmbH and OSYPKA MEDICAL. They specialize in specific products and cater to niche markets, adding diversity to the industry.
Overall, while Tier 1 companies are the primary drivers of the market, Tier 2 and 3 companies also make significant contributions, ensuring the temporary pacing leads sales remains dynamic and competitive.
Country-wise Insights
The section below covers the industry analysis for the temporary pacing leads market for different countries. Market demand analysis on key countries in several regions of the globe, including North America, Asia Pacific, Europe, and others, is provided.
The United States is anticipated to remain at the forefront in North America, with higher market share through 2034. In South Asia Pacific, India is projected to witness a CAGR of 6.6% by 2034.
.webp)
| Countries | Value CAGR (2026 to 2036) |
|---|---|
| USA | 2.1% |
| Germany | 1.9% |
| China | 6.2% |
| France | 4.9% |
| India | 6.6% |
| Spain | 3.0% |
| Australia & New Zealand | 4.9% |
| South Korea | 5.8% |
Increasing Number of Cardiovascular Disease Cases in the United States
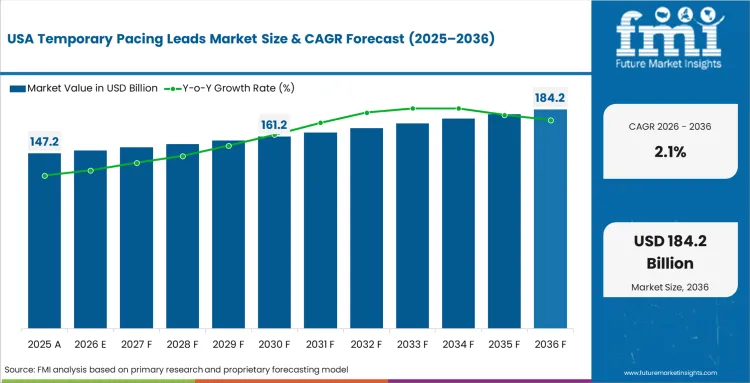
USA temporary pacing leads market is poised to exhibit a CAGR of 2.1% between 2026 and 2036. Currently, USA holds lion’s share in the North American Temporary Pacing Leads market, and the trend is expected to continue during the forecast period.
Increasing prevalence of cardiovascular disorders, rising number of cardiac procedures and demand for temporary pacing leads in market in the USA is developing mainly owing to the increase in the prevalence of cardiovascular diseases. Heart disease continues to be the major cause of mortality in the country, which fosters the demand for advanced cardiac care solutions even more.
A large population aged above 65 years and a high incidence of various conditions like bradycardia and arrhythmias are some of the major reasons contributing to this trend. Moreover, the strong healthcare infrastructure and awareness about treatment in USA promotes the adoption of the such medical technologies, which further accelerates the growth of the market for easy availability and usage of temporary pacing leads.
China's Strategic Emphasis on developing new Pacing Leads has Led to Increased Investment in Research and Development in Cardiovascular Procedures
China’s temporary pacing leads market is poised to exhibit a CAGR of 6.2% between 2026 and 2036. Currently, it holds the highest share in the East Asia market, and the trend is expected to continue during the forecast period.
The China’s healthcare sector is developing owing to higher investment by the government in the country's, are contributing factors to the continuous growth of China's temporary pacing lead market. The key reason is that cardiac care services have been emphasized in general healthcare reforms undertaken by the Chinese government.
The National Health Commission of China announced that the country would continue to promote access to high-value medical devices, such as temporary pacing leads, in both urban and rural areas. As such, the move is driving adoption, especially as more hospitals are acquiring the necessary technologies to conduct complex cardiac procedures.
The Increasing Awareness and Early Diagnosis of Cardiovascular Conditions Propelling the Temporary Pacing Leads Market in India
India’s temporary pacing leads market is poised to exhibit a CAGR of 6.6% between 2026 and 2036. Currently, it holds the highest share in the South Asia & Pacific market, and the trend is expected to continue during the forecast period.
This growing awareness of cardiovascular conditions and early diagnoses is driving growth within the temporary pacing leads market in India. The public health campaigns and the efforts by organizations such as the Indian Heart Association have brought much awareness about heart diseases, thus leading to timely interventions.
For instance, in September 2022, the Hindu’s Future India Club, in association with the city’s Andhra Hospital has organized awareness programme on the occasion of World Heart Day in Vijayawada, Andhra Pradesh.
It can also be attributed to a growing middle-class population that enjoys better access to health care. Because of early diagnosis and treatment, more patients require temporary pacing lead, so the demand is getting higher, especially in towns and cities where health care facilities are getting better.
Category-wise Insights
Owing to having Wide Application and Advanced Technology available in Cardiac Pacing Leads/Catheters has Higher Adoption Rate.
The section contains information about the leading segments in the industry. By product, the cardiac pacing leads/catheter segment holds the highest market share of 95.7% in 2025
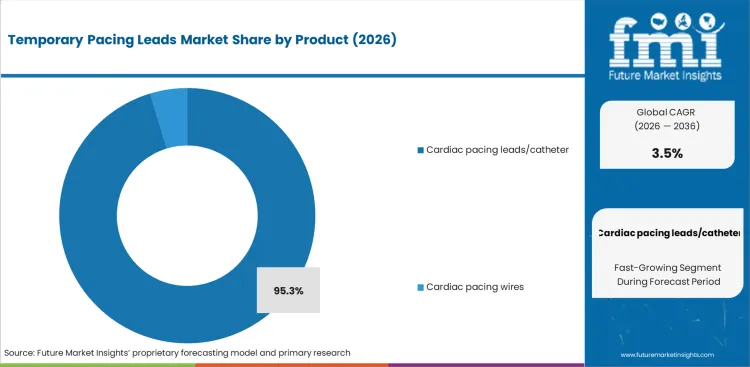
| By Product | Cardiac Pacing Leads/Catheter |
|---|---|
| Value Share (2025) | 95.3% |
The wide application and advanced technology available in cardiac pacing leads/catheters has hold significant market share of 95.3% in 2026. Cardiac pacing leads/catheters have a wider range of application from temporary to permanent pacing in all cardiac procedural settings.
Their unique design also allows for precise placement and reliable performance, essential in the management of arrhythmias and heart block. Cardiac pacing wires, on the other hand, are usually indicated in more specific, limited scenarios, including short-term pacing after cardiac surgery. The versatility, along with reliability and advanced features bestowed on leads/catheters, propels their dominance in the market.
TAVR offers a Minimally Invasive Alternative to Open-heart Surgery Hence Increasing Preference from Healthcare Experts
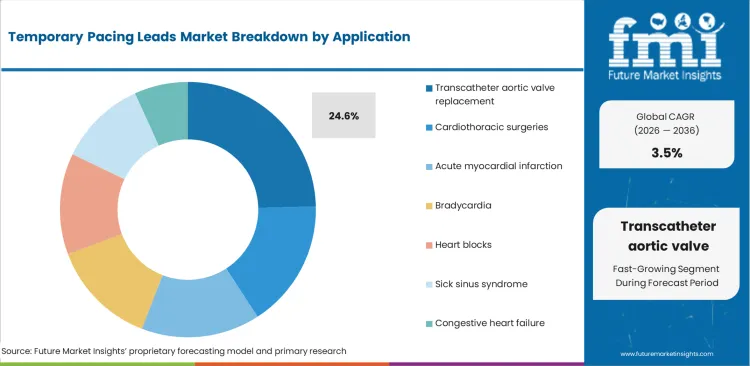
| By Formulation | Transcatheter aortic valve replacement (TAVR) |
|---|---|
| Value Share (2025) | 24.6% |
The segment of TAVR holds a share of 24.6% compared with other applications, due to the increased adoption of minimally invasive procedures for the treatment of severe aortic stenosis. TAVR offers much less invasiveness than an open-heart surgical option, especially in a high-risk patient such as an elderly or any comorbid patient.
Performances from TAVR procedures are increasing with the growth in the aging population and its increasing prevalence rate of aortic stenosis. Success with TAVR and lower recovery times, along with rapid advances in transcatheter technologies, make the application preferred over cardiothoracic surgeries or treatments for acute myocardial infarction.
Competitive Landscape
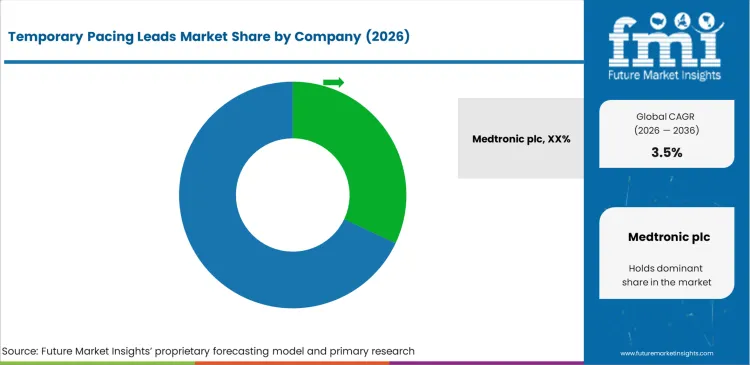
The market players are using strategies to stay competitive, such as product differentiation through innovative formulations, strategic partnerships with healthcare providers for distribution. Another key strategic focus of these companies is to actively look for strategic partners to bolster their product portfolios and expand their global market presence.
Recent Industry Developments in Temporary Pacing Leads Market
- In October 2022, Medtronic received approval for the expanded label of cardiac lead. These cardiac lead taps into the heart's natural electrical system and provides patients with needed therapy.
- In October 2022, Merit Medical Systems, Inc., acquired Tempo Lead from BioTrace Medical and expanded its product portfolio in the market.
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 396.09 Million to USD 558.73 Million, at a CAGR of 3.5% |
| Market Definition | The temporary pacing leads market encompasses single-use electrode catheters and pacing wires designed for transient electrical stimulation of the cardiac myocardium during acute cardiac events, cardiothoracic surgical p... |
| Product Segmentation | Cardiac Pacing Leads/Catheter, Cardiac Pacing Wires |
| Application Segmentation | Transcatheter Aortic Valve Replacement, Cardiothoracic Surgeries, Acute Myocardial Infarction, Bradycardia, Heart Blocks, Sick Sinus Syndrome, Congestive Heart Failure |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | India, China, USA, Germany, France, UK, Japan, Brazil, and 40 plus countries |
| Key Companies Profiled | Medtronic plc, BioTrace Medical (Merit Medical Systems Inc.), B. Braun SE, Oscor Inc., OSYPKA MEDICAL, BD, Abbott, APC Cardiovascular Ltd. |
| Forecast Period | 2026 to 2036 |
| Approach | Bottom-up methodology applied to segment-level deployment data, cross-validated against publicly reported manufacturer and operator revenue disclosures |
Key Segments of Temporary Pacing Leads Market
By Product:
In terms of product, the industry is divided into-cardiac pacing leads/catheter (temporary unipolar pacing leads, temporary bipolar pacing leads [bipolar pacing leads with flexible ends] bipolar pacing leads with balloons, temporary quadripolar pacing leads) and cardiac pacing wires (uni-polar temporary pacing wires, bi-polar temporary pacing wires, quadri-polar temporary pacing wires).
By Application:
In terms of application, the industry is segregated into cardiothoracic surgeries, acute myocardial infarction, bradycardia, heart blocks, sick sinus syndrome, congestive heart failure (CHF), and transcatheter aortic valve replacement (TAVR).
By Age Group:
In terms of age group, the industry is segregated into adult and pediatric.
By Technique:
In terms of technique, the industry is segregated into epicardial pacing transvenous pacing and transcutaneous pacing.
By End User:
In terms of end user, the industry is segregated into hospitals (less than 100 bed hospitals, 100-200 bed hospitals, more than 200 beds hospitals) and independent catheterization labs.
By Region:
Key countries of North America, Latin America, Western Europe, Eastern, South Asia & Pacific, East Asia and Middle East and Africa (MEA) have been covered in the report.
Bibliography
- World Health Organization. (2025, May). Global atlas on cardiovascular disease prevention and control: Cardiac intervention capacity data. WHO. https://www.who.int/publications/i/item/9789241564373
- Centers for Medicare and Medicaid Services. (2025, September). Transcatheter aortic valve replacement: National coverage determination and procedure volume data. CMS. https://www.cms.gov/medicare-coverage-database
- European Society of Cardiology. (2025, August). ESC guidelines on the management of valvular heart disease: TAVI procedure standards. ESC. https://www.escardio.org/Guidelines
- National Health Service England. (2025, June). National Cardiac Audit Programme: Structural heart intervention procedure volume report 2023-24. NHS England. https://www.nicor.org.uk
- Ministry of Health and Family Welfare, Government of India. (2025, March). Ayushman Bharat PMJAY: Cardiac procedure reimbursement coverage expansion update. MoHFW. https://pmjay.gov.in
- National Healthcare Security Administration, People's Republic of China. (2025, July). Medical device reimbursement catalogue update: Cardiac intervention consumables. NHSA. https://www.nhsa.gov.cn
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with primary research documentation.
Frequently Asked Questions
How large is the Temporary Pacing Leads Market in 2026?
The temporary pacing leads is estimated to be valued at USD 396.09 Million in 2026.
What will the Temporary Pacing Leads Market size be by 2036?
The market is projected to reach USD 558.73 Million by 2036.
What is the expected CAGR for the Temporary Pacing Leads Market between 2026 and 2036?
The market is expected to grow at a CAGR of 3.5% between 2026 and 2036.
Which product category leads the Temporary Pacing Leads Market?
Cardiac Pacing Leads/Catheter accounts for 95.3% in 2026 and leads due to its broad deployment across primary end-use applications and established procurement frameworks.
Which country records the fastest growth in the Temporary Pacing Leads Market?
India leads with a 6.6% CAGR through 2036, driven by expanding cardiac catheterisation laboratory network under Ayushman Bharat and PMJAY scheme coverage of structural heart procedure reimbursement.
What is driving demand in China?
China is advancing at 6.2% CAGR, supported by National Reimbursement Drug List and medical device reimbursement expansion covering TAVI procedures across designated cardiac centres.
Who are the key players in the Temporary Pacing Leads Market?
Key companies include Medtronic plc, BioTrace Medical (Merit Medical Systems Inc.), B. Braun SE, Oscor Inc., OSYPKA MEDICAL, and others detailed in the competitive landscape section.
What incremental opportunity does the Temporary Pacing Leads Market offer through 2036?
FMI analysts project an incremental market opportunity of USD 162.64 Million over the 2026 to 2036 forecast period.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product , 2026 to 2036
- Cardiac pacing leads/catheter
- temporary unipolar pacing leads
- temporary bipolar pacing leads
- _bipolar pacing leads with flexible ends
- bipolar pacing leads with balloons
- temporary quadripolar pacing leads
- Cardiac pacing wires
- uni-polar temporary pacing wires
- bi-polar temporary pacing wires
- quadri-polar temporary pacing wires
- Cardiac pacing leads/catheter
- Y to o to Y Growth Trend Analysis By Product , 2021 to 2025
- Absolute $ Opportunity Analysis By Product , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Transcatheter aortic valve replacement
- Cardiothoracic surgeries
- Acute myocardial infarction
- Bradycardia
- Heart blocks
- Sick sinus syndrome
- Congestive heart failure
- Transcatheter aortic valve replacement
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Application
- Competition Analysis
- Competition Deep Dive
- Medtronic plc
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- BioTrace Medical (Merit Medical Systems Inc.)
- B. Braun SE
- Oscor Inc
- OSYPKA MEDICAL
- BD
- Abbott
- APC Cardiovascular Ltd
- Teleflex Incorporated
- Lotus Surgicals Pvt Ltd
- Centenial Surgical Suture Ltd
- Taisho Biomed Instruments Co., Ltd.
- Edwards Lifesciences Services GmbH
- Zimmer Biomet
- Medtronic plc
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Product
- Figure 23: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Application
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Product
- Figure 30: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Application
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Product
- Figure 37: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Application
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Product
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Application
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Product
- Figure 51: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Application
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

