The decision to outsource pharmacovigilance activities reflects fundamental changes in how pharmaceutical companies evaluate internal capabilities versus external expertise. Internal pharmacovigilance operations require substantial fixed investments in specialized personnel, technology platforms, and regulatory compliance systems that many companies cannot justify across their product portfolios.
Building internal capabilities demands hiring qualified persons for pharmacovigilance, safety scientists, regulatory affairs specialists, and database administrators who command premium salaries due to their specialized knowledge. These personnel must maintain continuous training on evolving regulatory requirements across multiple jurisdictions, creating ongoing educational expenses that compound over time.
Technology infrastructure represents another significant barrier to internal operations. Pharmacovigilance requires sophisticated safety databases capable of handling multiple data formats, automated case processing workflows, electronic submission capabilities for regulatory authorities, and integration with clinical trial management systems. These platforms require substantial licensing fees, customization costs, and ongoing maintenance expenses.
The complexity of global regulatory requirements makes internal operations particularly challenging for companies operating across multiple markets. Each regulatory authority maintains distinct reporting requirements, submission formats, and compliance expectations that demand specialized knowledge and dedicated resources to manage effectively.
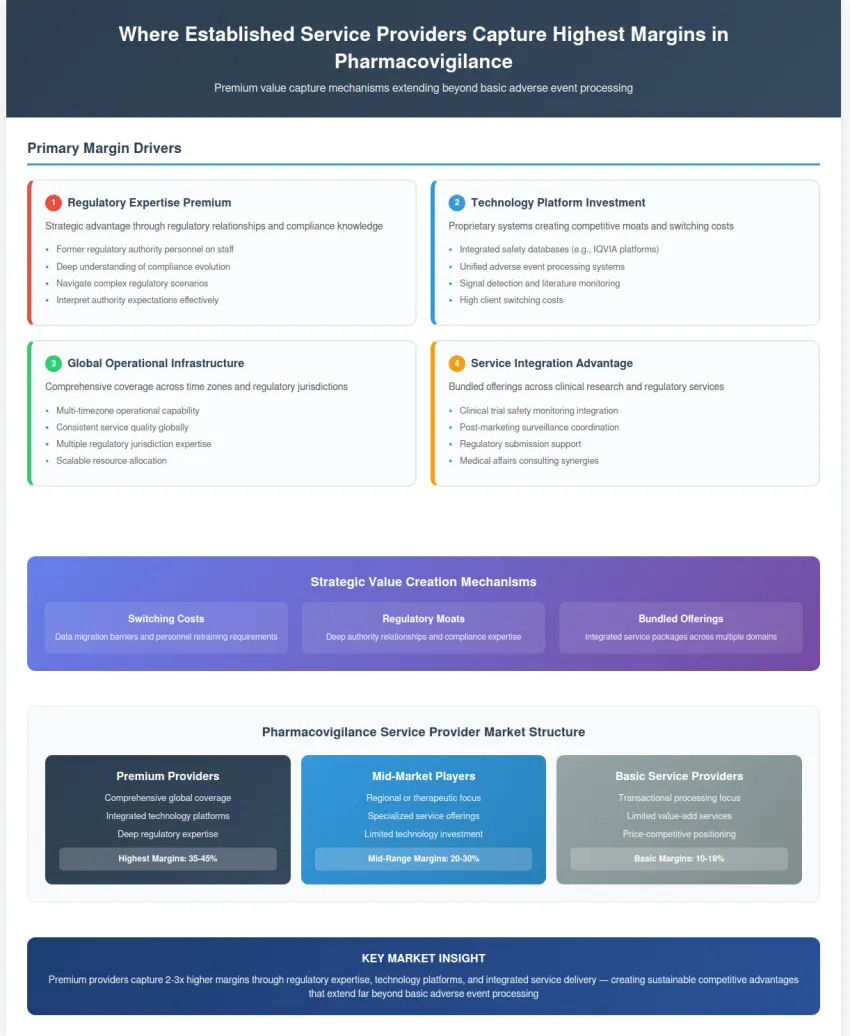
Premium pharmacovigilance service providers have developed sophisticated value capture mechanisms that extend far beyond basic adverse event processing. Their margin structure reflects the strategic positioning they have achieved through regulatory relationships, technology investments, and comprehensive service integration.
Regulatory expertise represents perhaps the most valuable component of their service offering. Established providers maintain teams of former regulatory authority personnel who understand not only current requirements but also how regulations evolve and how authorities interpret compliance expectations. This knowledge enables them to guide clients through complex regulatory scenarios that smaller providers cannot navigate effectively.
Technology platform development creates another layer of competitive advantage. Companies like IQVIA have invested heavily in proprietary safety databases that integrate adverse event processing, signal detection, literature monitoring, and regulatory reporting into unified platforms. These systems create switching costs for clients who would need to migrate data and retrain personnel to change providers.
Global operational infrastructure enables premium providers to offer comprehensive coverage across multiple time zones and regulatory jurisdictions. This capability is particularly valuable for multinational pharmaceutical companies that need consistent service quality and regulatory compliance across their entire product portfolios.
The integration of pharmacovigilance with broader clinical research and regulatory services creates additional value capture opportunities. Providers can offer bundled services that include clinical trial safety monitoring, post-marketing surveillance, regulatory submission support, and medical affairs consulting within integrated service packages.
Small and medium-sized pharmacovigilance service providers face structural disadvantages that extend beyond simple economies of scale. These challenges create barriers that prevent them from effectively competing with established players even when they offer substantially lower pricing.
Technology infrastructure requirements represent a particularly challenging barrier for smaller providers. Pharmacovigilance databases require substantial upfront investments and ongoing maintenance costs that must be amortized across client bases. Smaller providers often cannot justify these investments or must rely on generic platforms that lack the customization capabilities that larger clients expect.
Regulatory expertise concentration among established providers creates knowledge gaps that smaller competitors struggle to bridge. The specialized nature of pharmacovigilance regulations and the limited pool of qualified personnel mean that smaller providers often cannot attract or retain the regulatory expertise necessary to serve complex clients effectively.
Quality assurance and validation requirements imposed by pharmaceutical company clients create additional barriers for smaller providers. Large pharmaceutical companies typically require extensive quality audits, validation documentation, and compliance certifications that smaller providers may not have the resources to maintain.
Global operational capabilities represent another structural disadvantage for smaller providers. Pharmacovigilance requires continuous monitoring and rapid response capabilities across multiple time zones, which smaller providers cannot cost-effectively maintain without substantial infrastructure investments.

Sources
Pharmacovigilance involves complex regulatory compliance requirements, specialized technology infrastructure, and expert interpretation of safety data that extends far beyond simple adverse event reporting. Premium providers offer integrated solutions that include regulatory strategy, global compliance management, and advanced analytics capabilities that justify higher pricing.
Regulatory fees represent substantial fixed costs that pharmaceutical companies must pay regardless of whether they maintain internal pharmacovigilance operations or outsource these functions. Service providers must factor these regulatory costs into their pricing structures, often leading to higher total costs for companies with complex product portfolios.
Smaller companies can access quality pharmacovigilance services through specialized providers that focus on serving smaller clients, but they may need to accept more standardized service offerings rather than fully customized solutions. Some providers offer tiered service models that provide basic compliance coverage at lower price points.
Technology investments enable service providers to offer faster case processing, more accurate signal detection, improved data quality, and enhanced regulatory compliance capabilities. These improvements can reduce regulatory risks for pharmaceutical companies while potentially lowering long-term pharmacovigilance costs through operational efficiencies.
Companies should evaluate regulatory expertise and compliance track records, technology platform capabilities, global operational coverage, quality assurance processes, and integration capabilities with existing systems. The lowest-cost provider may not offer the comprehensive capabilities necessary to manage complex regulatory requirements effectively.
This report covers the coronary heart disease diagnostic imaging market through analysis of market size, revenue forecast, competitive landscape, demand outlook, growth drivers, restraints, imaging pathway shifts, supply-side developments, and strategic growth opportunities.
This report covers the mRNA vaccine formulation stability and stress test systems market through analysis of market size, market share, revenue forecast, system pricing benchmarks, company positioning, competitive landscape, demand outlook, growth drivers, restraints, technology trends, formulation testing requirements, assay integration, analytical method continuity, end-user adoption, supply chain developments, regulatory and quality expectations, investment opportunities, profitability outlook, and strategic growth opportunities during 2026 to 2036.
This report covers the cell therapy cryopreservation and thaw performance test platforms market through analysis of market size, market share, revenue forecast, platform segmentation, assay readout trends, cell type demand, preservation format usage, automation level adoption, end-user demand, workflow stage requirements, regional outlook, technology developments, handling standardization needs, method transfer practices, comparability testing, supply chain dynamics, regulatory and quality expectations, investment opportunities, profitability outlook, and strategic growth opportunities during 2026 to 2036.
The Gene Editing Off-Target Effect Detection Test Systems Market is segmented by Detection Approach (Cell-based Assays, Biochemical Assays, In Silico Tools, Orthogonal Validation), Product Format (Services, Assay Kits, Software, Instruments, Reagents), Workflow Stage (Site Nomination, Site Confirmation, Structural Follow-up, Release Support), Editing Modality (CRISPR-Cas9, Base Editing, Prime Editing, Cas12 Systems, Other Editors), Readout Technology (Amplicon NGS, Short-read WGS, Single-cell DNA, ChIP-seq Workflows, Long-read Sequencing), End User (Biopharma Companies, Academic Laboratories, CROs, CDMOs, Hospitals), Application (IND-enabling Studies, Discovery Screening, Cell Therapy Quality Assessment, In vivo Profiling, Comparability Studies), Deployment (Outsourced Testing, In-house Testing, Hybrid Models), and Region. Forecast for 2026 to 2036.
This report covers the at-home blood collection kits and devices market through analysis of market size, revenue forecast, competitive landscape, demand outlook, growth drivers, restraints, self-collection workflow trends, regulatory developments, and strategic growth opportunities.