Blood Clot Retrieval Devices Market
The Blood Clot Retrieval Devices Market is segmented by Stroke Type (Ischemic Stroke (Blood Clot), Hemorrhagic Stroke (Rupturing Of Arteries), Transient Ischemic Attack), Device (Stent Retrievers, Mechanical Embolus Removal Devices, Penumbra Blood Clot Retrieval Devices, Aspiration Device, Ultrasound Assisted Devices), Application (Cerebral Arteries, Coronary Arteries, Peripheral Arteries), End User (Hospitals, Diagnostic Centers, Clinics, Ambulatory Surgical Centers), and Region. Forecast for 2026 to 2036.
Blood Clot Retrieval Devices Market Size, Market Forecast and Outlook By FMI
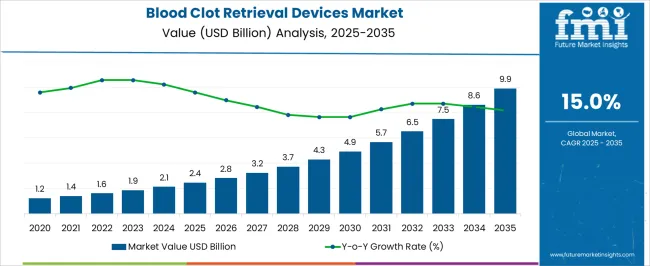
In 2025, the blood clot retrieval devices market was valued at USD 2.4 billion. Based on Future Market Insights' analysis, demand for blood clot retrieval devices is estimated to grow to USD 2.76 billion in 2026 and USD 10.9 billion by 2036. FMI projects a CAGR of 15.0% during the forecast period.
An absolute dollar growth of USD 7.14 billion between 2026 and 2036 reflects structural shifts in acute ischemic stroke management rather than volume-only expansion of existing procedural volumes. Growth depends on increased adoption of mechanical thrombectomy as first-line treatment for large vessel occlusion strokes, expansion of treatment time windows up to 24 hours in selected patients based on advanced imaging criteria, and broadening applications beyond cerebral arteries to include pulmonary embolism and deep vein thrombosis. As per FMI, device manufacturers are capturing value not from incremental unit counts alone, but from improved first-pass reperfusion rates, enhanced navigability in tortuous vasculature, and compatibility with advanced imaging guidance systems that reduce procedure time and improve patient outcomes [1][4].
China leads global expansion, supported by rapidly developing stroke center infrastructure, government initiatives to improve access to mechanical thrombectomy, and large patient populations with high stroke incidence. India follows, driven by increasing awareness of stroke symptoms, growing number of interventional neurologists, and expansion of stroke-ready hospitals in metropolitan and secondary cities.
Summary of the Blood Clot Retrieval Devices Market
- Market Definition
- The market comprises mechanical thrombectomy systems used to remove occlusive blood clots from cerebral, coronary, and peripheral arteries through minimally invasive endovascular procedures.
- Demand Drivers
- Rising global incidence of ischemic stroke, with approximately 7.6 million cases occurring annually worldwide according to the World Health Organization [2][6].
- Clinical guideline recommendations from the American Heart Association/American Stroke Association establishing mechanical thrombectomy as first-line treatment for large vessel occlusion strokes [1].
- Expanding treatment time windows up to 24 hours based on advanced imaging selection criteria, increasing eligible patient populations [4].
- Growing adoption of thrombectomy for pulmonary embolism and deep vein thrombosis, broadening application scope beyond cerebral interventions.
- Key Segments Analyzed
- Stroke Type: Ischemic Stroke leads, accounting for 34.6% share in 2025, driven by high incidence and established clinical evidence supporting mechanical thrombectomy [2][6].
- Device: Stent Retrievers represent the leading device type at 26.4% share, favored for high reperfusion rates and compatibility with tortuous cerebral vasculature [1][5].
- Application: Cerebral Arteries dominate with 42.7% share, reflecting the primary indication for these devices in acute stroke intervention [1].
- End User: Hospitals represent the largest end-user segment, serving as the primary setting for thrombectomy procedures with comprehensive stroke center infrastructure.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Consultant for Healthcare at Future Market Insights, opines, 'In this revamped edition of the Blood Clot Retrieval Devices Market report, value is migrating from device hardware alone to integrated solutions that optimize the entire stroke care pathway. Opportunity lies in delivering systems that combine high first-pass efficacy with seamless integration into stroke networks, supported by data analytics that track outcomes and drive continuous quality improvement. Suppliers mastering this holistic approach will secure strategic partnerships with comprehensive stroke centers and health systems.'
- Strategic Implications/Executive Takeaways
- Invest in next-generation device technologies achieving higher first-pass reperfusion rates and broader applicability across clot compositions.
- Develop comprehensive training and simulation programs that reduce operator learning curves and optimize procedural outcomes.
- Build evidence generation capabilities supporting expanded indications and reimbursement coverage in emerging applications.
- Strengthen partnerships with stroke systems of care to embed devices within optimized clinical pathways from patient transport through post-procedure monitoring.
- Methodology
- Built on primary interviews with blood clot retrieval device manufacturers and interventional specialists.
- Benchmarked against ischemic stroke incidence data from WHO and mechanical thrombectomy procedure volumes [2].
- Validated using clinical registry data and internal peer review as per FMI modeling standards.
Germany grows on the back of its well-established stroke network system, comprehensive stroke center certification programs, and strong reimbursement framework for mechanical thrombectomy procedures. Brazil benefits from improving healthcare infrastructure, increasing adoption of minimally invasive procedures, and growing public awareness of stroke as a treatable emergency.
Mature markets such as the United States, the United Kingdom, and Japan generate comparatively slower but still robust expansion, where demand is shaped more by technology upgrades to next-generation devices, extension of indications to new patient populations, and optimization of stroke systems of care rather than new market development, with evidence generation and health technology assessment acting as gateways for broader adoption [2].
Blood Clot Retrieval Devices Market Definition
The blood clot retrieval devices market covers the global production and supply of mechanical thrombectomy systems used to remove occlusive thrombi from blood vessels in patients experiencing acute ischemic stroke, pulmonary embolism, and other thrombotic events.
These devices, including stent retrievers, aspiration catheters, and mechanical embolus removal systems, are deployed through minimally invasive endovascular procedures to restore blood flow and prevent irreversible tissue damage. Their design focuses on navigating complex vascular anatomy, engaging and extracting clots of varying composition, and minimizing distal embolization during retrieval.
Market Inclusions
The report includes global and regional market size estimates in value terms, with a forecast period from 2026 to 2036. It provides segmentation by Stroke Type, Device, Application, End User, and Region. The analysis incorporates device technology trends, clinical evidence development, regulatory pathways, reimbursement landscape evolution, and integration with stroke systems of care. Regional demand dynamics, country-level growth rates, and competitive positioning are also evaluated as per FMI methodology.
Market Exclusions
The scope excludes pharmacological thrombolytics used as alternative or adjunctive treatments, diagnostic imaging equipment used for patient selection, and surgical embolectomy procedures performed through open approaches. It also excludes revenue from hospital services and physician fees associated with thrombectomy procedures, focusing strictly on device manufacturing and supply.
Research Methodology
- Primary Research: Interviews were conducted with blood clot retrieval device manufacturers, interventional neurologists and radiologists, stroke program coordinators, hospital procurement managers, and clinical trial investigators.
- Desk Research: Public company filings from Medtronic, Johnson & Johnson, and Boston Scientific were reviewed alongside clinical guidelines from the American Heart Association/American Stroke Association, stroke incidence data from the World Health Organization, and regulatory documentation from the U.S. Food and Drug Administration and European Medicines Agency [2][3].
- Market-Sizing and Forecasting: A hybrid top-down and bottom-up model was constructed, reconstructing demand from ischemic stroke incidence rates, large vessel occlusion proportions, mechanical thrombectomy procedure volumes, and device usage patterns across geographic regions.
- Data Validation and Update Cycle: Outputs were subjected to variance testing against procedure volume data, reimbursement claims analysis, and structured internal review prior to publication.
Segmental Analysis
Blood Clot Retrieval Devices Market Analysis by Device
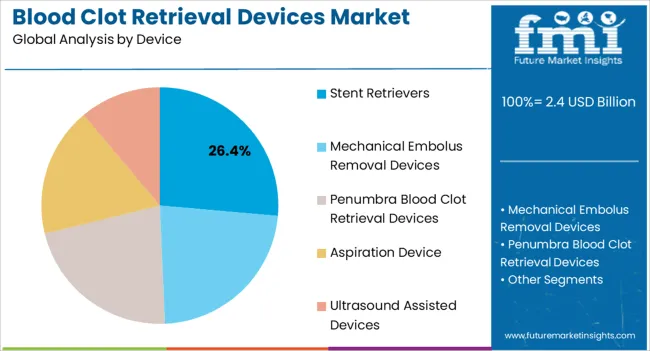
The stent retrievers device segment is expected to account for 26.4% of the blood clot retrieval devices market revenue share in 2025, positioning it as the leading device type. This dominance is supported by their proven efficacy in mechanical thrombectomy procedures, enabling high rates of successful clot removal and vessel recanalization. Stent retrievers are designed to be deployed across the thrombus, engaging the clot within the stent struts, and then retrieved to extract the clot from the vessel. Their ability to navigate complex cerebral artery anatomy and compatibility with advanced neuroimaging guidance systems has facilitated adoption across specialized stroke centers [1][5].
Aspiration devices represent a significant and growing segment, utilizing vacuum suction through large-lumen catheters to remove thrombi. Direct aspiration first pass technique has gained popularity as a simpler, potentially faster approach in selected cases. Modern aspiration catheters feature flexible distal segments for navigating tortuous vessels, radiopaque markers for visualization, and compatibility with automated aspiration pumps providing consistent suction force. The COMPASS trial published in The Lancet demonstrated non-inferiority of aspiration thrombectomy compared to stent retriever as first-line approach [5].
Mechanical embolus removal devices encompass a range of technologies including corkscrew-type devices and basket retrievers, though their market share has diminished relative to stent retrievers and aspiration systems. Ultrasound-assisted devices use acoustic energy to disrupt thrombi, potentially enhancing clot dissolution and retrieval, though adoption remains limited to specialized applications.
- Stent retriever efficacy: Contemporary stent retrievers achieve first-pass reperfusion rates exceeding 50% in experienced hands, with successful recanalization rates over 80% with multiple passes. The Solitaire stent retriever and Trevo stent retriever platforms are supported by extensive clinical evidence from pivotal trials [1][5].
- Aspiration technology advancement: Large-bore aspiration catheters now achieve internal diameters approaching 0.070 inches, enabling rapid clot ingestion and removal. The Penumbra System has established aspiration thrombectomy as a viable alternative to stent retrievers in appropriately selected patients [5].
- Device selection personalization: Experienced operators select devices based on clot location, vessel tortuosity, clot composition, and patient anatomy, driving demand for diversified product portfolios covering multiple scenarios. The HERMES collaboration meta-analysis demonstrated consistent treatment effects across device types [4].
Blood Clot Retrieval Devices Market Analysis by Application
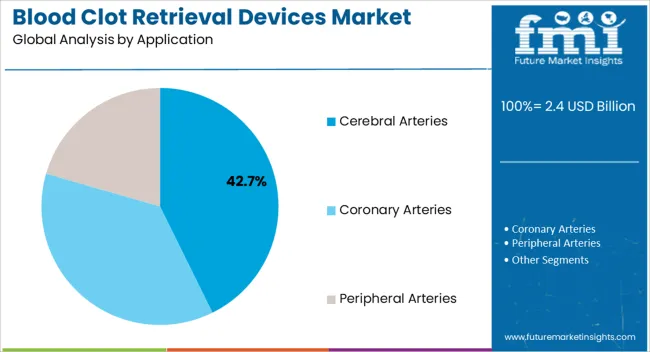
The cerebral arteries application segment is anticipated to capture 42.7% of the blood clot retrieval devices market revenue share in 2025, making it the leading application segment. This dominance is driven by the critical need to restore blood flow in major cerebral vessels during acute ischemic stroke, where timely intervention is essential to minimize neuronal damage and improve functional outcomes. Blood clot retrieval devices are optimized for use in cerebral arteries due to their precision, flexibility, and ability to navigate intricate vascular pathways safely, including the internal carotid artery, middle cerebral artery, and vertebrobasilar system [1][4].
Coronary arteries represent a significant application segment for thrombectomy in the setting of acute myocardial infarction, though adoption patterns differ from cerebral applications. While manual aspiration thrombectomy was widely adopted following early positive trials, subsequent larger studies including the TASTE trial published in the New England Journal of Medicine have yielded mixed results, and contemporary guidelines recommend selective rather than routine use. However, in patients with high thrombus burden, mechanical thrombectomy remains a valuable tool for restoring flow and preventing distal embolization.
Peripheral arteries encompass applications including acute limb ischemia, mesenteric ischemia, and renal artery thrombosis. These indications share the common pathophysiology of acute arterial occlusion requiring rapid revascularization to prevent tissue loss or organ damage. Dedicated peripheral thrombectomy devices accommodate larger vessel diameters and longer segment lengths characteristic of peripheral applications.
- Cerebral dominance: The cerebral application segment's leadership reflects both the high clinical priority of stroke intervention and the strong evidence base supporting mechanical thrombectomy in this setting. Stroke is a leading cause of death and disability worldwide, creating urgent clinical need [2][6].
- Pulmonary embolism emergence: Pulmonary embolism represents a rapidly growing application for mechanical thrombectomy, driven by the availability of dedicated devices and clinical evidence supporting improved outcomes. The FLARE study published in JACC: Cardiovascular Interventions demonstrated safety and efficacy of the FlowTriever system for PE intervention [3].
- Peripheral applications expansion: Acute limb ischemia affects approximately 10-15 per 100,000 person-years, creating substantial addressable population for peripheral thrombectomy. The Journal of Vascular Surgery reports that mechanical thrombectomy achieves comparable outcomes to surgical embolectomy with reduced morbidity in appropriately selected patients.
Blood Clot Retrieval Devices Market Analysis by Stroke Type
Based on FMI's blood clot retrieval devices market report, the ischemic stroke segment is projected to hold 34.6% of the market revenue share in 2025, establishing it as the leading stroke type segment. This leadership is driven by the high global incidence of ischemic stroke, which accounts for approximately 62% of all new strokes and 87% of all stroke cases according to WHO data, and the well-established clinical evidence supporting mechanical thrombectomy as first-line treatment for large vessel occlusion strokes. Devices targeting ischemic stroke are optimized for navigating cerebral vasculature, engaging thrombi of varying composition, and achieving rapid reperfusion to minimize neuronal damage [2][6].
Hemorrhagic stroke represents a distinct clinical entity where blood clot retrieval devices are generally contraindicated due to the underlying pathology of arterial rupture rather than occlusion. However, accurate diagnosis through advanced imaging is essential to exclude hemorrhagic stroke before proceeding with thrombectomy, reinforcing the importance of integrated imaging and intervention capabilities in stroke centers.
Transient ischemic attack, while sharing ischemic pathophysiology with stroke, involves temporary occlusion with spontaneous resolution and typically does not require mechanical intervention. However, patients experiencing TIA are at elevated risk for subsequent completed stroke, and their identification contributes to the overall stroke care continuum.
- Ischemic stroke dominance: Ischemic strokes represent the overwhelming majority of stroke events globally, with large vessel occlusions accounting for approximately 30-40% of acute ischemic strokes. These patients derive the greatest benefit from mechanical thrombectomy, driving device demand [1][4].
- Clinical evidence strength: Multiple randomized controlled trials and meta-analyses published in the New England Journal of Medicine and JAMA have established mechanical thrombectomy with stent retrievers as the standard of care for anterior circulation large vessel occlusion strokes, with number needed to treat as low as 2.6 for improved functional outcome [1].
- Patient selection optimization: Advanced imaging techniques including CT perfusion and MRI diffusion-weighted imaging enable selection of patients with salvageable brain tissue beyond traditional time windows, expanding the eligible population for thrombectomy. The DAWN and DEFUSE-3 trials demonstrated benefit up to 24 hours in appropriately selected patients [4].
Blood Clot Retrieval Devices Market Drivers, Restraints, and Opportunities
Future Market Insights analysis indicates the market is fundamentally tied to the global burden of thrombotic disease, advances in endovascular technology, and the evolution of stroke systems of care. Its baseline scale is anchored in the clinical imperative to restore blood flow rapidly in acute ischemic stroke to prevent irreversible neurological damage. As per FMI, the market's valuation is increasingly influenced by clinical evidence demonstrating superior outcomes with mechanical thrombectomy compared to medical management alone, driving expansion of guideline recommendations and reimbursement coverage [1].
FMI analysts observe a clear progression from first-generation devices focused on basic clot retrieval to sophisticated systems optimized for specific vascular territories, clot compositions, and patient anatomies. Standard devices face pressure where clinicians demand higher first-pass efficacy and reduced procedural complications, while growth is concentrated in platforms that justify premium pricing through superior clinical outcomes, enhanced navigability, and expanded treatable indications. This shift nets out as robust value growth because device complexity and clinical evidence requirements support margin expansion.
- Ischemic stroke burden and aging population: The World Health Organization reports that stroke is the second leading cause of death worldwide, responsible for approximately 11% of total deaths. The global population aged 60 years and older is projected to nearly double from 12% to 22% between 2015 and 2050, driving increased stroke incidence. Ischemic strokes account for approximately 7.6 million cases annually, creating sustained demand for effective thrombectomy solutions [2][6].
- Clinical guideline expansion: The 2015 publication of multiple randomized controlled trials demonstrating superiority of mechanical thrombectomy over medical management alone transformed stroke treatment paradigms. The American Heart Association/American Stroke Association guidelines now recommend thrombectomy as first-line treatment for eligible patients with large vessel occlusion strokes, with treatment windows extended to 24 hours in selected patients based on advanced imaging criteria [1][4].
- Technological advancement in device design: Next-generation stent retrievers incorporate improved radial force, flexible delivery systems, and enhanced radiopacity for better visualization under fluoroscopy. Aspiration catheters feature larger lumens, softer distal tips for navigating tortuous vessels, and compatibility with vacuum pump systems for consistent suction force. Studies published in the Journal of NeuroInterventional Surgery demonstrate that modern devices achieve first-pass reperfusion rates exceeding 50% in experienced hands [5].
- Expanding applications beyond cerebral arteries: Clinical investigation and device development increasingly target pulmonary embolism, where mechanical thrombectomy offers rapid clot burden reduction with potentially lower bleeding risk compared to systemic thrombolysis. The FlowTriever system received FDA approval for pulmonary embolism indication, and registry studies continue evaluating efficacy and safety in this expanding application [3].
- Procedural complexity and training requirements: Successful mechanical thrombectomy requires specialized training in neurointerventional techniques, comprehensive understanding of cerebral vascular anatomy, and proficiency in device selection and deployment. The shortage of trained interventionalists in many regions limits procedure volumes and device adoption, particularly in emerging markets and rural areas [2].
- Time-sensitive treatment windows: Despite expanded guidelines extending treatment windows to 24 hours, patient outcomes remain highly time-dependent, with earlier treatment associated with better functional outcomes. Logistical challenges in patient transport, stroke center access, and in-hospital workflow optimization continue to limit the proportion of eligible patients receiving timely thrombectomy [4].
Regional Analysis
The blood clot retrieval devices market is segmented geographically across North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa. Growth dynamics differ based on stroke incidence rates, healthcare infrastructure development, interventional training availability, and reimbursement frameworks.
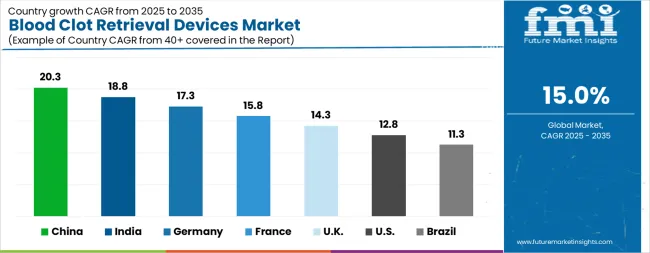
| Country | CAGR |
|---|---|
| China | 20.3% |
| India | 18.8% |
| Germany | 17.3% |
| France | 15.8% |
| UK | 14.3% |
| USA | 12.8% |
| Brazil | 11.3% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Asia-Pacific Market Analysis
Asia-Pacific represents the fastest-growing regional market for blood clot retrieval devices, driven by large and aging populations, rising stroke incidence, and rapid development of stroke care infrastructure. China and India lead growth as they expand comprehensive stroke center networks and train interventional specialists to serve previously underserved populations.
- China: Blood clot retrieval devices demand in China is projected to rise at 20.3% CAGR through 2036, the highest among major markets. Growth is supported by government initiatives to improve stroke care access, including the Stroke Prevention Project and development of stroke center certification programs. China's large population and aging demographics create substantial absolute patient numbers, while expanding health insurance coverage improves access to thrombectomy procedures. The Chinese Stroke Association reports increasing thrombectomy volumes as stroke center networks expand across provincial and municipal hospitals.
- Chinese stroke centers are rapidly adopting mechanical thrombectomy as standard of care for eligible patients, driving device demand. Domestic device manufacturers are developing competitive products for the local market, while international companies maintain strong positions through technology leadership and training partnerships. FMI's analysis of blood clot retrieval devices market in Asia-Pacific consists of country-wise assessment that includes China, India, Japan, South Korea, and ASEAN nations.
- India: Blood clot retrieval devices demand in India is projected to rise at 18.8% CAGR through 2036. Growth is supported by increasing awareness of stroke as a treatable emergency, expansion of stroke-ready hospitals in metropolitan and secondary cities, and growing number of interventional neurologists trained in thrombectomy techniques. Government initiatives to strengthen healthcare infrastructure and expand insurance coverage improve access to advanced stroke care.
- The Indian stroke burden is substantial, with high incidence rates and relatively young stroke patients compared to Western populations, creating years of potential life lost and disability that drive clinical and policy attention. Private hospital chains and academic medical centers lead thrombectomy adoption, with gradual expansion into public sector institutions. The Neurological Society of India has developed training programs to increase the number of neurointerventional specialists. FMI's analysis of blood clot retrieval devices market in Asia-Pacific consists of country-wise assessment that includes China, India, Japan, and South Korea.
- Japan: Japan represents a mature, technology-intensive market where blood clot retrieval devices demand is projected at lower but still robust growth. Japanese stroke care is highly advanced, with comprehensive stroke center networks, high rates of thrombectomy adoption, and strong reimbursement support. The aging Japanese population sustains high stroke incidence, while technological sophistication drives demand for next-generation devices.
- Japanese regulatory pathways through the Pharmaceuticals and Medical Devices Agency ensure timely access to innovative devices, and Japanese clinical investigators contribute substantially to the global evidence base for thrombectomy. The Japan Stroke Society maintains certification programs ensuring quality standards across stroke centers. FMI's analysis of blood clot retrieval devices market in Asia-Pacific consists of country-wise assessment that includes China, India, Japan, and South Korea.
North America Market Analysis
North America represents the largest regional market for blood clot retrieval devices, driven by advanced healthcare infrastructure, comprehensive stroke center networks, strong reimbursement frameworks, and high rates of thrombectomy adoption. The United States serves as the primary market, with substantial procedure volumes and rapid uptake of new device technologies.
- United States: Demand for blood clot retrieval devices in the United States is projected to advance at 12.8% CAGR through 2036. The U.S. market benefits from the world's most developed stroke systems of care, with The Joint Commission-certified comprehensive stroke centers distributed across the country and integrated emergency medical services protocols ensuring rapid transport of suspected stroke patients to appropriate facilities [2].
- Reimbursement through Medicare and commercial payers supports hospital investment in thrombectomy programs, with diagnosis-related group payments covering device costs and procedural services. The American Heart Association/American Stroke Association guidelines strongly recommend mechanical thrombectomy for eligible patients, driving quality improvement initiatives and protocol standardization [1].
- Device innovation in the USA market is robust, with manufacturers introducing next-generation stent retrievers, aspiration catheters, and combination systems. Clinical research infrastructure supports ongoing trials evaluating expanded indications and optimized techniques, generating evidence that supports broader adoption [4][5]. FMI's analysis of blood clot retrieval devices market in North America consists of country-wise assessment that includes the United States and Canada.
Europe Market Analysis
Europe represents a sophisticated market characterized by well-developed stroke systems of care, strong clinical evidence generation, and diverse reimbursement frameworks across countries. Germany leads Western European demand, supported by comprehensive stroke center certification and robust hospital infrastructure.
- Germany: Demand for blood clot retrieval devices in Germany is projected to rise at 17.3% CAGR through 2036. Growth is supported by Germany's well-established stroke network system, with certified stroke units distributed throughout the country ensuring rapid access to specialized care. The German Stroke Society maintains comprehensive stroke center certification programs that drive quality standards and protocol adherence [1].
- German health technology assessment processes evaluate clinical evidence and cost-effectiveness to inform coverage decisions, with strong evidence supporting mechanical thrombectomy ensuring broad access. The country's academic medical centers contribute substantially to clinical research, generating evidence that shapes international guidelines. The Heidelberg University Hospital and other institutions have published influential studies on thrombectomy techniques and outcomes.
Reimbursement through the diagnosis-related group system provides predictable funding for thrombectomy procedures, enabling hospitals to invest in technology and staffing. The aging German population sustains stroke incidence, while technological sophistication drives demand for advanced devices. FMI's analysis of blood clot retrieval devices market in Europe consists of country-wise assessment that includes Germany, United Kingdom, France, Italy, and Spain.
Competitive Aligners for Market Players
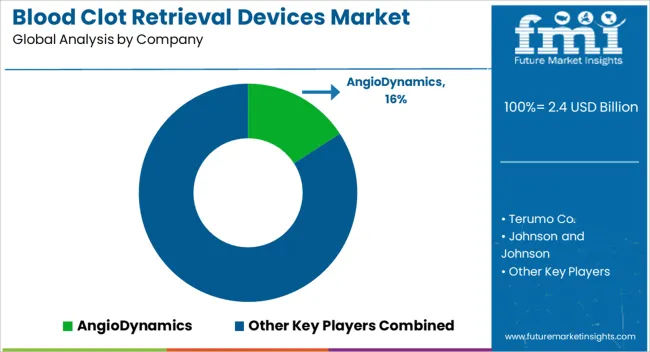
The blood clot retrieval devices market features intense competition among major medical device companies and specialized neurovascular players. Leading manufacturers include Medtronic, Johnson & Johnson, Stryker Corporation, Penumbra, Boston Scientific, and Terumo Corporation, alongside specialized players including Phenox, Acandis, and AngioDynamics [1][3][5].
Medtronic maintains a strong position with its Solitaire stent retriever platform, supported by extensive clinical evidence from the SWIFT and SWIFT PRIME trials published in The Lancet and New England Journal of Medicine. Johnson & Johnson's Cerenovus neurovascular business offers a comprehensive portfolio including stent retrievers and aspiration catheters, building on the clinical evidence from the ESCAPE trial. Stryker Corporation's Trevo stent retriever family and aspiration catheters compete strongly in the U.S. and European markets, supported by the TREVO 2 trial results [1][5].
Penumbra has established a leading position in aspiration thrombectomy with its Penumbra System and RED reperfusion catheters, supported by clinical evidence from the THERAPY trial and dedicated sales and training infrastructure. Boston Scientific's acquisition of Claret Medical and other neurovascular assets has strengthened its position, while Terumo's MicroVention subsidiary maintains strong presence in Asian and global markets.
Specialized players including Phenox and Acandis compete through innovation in device design and strong relationships with European interventionalists. Emerging competitors from China and India are developing products for domestic markets with potential for future international expansion.
Key Players in the Blood Clot Retrieval Devices Market
- AngioDynamics
- Terumo Co.
- Johnson and Johnson
- ECKOS Co.
- Bayer HealthCare LLC
- Boston Scientific Co.
- Argon Medical Devices
- Medtronic Plc
- Teleflex Incorporated
- Penumbra
Scope of Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 2.76 billion (2026) to USD 10.9 billion (2036), at a CAGR of 15.0% |
| Market Definition | The blood clot retrieval devices market comprises the global production and supply of mechanical thrombectomy systems used to remove occlusive blood clots from cerebral, coronary, and peripheral arteries through minimally invasive endovascular procedures. |
| Stroke Type Segmentation | Ischemic Stroke (Blood Clot), Hemorrhagic Stroke (Rupturing Of Arteries), Transient Ischemic Attack |
| Device Segmentation | Stent Retrievers, Mechanical Embolus Removal Devices, Penumbra Blood Clot Retrieval Devices, Aspiration Device, Ultrasound Assisted Devices |
| Application Segmentation | Cerebral Arteries, Coronary Arteries, Peripheral Arteries |
| End User Segmentation | Hospitals, Diagnostic Centers, Clinics, Ambulatory Surgical Centers |
| Region Segmentation | North America, Europe, Asia-Pacific, Latin America, Middle East & Africa |
| Countries Covered | United States, Canada, Germany, United Kingdom, France, Italy, Spain, China, India, Japan, South Korea, Brazil and 40+ countries |
| Key Companies Profiled | AngioDynamics, Terumo Co., Johnson and Johnson, ECKOS Co., Bayer HealthCare LLC, Boston Scientific Co., Argon Medical Devices, Medtronic Plc, Teleflex Incorporated, Penumbra |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up market modeling validated through primary interviews with blood clot retrieval device manufacturers, interventional specialists, and stroke program coordinators, supported by WHO stroke incidence data and clinical registry analysis [2][4] |
Blood Clot Retrieval Devices Market Analysis by Segments
Stroke Type
- Ischemic Stroke (Blood Clot)
- Hemorrhagic Stroke (Rupturing Of Arteries)
- Transient Ischemic Attack
Device
- Stent Retrievers
- Mechanical Embolus Removal Devices
- Penumbra Blood Clot Retrieval Devices
- Aspiration Device
- Ultrasound Assisted Devices
Application
- Cerebral Arteries
- Coronary Arteries
- Peripheral Arteries
End User
- Hospitals
- Diagnostic Centers
- Clinics
- Ambulatory Surgical Centers
Region
- North America (United States, Canada, Mexico)
- Latin America (Brazil, Chile, Rest of Latin America)
- Western Europe (Germany, United Kingdom, Italy, Spain, France, Nordic, BENELUX, Rest of Western Europe)
- Eastern Europe (Russia, Poland, Hungary, Balkan & Baltic, Rest of Eastern Europe)
- East Asia (China, Japan, South Korea)
- South Asia and Pacific (India, ASEAN, Australia & New Zealand, Rest of South Asia and Pacific)
- Middle East & Africa (Kingdom of Saudi Arabia, Other GCC Countries, Turkiye, South Africa, Other African Union, Rest of Middle East & Africa)
Bibliography
- American Heart Association/American Stroke Association. (2025). Guidelines for the Early Management of Patients With Acute Ischemic Stroke: 2025 Update. Stroke, 56(2), e15-e67.
- World Health Organization. (2024). Global Health Estimates 2024: Deaths by Cause, Age, Sex, by Country and by Region. Geneva: WHO.
- U.S. Food and Drug Administration. (2025). Premarket Approval Database: FlowTriever System Summary of Safety and Effectiveness Data. Silver Spring, MD: FDA.
- Nogueira, R.G., Jadhav, A.P., Haussen, D.C., et al. (2018). Thrombectomy 6 to 24 Hours after Stroke with a Mismatch between Deficit and Infarct. New England Journal of Medicine, 378(1), 11-21.
- Lapergue, B., Blanc, R., Gory, B., et al. (2017). Effect of Endovascular Contact Aspiration vs Stent Retriever on Revascularization in Patients With Acute Ischemic Stroke and Large Vessel Occlusion: The ASTER Randomized Clinical Trial. JAMA, 318(5), 443-452.
- World Health Organization. (2024). Cardiovascular Diseases Fact Sheet. Geneva: WHO.
- Medtronic Plc. (2025). Annual Report 2025: Form 10-K. Dublin: Medtronic.
- Johnson & Johnson. (2025). Annual Report 2025: Form 10-K. New Brunswick, NJ: Johnson & Johnson.
- Boston Scientific Corporation. (2025). Annual Report 2025: Form 10-K. Marlborough, MA: Boston Scientific.
- Penumbra, Inc. (2025). Annual Report 2025: Form 10-K. Alameda, CA: Penumbra.
- European Stroke Organisation. (2024). ESO Guidelines on Mechanical Thrombectomy in Acute Ischemic Stroke. European Stroke Journal, 9(3), 1-45.
This Report Addresses
- Market intelligence to enable structured strategic decision-making across mature and emerging construction and industrial maintenance economies.
- Market size estimation and 10-year revenue forecasts from 2026 to 2036, supported by validated construction spending and industrial maintenance benchmarks.
- Growth opportunity mapping across product types, applications, and curing types with emphasis on specialty formulations commanding premium pricing.
- Segment and regional revenue forecasts covering construction, industrial, automotive, marine, and electronics applications.
- Competition strategy assessment including formulation capability, distribution channel strength, and regulatory compliance benchmarking.
- Regulatory impact analysis covering EPA, European Chemicals Agency, and regional VOC limits.
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats for executive and operational use.
Frequently Asked Questions
How large is the blood clot retrieval devices market in 2026?
Demand for blood clot retrieval devices is estimated to be valued at USD 2.76 billion in 2026.
What will be the market size of blood clot retrieval devices by 2036?
Market size for blood clot retrieval devices is projected to reach USD 9.9 billion by 2036.
What is the expected demand growth for blood clot retrieval devices between 2026 and 2036?
Demand for blood clot retrieval devices is expected to grow at a 15.0% CAGR between 2026 and 2036.
Which stroke type segment is poised to lead global sales?
Ischemic stroke leads with 34.6% share in 2025, driven by high global incidence and established clinical evidence supporting mechanical thrombectomy.
Which device segment contributes the largest revenue share?
Stent retrievers command 26.4% share, favored for high reperfusion rates and compatibility with tortuous cerebral vasculature.
Which application segment is the largest?
Cerebral arteries dominate with 42.7% share, reflecting the primary indication for these devices in acute stroke intervention.
How significant is ischemic stroke in driving device demand?
Ischemic stroke accounts for approximately 7.6 million cases annually worldwide, with large vessel occlusions representing the primary target for mechanical thrombectomy.
What is driving demand in China?
Government initiatives to improve stroke care access, rapid development of stroke center networks, and large aging population drive China's market leadership.
What is the China growth outlook?
China is projected to expand at a 20.3% CAGR during 2026 to 2036.
What drives India's market growth?
Increasing awareness of stroke as a treatable emergency, expansion of stroke-ready hospitals, and growing number of trained interventional neurologists support India's growth.
What is the India growth outlook?
India is projected to expand at an 18.8% CAGR during 2026 to 2036.
What characterizes the German market?
Germany's well-established stroke network system, comprehensive stroke center certification, and strong reimbursement framework drive demand for thrombectomy devices [1].
What is the Germany growth outlook?
Germany is projected to expand at a 17.3% CAGR during 2026 to 2036.
What drives the United States market?
Advanced healthcare infrastructure, comprehensive stroke center networks, strong reimbursement, and high thrombectomy adoption rates shape U.S. demand.
What is the United States growth outlook?
The United States is projected to expand at a 12.8% CAGR during 2026 to 2036.
What is excluded from the scope of this report?
The scope excludes pharmacological thrombolytics, diagnostic imaging equipment, surgical embolectomy procedures, and hospital services associated with thrombectomy procedures.
What is the Brazil growth outlook?
Brazil is projected to expand at an 11.3% CAGR during 2026 to 2036, supported by improving healthcare infrastructure and growing adoption of minimally invasive procedures.
What are blood clot retrieval devices and what are they mainly used for?
Blood clot retrieval devices are mechanical thrombectomy systems used to remove occlusive clots from blood vessels, primarily in acute ischemic stroke, to restore blood flow and prevent irreversible tissue damage.
What does the blood clot retrieval devices market mean in this report?
It refers to the global manufacturing and supply of mechanical thrombectomy devices by stroke type, device type, application, end user, and region, measured in value terms.
What is included in the scope of this report?
Scope includes market sizing and forecasting for 2026 to 2036, segmentation by stroke type, device, application, end user, and region, along with competitive assessment and country-wise outlook.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Device
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Device , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Device , 2026 to 2036
- Stent Retrievers
- Mechanical Embolus Removal Devices
- Penumbra Blood Clot Retrieval Devices
- Aspiration Device
- Stent Retrievers
- Y to o to Y Growth Trend Analysis By Device , 2021 to 2025
- Absolute $ Opportunity Analysis By Device , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Cerebral Arteries
- Coronary Arteries
- Peripheral Arteries
- Cerebral Arteries
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Device
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Device
- By Application
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Device
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Device
- By Application
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Device
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Device
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Device
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Device
- By Application
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Device
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Device
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Device
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Device
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Device
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Device
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Device
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Device
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Device
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Device
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Device
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Device
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Device
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Device
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Device
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Device
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Device
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Device
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Device
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Device
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Device
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Device
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Device
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Device
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Device
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Device
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Device
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Device
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Device
- By Application
- Competition Analysis
- Competition Deep Dive
- AngioDynamics
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Terumo Co.
- Johnson and Johnson
- ECKOS Co.
- Bayer HealthCare LLC
- Boston Scientific Co.
- Argon Medical Devices
- AngioDynamics
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Device , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Device , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Device , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Device , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Device , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Device , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Device , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Device , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Device , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Device , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Device
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Device , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Device , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Device
- Figure 23: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Application
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Device , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Device , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Device
- Figure 30: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Application
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Device , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Device , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Device
- Figure 37: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Application
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Device , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Device , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Device
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Application
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Device , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Device , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Device
- Figure 51: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Application
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Device , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Device , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Device
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Device , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Device , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Device
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

