The recycled content polycarbonate for non-critical medical and lab devices market is valued at USD 111 million in 2026 and is projected to grow at a CAGR of 10.9%, reflecting steady expansion toward USD 312.3 million by 2036. Growth is driven by increasing demand for sustainable materials in laboratory and non-critical medical applications, supported by post-consumer and post-industrial polycarbonate streams. Product grades include blended recycled, enhanced purity, and standard post-consumer resins. Cost structures reflect feedstock collection, reprocessing complexity, and quality validation, while margin concentration favors operators able to deliver consistent, verified outputs rather than focusing solely on volume throughput.
Between 2026 and 2036, adoption varies by region according to laboratory density, medical device production, and regulatory alignment. Application focus spans non-critical medical housings, laboratory instrument components, benchtop equipment frames, and non-sterile labware. Key companies, including Veolia, Covestro, Eastman, Celanese, Teijin, and SABIC, capture value by integrating feedstock security, process standardization, and quality documentation. Operators prioritizing volume over compliance face weaker returns, whereas those delivering verified, consistent recycled content achieve concentrated margins and sustainable, long-term revenue streams across the forecast period.
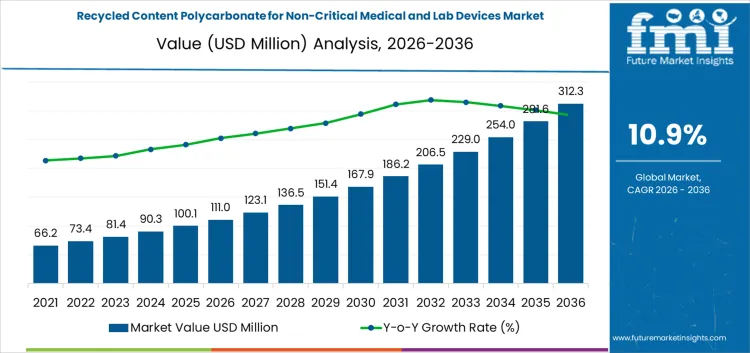
Between 2026 and 2031, the recycled content polycarbonate for non-critical medical and lab devices market is projected to grow from USD 111 million to USD 183 million, generating an absolute increase of USD 72 million and accounting for approximately 35.8% of total decade growth. Early-stage growth is driven by adoption of recycled polycarbonate in non-critical medical and laboratory devices, regulatory encouragement for circular medical plastics, and investment in decontamination and regranulation processes. Cost structures are influenced by sterilization, material handling, and quality assurance. Value capture favors operators delivering certified, traceable polycarbonate streams rather than prioritizing volume throughput.
From 2031 to 2036, the market is expected to expand from USD 183 million to USD 312.3 million, adding USD 129.3 million and representing roughly 64.2% of decade growth. Growth accelerates as recycled polycarbonate becomes integrated into mainstream non-critical medical and lab devices. Margins increasingly favor operators controlling feedstock sourcing, process reliability, and regulatory compliance. Competitive advantage shifts toward firms providing traceable, validated recycled polycarbonate, while late entrants focus on operational efficiency, standardization, and certification to capture incremental market share.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 111 million |
| Forecast Value (2036) | USD 312.3 million |
| Forecast CAGR (2026 to 2036) | 10.9% |
Recycled content polycarbonate for non-critical medical and lab devices is increasingly adopted to meet sustainability targets, regulatory guidance, and cost efficiency objectives. Historically, polycarbonate from medical devices and laboratory equipment was discarded due to contamination risk, variable additive content, and degradation during reprocessing.
Current processes focus on controlled collection, validated sterilization, and extrusion techniques that preserve mechanical and thermal properties suitable for housings, connectors, and non-critical components. Hospitals, laboratories, and device manufacturers prioritize feedstock traceability, reproducible polymer performance, and documented compliance. Value capture is concentrated on operational efficiency, regulatory alignment, and verified material reuse rather than volume throughput. Pilot programs inform best practices for decontamination, process control, and quality assurance, guiding broader implementation.
Future growth depends on regulatory support, institutional procurement policies, and circularity commitments rather than voluntary adoption alone. Compared with earlier experimental approaches, contemporary operations emphasize lot-to-lot consistency, controlled additive management, and validated sterilization processes.
Cost sensitivity is influenced by energy consumption, feedstock handling, and yield recovery, concentrating margins among operators capable of delivering certified polymer at scale. Adoption is strongest where recycled polycarbonate meets non-critical device specifications, maintains mechanical performance, and supports audit readiness. By 2036, recycled content polycarbonate is expected to be a standard solution for non-critical medical and laboratory device components, combining sustainability, compliance, and operational reliability.
The demand for recycled content polycarbonate (PC) in non-critical medical and laboratory devices is segmented by product grade and application. Product grades include post-consumer recycled PC, postindustrial recycled PC, blended recycled fractions, and enhanced purity recycled PC. Applications cover non-critical medical device housings, laboratory instrument components, benchtop equipment frames, and non-sterile labware and accessories. Segment selection is influenced by polymer quality, regulatory requirements, and functional suitability. Adoption is primarily driven by the ability to maintain mechanical properties, clarity, and durability while integrating recycled content. Material traceability, processing compatibility, and operational feasibility guide uptake rather than cost optimization.
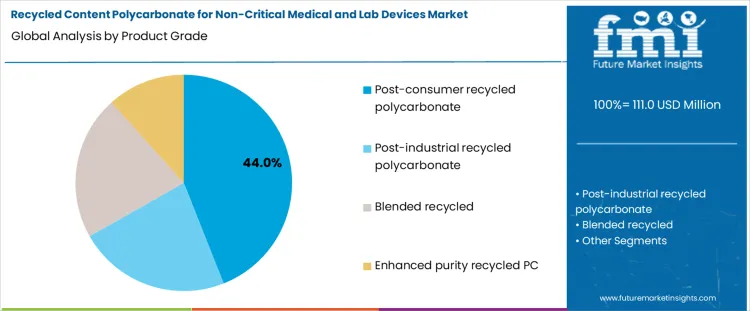
Post-consumer recycled PC accounts for approximately 44% of total product grade demand, making it the leading category. This share reflects its availability, traceable origin, and ability to meet quality requirements for non-critical applications. Recyclers process post-consumer material through washing, shredding, and regranulation to produce consistent output suitable for device housings and laboratory components. Operators favor this grade because it provides predictable mechanical performance and transparency, allowing manufacturers to integrate recycled content without compromising function or regulatory compliance.
Demand for post-consumer recycled PC is shaped by material reliability and sustainability objectives. Manufacturers require consistent melt flow, strength, and clarity for housings, frames, and benchtop instruments. Post-consumer feedstock enables demonstrable circularity while supporting ISO and quality management standards. The segment leads because it balances recycled content integration with operational feasibility, offering a validated pathway for producing non-critical medical and laboratory components from recovered polycarbonate.
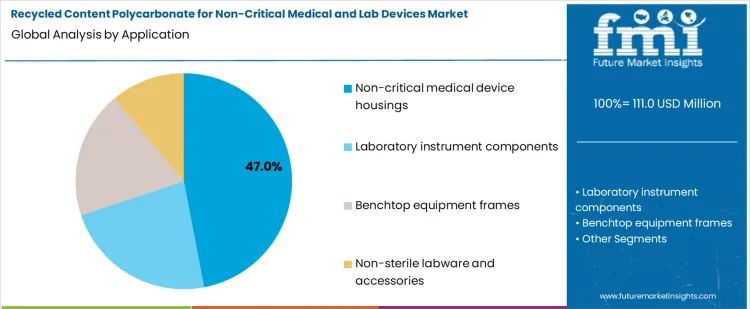
Non-critical medical device housings account for approximately 47% of total application demand, making them the largest segment. This reflects their high usage volume and moderate regulatory requirements compared with sterile or critical devices. Recycled polycarbonate in housings must maintain structural integrity, dimensional accuracy, and chemical resistance to support repeated handling and routine cleaning.
Demand in non-critical housings is driven by the combination of volume, process compatibility, and regulatory feasibility. Manufacturers favor applications where recycled content can be integrated without complex sterilization or performance validation challenges. Segmented recycling streams ensure consistent quality, traceability, and material performance. The segment leads because non-critical housings provide a high volume, low risk opportunity for incorporating recycled polycarbonate, supporting sustainability objectives while meeting functional requirements for medical and laboratory environments.
Recycled content polycarbonate for non-critical medical and lab devices is increasingly adopted in components such as casings, trays, and housings where sterilization and biocompatibility risks are lower. Adoption is driven by regulatory allowances for non-critical applications and institutional sustainability initiatives. Manufacturers prioritize polymer consistency, chemical resistance, and dimensional stability to ensure reliable performance. Growth is strongest in regions with defined recycled content targets and waste reduction mandates. Investment decisions emphasize process validation and traceable quality rather than throughput or cost alone. Regulatory alignment and operational reliability drive procurement for safe integration in non-critical healthcare and laboratory components.
Demand is influenced by country-specific healthcare and laboratory standards permitting recycled polymers in non-critical applications. Hospitals, labs, and device manufacturers seek materials that meet mechanical and chemical specifications while supporting circularity objectives. Supplier-provided documentation and batch traceability reduce audit risk and facilitate compliance with local regulations. Institutional sustainability initiatives encourage integration of recycled content, creating preference for certified material streams. The driver is regulatory certainty and environmental stewardship rather than cost advantage. Facilities capable of delivering consistent, traceable recycled polycarbonate gain stronger positioning among device manufacturers and institutional purchasers operating under sustainability mandates.
Barriers include feedstock variability, maintaining dimensional and chemical stability, and ensuring traceable quality for audits. Smaller recyclers may lack capacity to deliver certified, consistent batches. Process validation and documentation increase operational complexity and cost. Some laboratories and device manufacturers require pre-approval testing before integrating recycled material, extending timelines. Regional differences in acceptance criteria or recycled content guidelines further constrain adoption. These factors limit uptake to operators with technical expertise and established certification processes, concentrating use in well-resourced institutions and manufacturers with formal sustainability and compliance programs.
Trends emphasize modular processing systems, quality monitoring, and batch-level traceability to maintain polymer performance and compliance. Collaboration between recyclers, manufacturers, and institutional purchasers secures reliable feedstock and supports regulatory documentation. Pilot-scale validation allows demonstration of reproducibility and safety prior to full-scale deployment. Digital systems provide audit-ready records for batch certification. Focus is on operational reliability and regulatory alignment rather than throughput or cost efficiency. These collaborative models enable safe integration of recycled polycarbonate in non-critical medical and laboratory devices while meeting institutional sustainability targets and local compliance requirements.
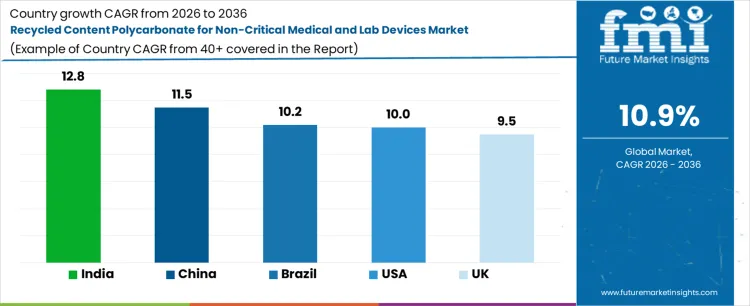
| Country | CAGR (%) |
|---|---|
| USA | 10.0% |
| UK | 9.5% |
| China | 11.5% |
| India | 12.8% |
| Brazil | 10.2% |
Demand for recycled content polycarbonate in non-critical medical and lab devices is rising as manufacturers aim to reduce material costs and improve sustainability without compromising performance. India leads with a 12.8% CAGR, driven by growing hospital and laboratory infrastructure, increasing device volumes, and adoption of recycled polycarbonate in non-critical applications such as instrument housings and lab trays. China follows at 11.5%, supported by large-scale device production and regulatory acceptance of recycled content for non-critical components. Brazil records 10.2% growth, reflecting adoption in private and public laboratories. The USA grows at 10.0%, shaped by manufacturer initiatives for cost optimization and circularity programs. The UK shows 9.5% CAGR, reflecting steady integration of recycled polycarbonate into non-critical lab and medical devices.
In the United States, the recycled content polycarbonate market for non-critical medical and lab devices is growing at a CAGR of 10%, supported by hospitals and manufacturers integrating recycled materials to meet sustainability goals and regulatory expectations. Recyclers are deploying cleaning, sterilization, and quality verification processes to produce high-quality polycarbonate suitable for non-critical components. Demand is concentrated among certified recyclers, OEMs, and hospital networks seeking traceable, compliant feedstock. Investments focus on process validation, contamination control, and consistency rather than rapid capacity expansion. Growth reflects strategic substitution for non-critical applications while maintaining material safety and operational reliability.
In the United Kingdom, recycled content polycarbonate for non-critical medical and lab devices is expanding at a CAGR of 9.5%, driven by healthcare regulations and environmental policies promoting safe use of recovered plastics. Recyclers and OEMs implement validated cleaning, sterilization, and verification processes to produce material suitable for non-critical applications. Demand is concentrated among hospitals, certified recyclers, and device manufacturers complying with EU and national standards. Investments prioritize process standardization, traceability, and reproducibility rather than large-scale throughput expansion. Growth reflects careful integration of recycled polycarbonate into existing device lines.
China is witnessing strong growth in recycled content polycarbonate for non-critical medical and lab devices, with a CAGR of 11.5%, fueled by hospitals and manufacturers aligning with international sustainability and compliance expectations. Recyclers implement advanced cleaning, sterilization, and verification processes to recover polycarbonate suitable for non-critical applications. Demand is concentrated in urban hospital clusters and industrial regions producing export-oriented devices. Investments focus on process reliability, standardization, and traceable outputs. Growth is influenced by global regulatory requirements and brand sourcing expectations alongside domestic initiatives.
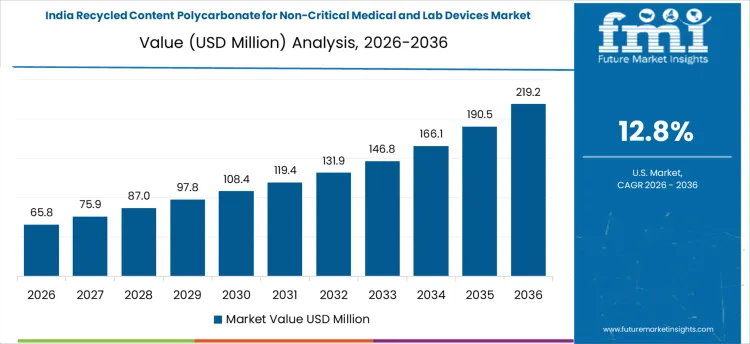
India shows the fastest growth in recycled content polycarbonate for non-critical medical and lab devices, expanding at a CAGR of 12.8%, driven by increasing hospital capacity and single-use medical device consumption. Recyclers are deploying cleaning, sterilization, and verification processes to reclaim polycarbonate for non-critical applications. Demand is concentrated among organized recyclers and hospital networks aligned with OEMs. Investments prioritize process reliability, contamination control, and certification. Growth reflects both higher device volumes and the need to supply certified recycled feedstock for domestic and export applications.
Brazil is recording steady growth in recycled content polycarbonate for non-critical medical and lab devices at a CAGR of 10.2%, supported by improvements in hospital waste management and regulatory enforcement. Recyclers are implementing cleaning, sterilization, and verification processes to recover high-quality polycarbonate suitable for non-critical applications. Demand is concentrated in urban hospitals and certified recyclers. Investments focus on process standardization, contamination control, and traceable documentation rather than rapid capacity expansion. Growth reflects gradual integration of recycled polycarbonate into non-critical medical and lab device applications.
Competition in the recycled content polycarbonate market for non-critical medical and laboratory devices centers on material quality, processing pathways, and alignment with device performance requirements. Veolia supports downstream supply by integrating medical plastic waste collection with sorting, decontamination, and recycling operations that deliver polymer streams suitable for non-critical device components. Covestro AG competes through advanced purification and conditioning technologies that enhance recycled polycarbonate clarity and mechanical consistency for applications where failure risk is low but reliability remains important. Eastman focuses on tailored recycling workflows and quality assurance frameworks that stabilize material properties for lab equipment housings, secondary instrument parts, and non-sterile components.
Other participants differentiate through polymer expertise and feedstock integration. Celanese develops controlled reprocessing systems that stabilize mixed feedstock and improve output uniformity, appealing to converters serving non-critical segments. Teijin applies solvent-level purification and conditioning to maintain product performance within targeted specifications. SABIC brings global polycarbonate knowledge and recycling partnerships that support scalable adoption across regions. Competitive differences arise in how firms balance processing complexity with cost, manage quality assurance for recycled content specifications, and integrate traceability systems that support customer confidence. Providers that combine consistent performance, documentation aligned with non-critical use cases, and secure feedstock channels tend to secure stronger positioning in this segment.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD million |
| Product Grade | Post-consumer recycled polycarbonate, Post-industrial recycled polycarbonate, Blended recycled, Enhanced purity recycled PC |
| Application | Non-critical medical device housings, Laboratory instrument components, Benchtop equipment frames, Non-sterile labware and accessories |
| Region | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Key Countries Covered | China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, United States, Canada, Mexico, Brazil, Chile, Kingdom of Saudi Arabia, Other GCC Countries, Turkey, South Africa, Other African Union |
| Key Companies Profiled | Veolia, Covestro, Eastman, Celanese, Teijin, SABIC |
| Additional Attributes | Dollar sales by product grade and application, feedstock traceability, regulatory compliance metrics, audit-ready documentation, operational efficiency, integration with healthcare and laboratory supply chains, ISO/FDA compliance, validated polymer recovery, multi-stream capability |
How big is the recycled content polycarbonate for non-critical medical and lab devices market in 2026?
The global recycled content polycarbonate for non-critical medical and lab devices market is estimated to be valued at USD 111.0 million in 2026.
What will be the size of recycled content polycarbonate for non-critical medical and lab devices market in 2036?
The market size for the recycled content polycarbonate for non-critical medical and lab devices market is projected to reach USD 312.3 million by 2036.
How much will be the recycled content polycarbonate for non-critical medical and lab devices market growth between 2026 and 2036?
The recycled content polycarbonate for non-critical medical and lab devices market is expected to grow at a 10.9% CAGR between 2026 and 2036.
What are the key product grade in the recycled content polycarbonate for non-critical medical and lab devices market?
The key product grade in recycled content polycarbonate for non-critical medical and lab devices market are post‑consumer recycled polycarbonate, post‑industrial recycled polycarbonate, blended recycled and enhanced purity recycled PC.
Which application segment will contribute significant share in the recycled content polycarbonate for non-critical medical and lab devices market in 2026?
In terms of application, non‑critical medical device housings segment to command 47.0% share in the recycled content polycarbonate for non-critical medical and lab devices market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.