The polycarbonate regranulate from hemodialysis and blood purification devices market is valued at USD 144 million in 2026 and is projected to reach USD 408.9 million by 2036, reflecting a CAGR of 11.00%. Growth is driven by rising medical waste volumes and stricter regulations encouraging circular use of high-value polycarbonate components. Feedstock primarily comprises hemodialysis device scrap, blood purification housings, post-use single-service filters, and manufacturing off-cuts. Chemical recycling dominates at 45% share, supported by solvent-based purification and supercritical fluid depolymerization for high-purity outputs. Mechanical regranulation handles lower-contaminant off-cuts. Cost structures are influenced by process complexity, sterilization requirements, and certification overhead. Margin concentration favors operators capable of delivering validated medical-grade regranulates with traceability, rather than simply processing volume.
Regional adoption reflects infrastructure readiness, medical device manufacturing density, and regulatory enforcement. Western Europe and North America lead in collection and recycling integration, while other regions adopt more gradually due to logistical and regulatory barriers. Companies capturing high value include Eastman, Covestro, Teijin, Celanese, Veolia, Suez, and MBA Polymers, leveraging proprietary technologies, feedstock access, and multi-application certification. Value capture correlates with secure feedstock streams, validated processes, and the ability to supply diverse medical-grade applications including reprocessed device components and housings. Operators focusing solely on throughput face lower margins, while those combining compliance, technology, and traceable supply achieve sustainable returns throughout the forecast period.
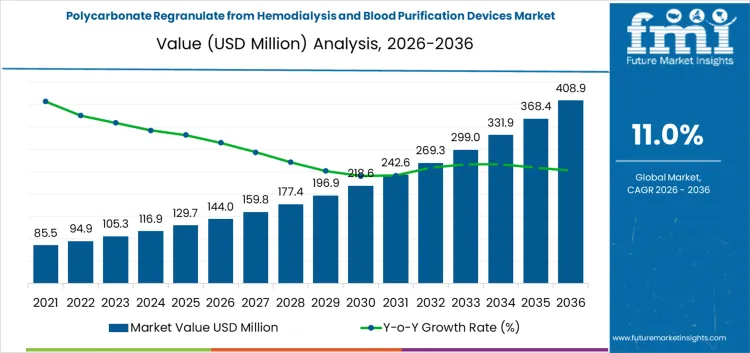
Between 2026 and 2031, the polycarbonate regranulate from hemodialysis and blood purification devices market is projected to grow from USD 144 million to USD 236 million, generating an absolute increase of USD 92 million and accounting for roughly 34.7% of total decade growth. This phase is shaped by early adoption of regranulation processes for single-use blood purification devices, driven by hospital sustainability initiatives and regulatory encouragement for circular medical plastics. Cost structures are influenced by decontamination, shredding, and material recovery technologies. Value capture favors operators providing validated, high-purity polycarbonate output suitable for medical applications rather than focusing on bulk volume.
From 2031 to 2036, the market is expected to expand from USD 236 million to USD 408.9 million, adding USD 172.9 million and representing 65.3% of decade growth. Growth accelerates as recycling processes scale and regranulated polycarbonate becomes fully integrated into medical device manufacturing. Margins increasingly favor firms controlling feedstock sourcing, sterilization, and quality assurance. Competitive advantage shifts toward operators offering traceable, compliant materials with consistent performance, while late entrants focus on operational efficiency, process optimization, and certification to capture incremental market share.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 144 million |
| Forecast Value (2036) | USD 408.9 million |
| Forecast CAGR (2026-2036) | 11.00% |
The polycarbonate regranulate market for hemodialysis and blood purification devices is shaped by regulatory requirements, polymer performance retention, and clinical waste management pressures. Historically, spent devices were incinerated or landfilled due to contamination risks, variable additive content, and sterilization challenges. Current recycling approaches focus on controlled collection, thorough decontamination, and extrusion processes that preserve polycarbonate mechanical and thermal properties suitable for non-critical medical applications. Hospitals, device manufacturers, and specialized recyclers collaborate to ensure traceable feedstock handling and compliance with healthcare regulations. Demand is driven by operational cost reduction, material circularity initiatives, and the need to document safe polymer reuse rather than purely volume based recovery, with process reliability and polymer integrity serving as primary value metrics.
Future expansion depends on standardized sterilization protocols, regulatory acceptance, and secure logistics for clinical waste streams rather than voluntary sustainability programs. Compared with early experimental programs, contemporary operations prioritize reproducible material properties, controlled additive management, and validated decontamination procedures. Energy consumption, contamination risk, and yield efficiency determine cost structures and concentrate margins among operators with integrated quality management systems. Hospitals and manufacturers adopt regranulate where it meets documented compliance and mechanical performance criteria. By 2036, polycarbonate regranulate from hemodialysis and blood purification devices will be embedded within controlled circularity programs, supporting regulatory alignment, operational efficiency, and safe material recovery in healthcare environments.
The demand for polycarbonate regranulate from hemodialysis and blood purification devices is segmented by feedstock source and recycling technology. Feedstock sources include hemodialysis device scrap, blood purification housings, post use single service filters, and manufacturing off cuts. Recycling technologies include chemical recycling, solvent based purification, supercritical fluid depolymerization, and mechanical regranulation. Segment selection is influenced by regulatory requirements for medical grade material, polymer property retention, and contamination control. Adoption is shaped by traceability, sterility assurance, and the ability to produce consistent regranulate suitable for downstream production rather than cost optimization or volume alone.

Hemodialysis device scrap accounts for approximately 48% of total feedstock demand, making it the leading source. This reflects both the high volume of polycarbonate in dialysis components and the uniformity of the material. Devices are collected and sterilized prior to recycling to meet medical grade compliance. Feedstock homogeneity simplifies processing and ensures consistent mechanical and thermal properties in the regranulate. Operators favor hemodialysis scrap because it provides a reliable source of high quality polycarbonate with minimal variability across batches.
Demand for hemodialysis device scrap is driven by its availability, material consistency, and regulatory traceability. Collection programs at hospitals and treatment centers enable predictable supply. Scrap quality supports chemical or supercritical recycling processes, ensuring polymer clarity and strength for reuse. The segment maintains leadership because it allows recyclers to generate validated regranulate suitable for critical medical applications, meeting both safety and performance standards. Integration with existing medical waste streams further reinforces its adoption.
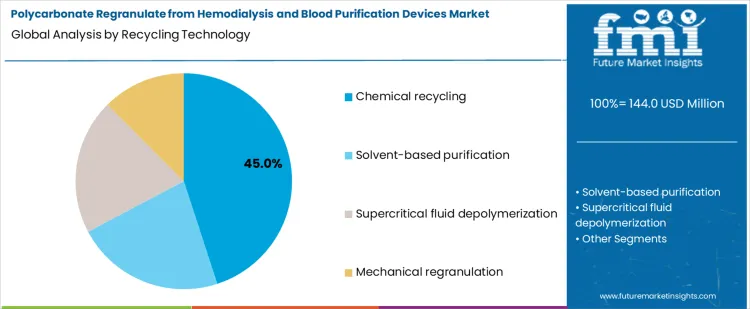
Chemical recycling represents approximately 45% of total technology adoption, making it the dominant route. This position reflects its ability to depolymerize polycarbonate into monomers while preserving chemical integrity for high grade reuse. Chemical recycling enables removal of contaminants, residual sterilization chemicals, and additives that mechanical reprocessing cannot fully address. Operators use controlled reaction conditions to produce consistent regranulate suitable for medical applications.
Demand for chemical recycling is shaped by regulatory requirements and material performance needs. Medical grade regranulate must meet stringent clarity, strength, and purity standards. Chemical recycling provides the highest level of control over these parameters compared with mechanical or solvent based methods. Adoption is reinforced in high value applications, where compliance, traceability, and polymer property retention outweigh throughput or capital cost considerations. The segment leads because it delivers validated, reproducible polycarbonate suitable for reuse in critical healthcare devices.
The polycarbonate regranulate market from hemodialysis and blood purification devices is shaped by stringent medical safety regulations and feedstock traceability requirements. Adoption focuses on processing spent devices while ensuring polymer integrity and removal of biological contaminants. Hospitals, device manufacturers, and specialized recyclers prioritize validated decontamination and quality control to comply with local health authority standards. Growth is concentrated where regulatory frameworks permit controlled recycling of single-use medical polymers. Procurement decisions emphasize certified process performance, batch consistency, and audit readiness rather than cost or volume expansion. Expansion reflects regulatory alignment rather than overall device consumption growth.
Demand is propelled by country-specific medical device waste management rules that require secure handling and sterilization of used hemodialysis and blood purification equipment. Recyclers must demonstrate process validation, bioburden removal, and polymer property retention to satisfy regulatory bodies. Device manufacturers and hospitals prefer suppliers providing traceable, compliant regranulate. Local authorities reward documented process control and third-party verification, creating a strong incentive for dedicated recycling lines. Investment is focused on compliance and risk mitigation rather than cost efficiency. This driver ensures recycled polycarbonate can be reused safely in approved industrial or non-critical applications under regional regulatory frameworks.
Adoption faces technical, operational, and regulatory constraints. Sterilization and decontamination processes must achieve near complete pathogen removal without degrading polymer properties. Feedstock collection from medical facilities is complex due to infection control and logistics. High capital expenditure for dedicated recycling lines limits participation to larger operators. Certification timelines and varying country regulations slow commercial deployment. Yield variability and contamination risk further challenge operational consistency. These barriers restrict adoption to specialized recyclers with technical expertise and institutional collaboration, preventing widespread integration despite growing interest in sustainable handling of single-use medical device polymers.
Trends emphasize advanced sterilization techniques, solvent or thermal purification, and continuous quality monitoring to meet regulatory standards. Collaborative models between hospitals, device manufacturers, and recyclers secure feedstock and streamline compliance documentation. Pilot scale validation increasingly precedes industrial deployment to demonstrate reproducibility and safety. Digital traceability ensures batch-level certification for audits. Investments focus on reproducibility, safety, and regulatory alignment rather than throughput or cost optimization. Partnerships and process standardization are essential to unlocking material reuse, enabling safe deployment of polycarbonate regranulate from hemodialysis and blood purification devices while maintaining compliance with national healthcare and medical device regulations.
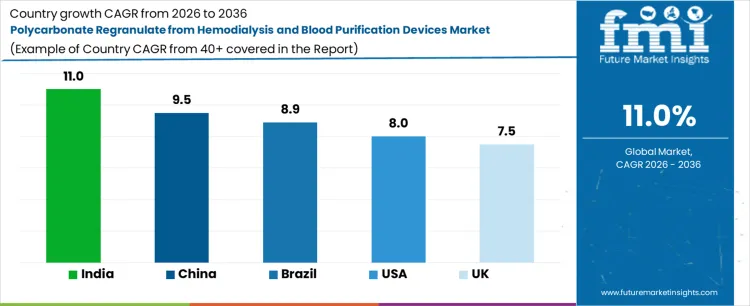
| Country | CAGR (%) |
|---|---|
| USA | 8.0 |
| UK | 7.5 |
| China | 9.5 |
| India | 11.0 |
| Brazil | 8.9 |
Demand for polycarbonate regranulate from hemodialysis and blood purification devices is growing as healthcare facilities and device manufacturers seek to recover high-value medical-grade polymers. India leads at 11.0% CAGR, driven by rapid expansion of dialysis centers, increasing patient volumes, and adoption of regranulation processes to reduce material costs while maintaining compliance. China follows at 9.5%, supported by large-scale production of medical disposables and integrated recycling initiatives. Brazil shows 8.9% growth, reflecting rising use of recycled polycarbonate in medical device manufacturing and secure material handling protocols. The USA grows at 8.0%, driven by regulatory-compliant recycling of single-use devices and supply chain optimization. The UK posts 7.5%, shaped by steady adoption of regranulation technologies in private and public healthcare systems.
The United States polycarbonate regranulate market from hemodialysis and blood purification devices is growing at a CAGR of 8.0%, supported by hospital sustainability programs and initiatives targeting medical device waste reduction. Recyclers are implementing controlled separation, cleaning, and sterilization processes to recover high-quality polycarbonate suitable for non-critical reuse or industrial applications. Demand is concentrated among hospitals, certified recycling operators, and medical device manufacturers seeking documented material performance. Investments focus on process validation, contamination control, and traceability rather than rapid capacity expansion. Growth reflects strategic recovery for selected device components while ensuring regulatory and biocompatibility compliance.
In the United Kingdom, the polycarbonate regranulate market from hemodialysis and blood purification devices is expanding at a CAGR of 7.5%, driven by healthcare regulations and environmental standards. Recycling operators focus on controlled cleaning, sterilization, and material verification to produce compliant regranulate. Demand is concentrated among hospitals, recycling service providers, and medical device manufacturers adhering to EU and national compliance frameworks. Investments prioritize process validation, consistent output quality, and traceable material flows rather than large-scale production. Growth reflects cautious integration into secondary applications where safety and documentation are critical.
China is experiencing strong growth in polycarbonate regranulate from hemodialysis and blood purification devices, with a CAGR of 9.5%, supported by expanding hospital infrastructure and increasing awareness of medical waste recycling. Recyclers are deploying solvent cleaning, sterilization, and quality control processes to recover polycarbonate suitable for industrial reuse. Demand is concentrated in urban healthcare clusters and large hospital networks. Investments emphasize process standardization, material verification, and traceable documentation to meet domestic and international safety requirements. Growth is influenced by increasing adoption of circular healthcare practices and environmental regulations.
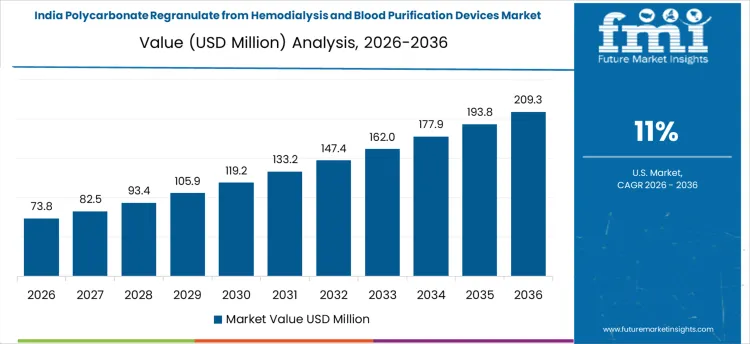
India shows rapid growth in polycarbonate regranulate from hemodialysis and blood purification devices, expanding at a CAGR of 11.0%, fueled by increasing hospital capacity and rising usage of disposable medical devices. Recyclers are implementing cleaning, sterilization, and quality verification processes to produce safe polycarbonate for non-critical or industrial applications. Demand is strongest among organized recyclers partnered with large hospital networks. Investments focus on process reliability, contamination control, and compliance alignment. Growth reflects both higher device volumes and the need for certified recycled feedstock.
Brazil is recording steady growth in polycarbonate regranulate from hemodialysis and blood purification devices at a CAGR of 8.9%, supported by improvements in hospital waste management and regulatory oversight. Recyclers are enhancing processes for cleaning, sterilization, and material verification to recover polycarbonate suitable for industrial applications. Demand is concentrated in urban healthcare facilities and hospitals engaged in recycling programs. Investment focuses on process standardization, contamination reduction, and traceable documentation rather than rapid expansion. Growth emphasizes safe material recovery and compliance with health and environmental standards.
The market for polycarbonate regranulate derived from hemodialysis and blood purification devices is developing in response to rising healthcare waste, cost sensitivity in treatment delivery, and growing interest in circular material systems. Hemodialysis and related blood purification devices contain polycarbonate components valued for clarity, impact resistance, and sterilization compatibility. Recycling these streams into regranulate requires robust sorting, decontamination, and quality control to meet stringent medical recycling criteria. Mechanical regranulation is common for high volume streams, but quality constraints prompt advanced purification in select applications. Demand emerges from device manufacturers, healthcare providers, and recyclers seeking validated pathways to reintroduce recovered polymer into non critical or indirectly contacted applications within the medical supply chain.
Market participants span polymer manufacturers, materials recyclers, and solution integrators focusing on process reliability and material performance. Eastman engages in polycarbonate regranulate production with controlled processing aimed at medical device derived feedstock. Covestro and Teijin contribute polymer expertise and conditioning technologies that support clarity and mechanical integrity in regranulate. Celanese focuses on sorting and reprocessing workflows that stabilize output quality. Recyclers such as Veolia, Suez, and MBA Polymers provide feedstock collection, decontamination, and mechanical regranulation capabilities, often in partnership with healthcare waste handlers. Competitive positioning in this market is defined by the ability to manage feedstock variability, achieve consistent material properties, and align supply with regulatory expectations for medical derived recyclates used in secondary or less critical applications.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD million |
| Feedstock Source | Hemodialysis device scrap, Blood purification housings, Post use single service filters, Manufacturing off cuts |
| Recycling Technology | Chemical recycling, Solvent based purification, Supercritical fluid depolymerization, Mechanical regranulation |
| Region | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Key Companies Profiled | Eastman, Covestro, Teijin, Celanese, Veolia, Suez, MBA Polymers |
| Additional Attributes | Dollar sales by feedstock source and recycling technology, medical-grade compliance and sterilization protocols, traceability and documentation, process reliability and yield efficiency, feedstock availability and contamination control, integration with medical device manufacturing, regulatory alignment with ISO/FDA standards |
How big is the polycarbonate regranulate from hemodialysis and blood purification devices market in 2026?
The global polycarbonate regranulate from hemodialysis and blood purification devices market is estimated to be valued at USD 144.0 million in 2026.
What will be the size of polycarbonate regranulate from hemodialysis and blood purification devices market in 2036?
The market size for the polycarbonate regranulate from hemodialysis and blood purification devices market is projected to reach USD 408.9 million by 2036.
How much will be the polycarbonate regranulate from hemodialysis and blood purification devices market growth between 2026 and 2036?
The polycarbonate regranulate from hemodialysis and blood purification devices market is expected to grow at a 11.0% CAGR between 2026 and 2036.
What are the key feedstock source types in the polycarbonate regranulate from hemodialysis and blood purification devices market?
The key feedstock source types in polycarbonate regranulate from hemodialysis and blood purification devices market are hemodialysis device scrap, blood purification housings, post‑use single‑service filters and manufacturing off‑cuts.
Which recycling technology segment to contribute significant share in the polycarbonate regranulate from hemodialysis and blood purification devices market in 2026?
In terms of recycling technology, chemical recycling segment to command 45.0% share in the polycarbonate regranulate from hemodialysis and blood purification devices market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.