The at-home blood collection kits and devices market was valued at an estimated USD 1.48 billion in 2025, is projected to reach USD 1.57 billion in 2026, and is forecast to expand to USD 2.83 billion by 2036 at a CAGR of 6.1%. The incremental opportunity during the forecast period 2026-2036 will be to the tune of USD 1.26 billion
| Parameter | Details |
|---|---|
| Market value (2026) | USD 1.57 billion |
| Forecast value (2036) | USD 2.83 billion |
| CAGR (2026 to 2036) | 6.1% |
| Estimated market value (2025) | USD 1.48 billion |
| Incremental opportunity | USD 1.26 billion |
| Leading product | Self-collection devices, 46.2% of product revenue |
| Leading application | Chronic disease monitoring, 36.9% of application revenue |
| Leading end use | Diagnostic laboratories and laboratory networks, 41.3% of end-use revenue |
| Key players | BD, Tasso, Babson Diagnostics, Neoteryx, Capitainer, Spot On Sciences |
Source: Analyst synthesis from authoritative sources, 2026.
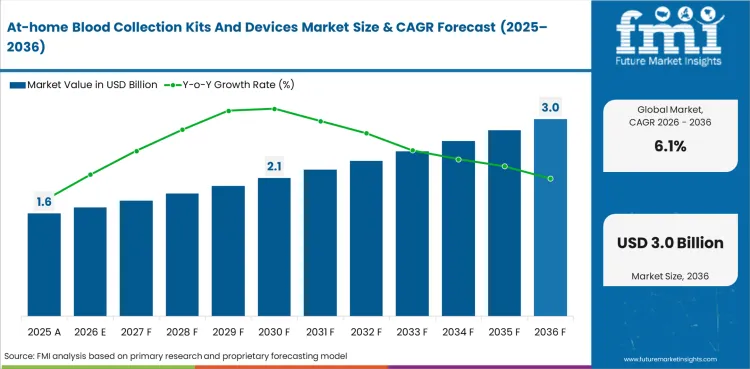
Chronic disease testing remains large, with CDC and NIDDK reporting 38.4 million people with diabetes in the United States alone. Validation studies increasingly show that home-collected blood microsamples can support usable laboratory workflows in research and selected clinical settings. Device makers and laboratory partners are also building alternate collection models that reduce dependence on phlebotomy centers and traditional venous draw infrastructure.
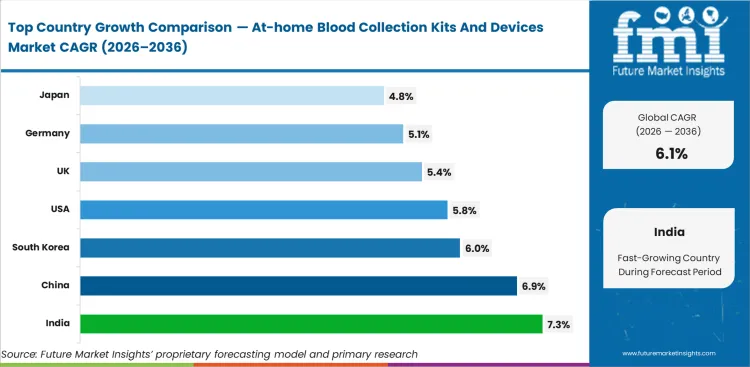
Across tracked geographies, India sets the pace at 7.3% CAGR. China follows at 6.9%. South Korea follows at 6.0%. The United States follows at 5.8%. The United Kingdom follows at 5.4%. Germany follows at 5.1%. Japan follows at 4.8%.
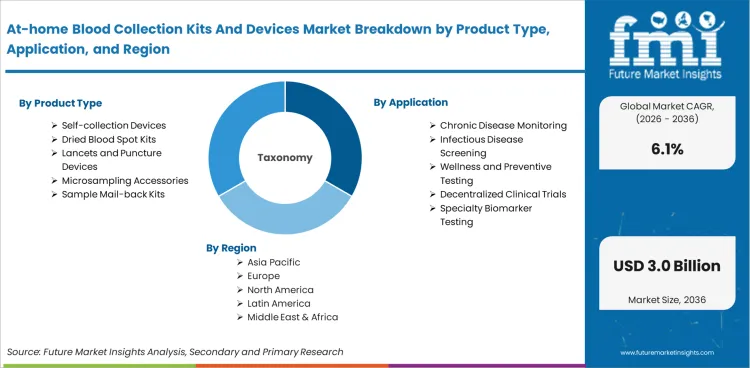
The at-home blood collection kits and devices market covers products used by consumers or non-phlebotomist operators to collect capillary or microsampled blood outside traditional laboratory draw sites for downstream clinical testing, research testing, screening, or monitoring. The market includes self-collection devices, collection kits, dried blood spot cards, volumetric microsampling tools, lancets, sample stabilization materials, and shipping components used in mail-back or decentralized blood testing workflows.
Market scope includes integrated self-collection devices, dried blood spot kits, volumetric absorptive microsampling tools, capillary collection tubes used in home or remote collection pathways, lancets bundled for home collection, sample mailers, stabilizers, and user instruction components. Revenue sizing spans products used for chronic disease monitoring, infectious disease screening, wellness testing, clinical trial sample collection, and specialty biomarker testing processed by central or partner laboratories.
The scope excludes conventional venipuncture systems used in hospitals and laboratories, fully integrated point-of-care analyzers that do not rely on sample return, saliva or urine collection kits, and laboratory assay revenue not linked to the sale or use of at-home blood collection kits or devices.
Recurring blood-based monitoring creates a large demand base. Diabetes, lipids, inflammatory markers, therapeutic drug monitoring, and infectious disease follow-up all benefit from lower-friction sample collection.
Clinical and research programs are moving toward decentralized sampling. Home collection reduces travel burden and can improve follow-up rates in longitudinal studies and remote monitoring programs.
Technology has moved beyond simple lancets. Newer devices combine capillary flow control, stabilized sampling formats, and user-friendly packaging that improve the usability of home collection.
The market is growing because the collection step has become the main bottleneck in many remote testing pathways. Laboratories can already process biomarker panels at scale, but patients still face inconvenience, needle fear, distance, and appointment friction when blood must be drawn in person. At-home collection addresses that friction directly.
Peer-reviewed studies support this shift. The PRE-MACE study found patient-centric remote dried blood sampling feasible in a cardiac setting, and later work in sexually transmitted infection testing showed that home self-collected dried blood spots can support HIV, hepatitis B, hepatitis C, syphilis, and creatinine assessment. These findings do not eliminate all quality concerns, but they improve confidence in remote blood collection workflows.
Demand is also supported by alternate-site care. BD stated that MiniDraw can extend blood collection to physician offices and retail sites without requiring phlebotomy-centered infrastructure. Tasso continues to expand partnerships that use home blood collection in research and home-based diagnostic pathways. As these models scale, product revenue shifts from niche research demand toward broader clinical and consumer-facing use cases.
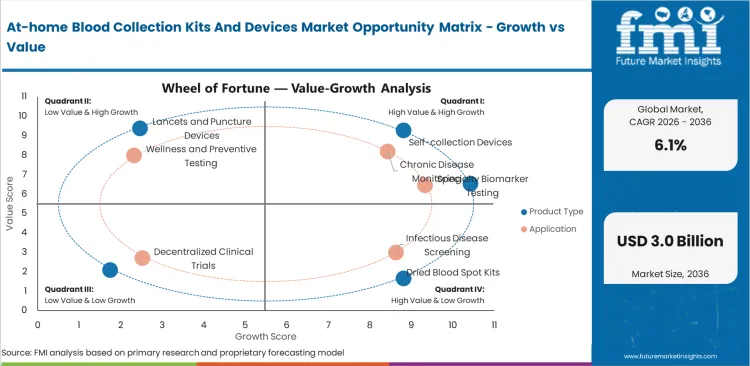
Self-collection devices command 46.2% of product revenue in 2026. The category leads because integrated devices simplify collection, reduce user error, and support broader adoption than stand-alone cards or accessories alone.
Chronic disease monitoring accounts for 36.9% of application revenue in 2026. This segment leads because diabetes, lipid, and cardiometabolic monitoring create repeat testing demand rather than one-time screening demand.
Diagnostic laboratories and laboratory networks hold 41.3% of end-use revenue in 2026. Central lab processing remains dominant because most home collection kits still feed into mail-back or partner-lab workflows rather than on-device analysis.
The market is segmented by product type, sample format, application, end use, distribution channel, and region. By product type, the market includes self-collection devices, dried blood spot kits, microsampling accessories, lancets and puncture devices, and sample mail-back kits. By application, the market covers chronic disease monitoring, infectious disease screening, wellness and preventive testing, decentralized clinical trials, and specialty biomarker testing.
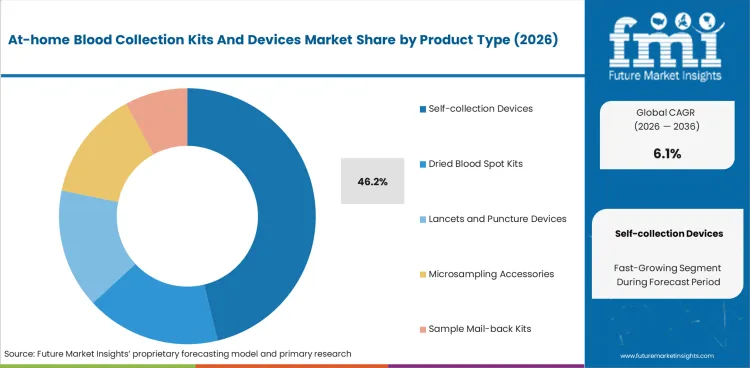
Self-collection devices are projected to hold 46.2% of product revenue in 2026. The segment benefits from easier user workflows, stronger perceived convenience, and ongoing investment by companies trying to replicate laboratory-grade sampling in home settings. Integrated devices also align better with premium pricing than cards or accessory-only kits.
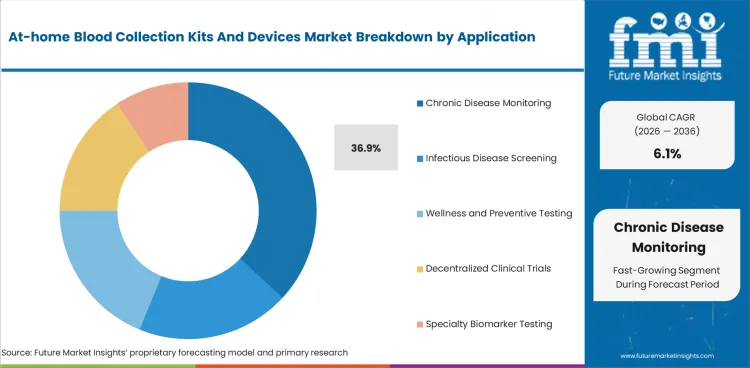
Chronic disease monitoring is expected to capture 36.9% of application revenue in 2026. Frequent testing needs in diabetes, lipids, thyroid, inflammation, and selected medication monitoring support recurring demand. This makes the segment more stable than outbreak-driven infectious disease demand or one-time wellness testing.
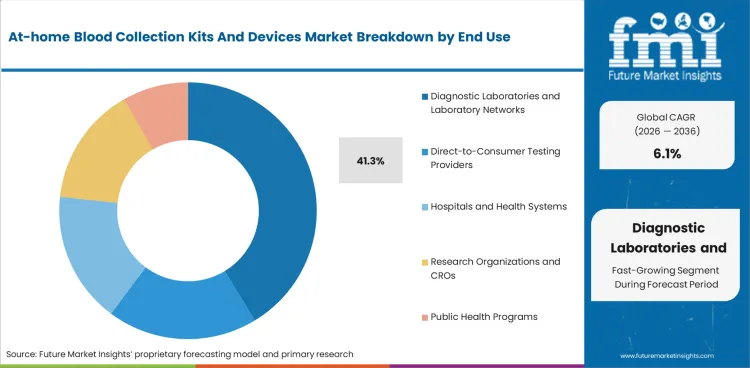
Diagnostic laboratories and laboratory networks are projected to account for 41.3% of end-use revenue in 2026. Most home-collected blood still requires central processing, quality review, and result reporting infrastructure. That keeps the value chain anchored to established laboratories even when collection shifts to the home.
Driver: recurring testing demand in chronic disease management and decentralized research is increasing the need for easier blood collection workflows.
Restraint: sample integrity, user error, transport stability, and regulatory limits still constrain broader clinical deployment.
Trend: integrated microsampling devices and mail-back kits are replacing simpler collection cards in higher-value workflows.
Growth rates differ by use case. Research, cardiometabolic monitoring, and specialty biomarker testing are advancing faster. Applications that require tighter pre-analytical control or product-specific regulatory clearance are growing more slowly.
Growth is supported by the simple fact that millions of patients need repeat blood tests. CDC and NIDDK diabetes statistics alone point to a large recurring monitoring base. When a collection workflow lowers inconvenience and needle aversion, adherence can improve and alternate settings become more viable.
Reliability under real-world conditions remains a core market constraint. FDA stated in October 2024 that it had not authorized blood sample self-collection kits for HIV testing and warned that self-collection raises issues around safe collection, shipping, and sample stability. FDA safety actions in capillary lead testing also show that collection devices can weaken diagnostic accuracy when quality systems fail.
Integrated platforms with user guidance, stabilized formats, and lab-linked software workflows are gaining share. Tasso partnerships and BD MiniDraw commercialization efforts point to more controlled and higher-value remote collection models.
.webp)
Asia is likely to deliver the fastest growth through 2036 because chronic disease burden is rising, digital health adoption is improving, and alternate collection models can bypass some traditional phlebotomy capacity constraints.
The United States remains the largest single-country revenue pool because remote diagnostics, decentralized trials, and retail health partnerships are more developed than in most other markets.
Western Europe grows steadily on the strength of public-health screening pilots, mail-back diagnostic workflows, and strong biosample logistics, but regulatory caution moderates the pace of adoption.
South Korea stands out within developed Asia because digital health infrastructure is strong and home-based health service adoption is relatively advanced.
| Country | CAGR |
|---|---|
| India | 7.3% |
| China | 6.9% |
| South Korea | 6.0% |
| United States | 5.8% |
| United Kingdom | 5.4% |
| Germany | 5.1% |
| Japan | 4.8% |
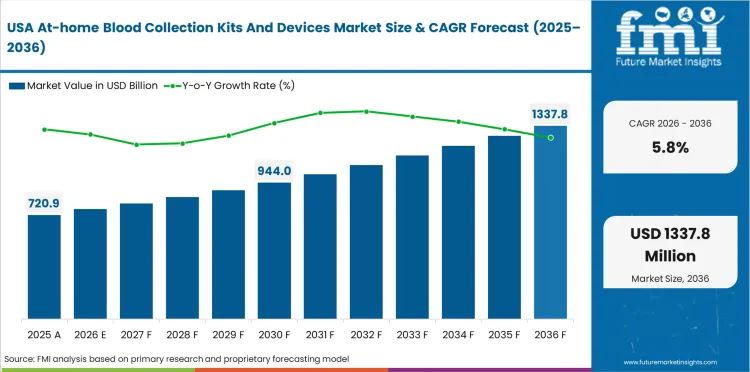
The United States is projected to grow at a CAGR of 5.8% through 2036. Growth is supported by FDA-cleared device availability, retail and laboratory partnerships, decentralized research adoption, and large chronic disease testing volumes.
The United Kingdom is projected to grow at a CAGR of 5.4% through 2036. Home testing acceptance is relatively high, and postal diagnostics infrastructure supports adoption, though public-system validation requirements keep scaling disciplined.
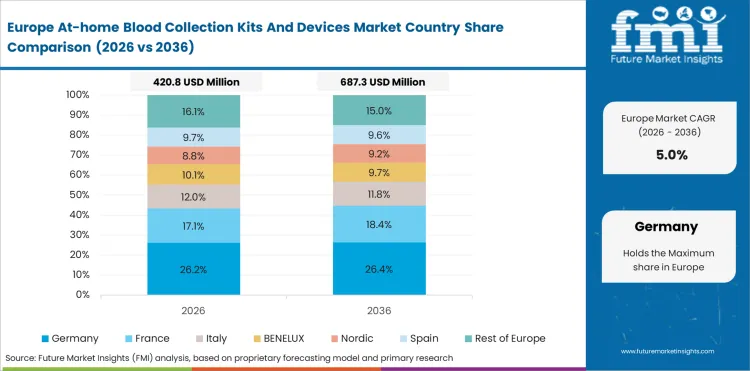
Germany is expected to grow at a CAGR of 5.1% through 2036. Laboratory quality expectations are high, which supports premium device positioning but can slow rapid diffusion of consumer-facing models.
Japan is projected to grow at a CAGR of 4.8% through 2036. The market benefits from aging-related monitoring demand, but mature healthcare delivery and careful workflow validation keep growth moderate.
China is expected to grow at a CAGR of 6.9% through 2036. The market is supported by rising chronic disease prevalence, rapid digital health scaling, and strong interest in alternate collection pathways linked to centralized testing.
India is projected to grow at a CAGR of 7.3% through 2036. Growth reflects expanding diagnostic demand, uneven access to traditional draw infrastructure, and strong potential for alternate collection pathways in urban and peri-urban populations.
The competitive landscape combines large specimen-management incumbents with smaller remote-sampling specialists. Competitive strength depends on regulatory status, user experience, sample quality, laboratory compatibility, and ability to support mail-back or decentralized workflows.
BD holds an advantage in scale, regulatory experience, and healthcare relationships. Tasso holds a strong position in patient-centric remote blood collection and continues to add research and diagnostic partners. Babson Diagnostics occupies a hybrid position by pairing capillary collection with a laboratory workflow and retail collection model.
Neoteryx, Capitainer, and similar microsampling firms compete on dried or volumetric sampling formats that address stability and transport requirements. These models remain attractive in research and specialty biomarker testing, even where they are not yet the default in broad consumer diagnostics.
The market is still fragmented enough for specialized companies to win. Larger companies bring manufacturing and distribution strength, while smaller firms often move faster on user design and decentralized care use cases.
Major Industry Players
BD and Tasso stand out as the most visible companies shaping the category through regulatory milestones and partnership activity. BD offers scale and conventional diagnostic relationships, while Tasso has built a strong patient-centric remote collection identity.
Babson Diagnostics strengthens the category by pairing device-enabled capillary collection with central laboratory processing and alternate-site workflows. Its positioning sits between device maker, collection workflow provider, and testing service partner.
Neoteryx, Capitainer, and Spot On Sciences retain relevance in microsampling-intensive applications where dried or stabilized collection formats matter for transport, research, and specialty testing.
| Company | Remote Collection Strength | Regulatory Position | Lab Workflow Integration | Geographic Reach |
|---|---|---|---|---|
| BD | High | High | High | Strong |
| Tasso | High | Medium | High | Moderate |
| Babson Diagnostics | Medium | Medium | High | Moderate |
| Neoteryx | Medium | Medium | Medium | Moderate |
| Capitainer | Medium | Medium | Medium | Moderate |
| Spot On Sciences | Medium | Low | Medium | Limited |
Source: Analyst synthesis from authoritative sources, 2026. Ratings reflect relative positioning based on disclosed capabilities and market presence.
Key Developments in At-home Blood Collection Kits And Devices Market
Major Global Players:
Emerging Players/Startups
| Metric | Value |
|---|---|
| Quantitative Units | USD 1.57 billion to USD 2.83 billion, at a CAGR of 6.1% |
| Market Definition | Products used to collect blood at home or in remote settings for downstream laboratory testing, screening, research, or monitoring. |
| Segmentation | Product type: self-collection devices, dried blood spot kits, lancets and puncture devices, microsampling accessories, sample mail-back kits. Sample format: capillary liquid blood, dried blood spot, volumetric absorptive microsampling, stabilized microsample formats. Application: chronic disease monitoring, infectious disease screening, wellness and preventive testing, decentralized clinical trials, specialty biomarker testing. End use: diagnostic laboratories and laboratory networks, direct-to-consumer testing providers, hospitals and health systems, research organizations and CROs, public health programs. Distribution channel: online direct sales, laboratory partner channels, pharmacy and retail health, institutional procurement. |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | United States, Canada, United Kingdom, Germany, France, Japan, South Korea, China, India, Australia, Brazil, GCC countries, South Africa |
| Key Companies Profiled | BD, Tasso, Babson Diagnostics, Neoteryx, Capitainer, Spot On Sciences |
| Forecast Period | 2026 to 2036 |
| Approach | Analyst-built hybrid top-down and bottom-up model using chronic disease testing need, remote collection validation evidence, regulatory status, and official company disclosures. |
| Historical Period | 2020 to 2025 |
How large is the demand for At-home Blood Collection Kits And Devices in the global market in 2026?
Demand for at-home blood collection kits and devices in the global market is estimated to be valued at USD 1.57 billion in 2026.
What will be the market size by 2036?
Market size is projected to reach USD 2.83 billion by 2036.
What is the expected demand growth between 2026 and 2036?
Demand is expected to grow at a CAGR of 6.1% between 2026 and 2036.
Which product type is poised to lead by 2026?
Self-collection devices lead the market in 2026 with 46.2% of product revenue.
How are diagnostic laboratories and laboratory networks driving adoption?
Diagnostic laboratories and laboratory networks account for 41.3% of end-use revenue because most home-collected samples still require centralized processing, quality review, and result reporting.
What is driving demand in the United States?
The United States is projected to grow at a CAGR of 5.8% through 2036, supported by FDA-cleared device availability, laboratory partnerships, decentralized research adoption, and large chronic disease testing volumes.
What does the market definition mean?
The market includes products used to collect blood at home or in remote settings for downstream laboratory testing, screening, research, or monitoring.
How does the analyst validate the forecast?
The forecast is validated through a hybrid model using chronic disease testing need, remote collection feasibility evidence, regulatory status, and official company disclosures, with cross-checks on product mix, application mix, and country adoption.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.