Reprocessed Medical Devices Market
This report offers a thorough overview of the reprocessed medical devices market, spanning market size analysis, revenue projections, competitive landscape assessment, product type trends, demand dynamics, key drivers, restraints, supply chain evaluation, and growth opportunity mapping.
Reprocessed Medical Devices Market Size, Market Forecast and Outlook By FMI
The reprocessed medical devices market was valued at USD 2.30 billion in 2025, projected to reach USD 2.69 billion in 2026, and is forecast to expand to USD 12.94 billion by 2036 at a 17.00% CAGR. Cost containment pressures across hospital networks and ambulatory surgical centers are accelerating the institutional adoption of reprocessed single-use devices, particularly in cardiac catheterization suites and laparoscopic procedure theatres. Medical device reprocessing has transitioned from a niche cost-saving initiative to a compliance-integrated supply chain strategy embedded in group purchasing organization contract cycles, a shift directly tied to waste reduction mandates and facility-level sustainability targets.
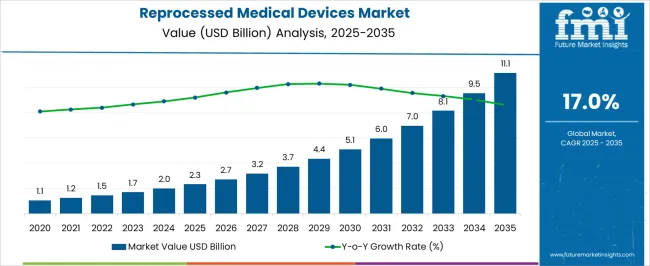
Regulatory harmonization efforts between the USA FDA and European MDR frameworks are expanding the cleared device categories eligible for reprocessing, creating new addressable volumes in electrophysiology catheters and ultrasonic scalpels. Hospital supply chain directors now evaluate reprocessing compatibility as a pre-qualification criterion when onboarding new single-use device vendors. The convergence of medical waste management protocols and value analysis committee procurement mandates positions reprocessing as a structural operating expense reduction lever rather than a discretionary initiative.
China sets the pace at a 22.9% CAGR, driven by national hospital procurement reform campaigns mandating cost reduction benchmarks for Class II consumables. India follows with 19.8% growth as state-level health mission budgets prioritize device reuse to extend tertiary care coverage. Brazil advances at 16.5% with Anvisa regulatory alignment enabling broader reprocessing clearances. USA operations expand at 15.3% supported by CMS payment bundling incentives that reward lower per-procedure device costs. Germany registers 14.7% growth under medical device regulation compliance timelines that include reprocessing pathway validation. UK maintains 14.1% expansion anchored by NHS sustainability framework targets. Japan tracks at 13.8% driven by MHLW device approval pathway updates for reprocessed cardiovascular consumables.
Summary of Reprocessed Medical Devices Market
- Reprocessed Medical Devices Market Definition
- Reprocessed medical devices are single-use instruments restored through validated reprocessing procedures to meet original performance standards, spanning cardiovascular, laparoscopic, orthopedic, and general surgical device categories.
- Demand Drivers in the Market
- Hospital group purchasing organizations increasingly mandate reprocessing-compatible device specifications as a pre-condition for vendor contract awards, compressing per-procedure consumable budgets by 40% to 60% on eligible categories.
- Regulatory pathway expansion under FDA supplemental 510(k) clearances and EU MDR Annex VIII reprocessing validations is widening the addressable device portfolio beyond traditional electrophysiology catheters into ultrasonic dissection and endoscopic accessories.
- Facility-level medical waste diversion targets, linked to Joint Commission sustainability metrics and NHS Green Plan benchmarks, are converting reprocessing from a discretionary savings initiative into a compliance obligation.
- Key Segments Analyzed in the FMI Report
- Cardiovascular Devices by Product Type: 35.4% share in 2026, reflecting the high unit cost of electrophysiology catheters and ablation devices that generate the largest per-unit reprocessing savings.
- Hospitals by End User: 50.2% share in 2026, as integrated delivery networks consolidate reprocessing contracts under system-wide group purchasing agreements.
- China: 22.9% compound growth driven by national hospital procurement reform campaigns mandating cost reduction benchmarks for Class II medical consumables.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Consultant for Healthcare observe that the reprocessed medical devices sector is entering a structural inflection where regulatory clearance expansion, group purchasing mandate integration, and sustainability compliance convergence are compressing the adoption decision cycle from multi-year evaluation to single-budget-cycle implementation. Hospital supply chain directors who delay reprocessing contract integration face compounding cost disadvantages as peer networks lock in volume-tiered pricing structures. The critical procurement risk centers on cleared device category breadth, as facilities operating narrow reprocessing portfolios forfeit the per-procedure savings available across broader consumable baskets.
- Strategic Implications / Executive Takeaways
- Hospital supply chain directors must integrate reprocessing compatibility as a mandatory specification in new single-use device vendor onboarding criteria to capture full per-procedure savings across expanding cleared categories.
- Reprocessing service providers must secure validated clearance pathways for next-generation ultrasonic and energy-based surgical devices before competitor facilities establish exclusivity agreements with health system networks.
- Group purchasing organization contract architects must embed reprocessing volume tiers into multi-year device supply agreements to prevent hospitals from reverting to full-price single-use procurement during contract renewal cycles.
Reprocessed Medical Devices Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 2.69 Billion |
| Industry Value (2036) | USD 12.94 Billion |
| CAGR (2026-2036) | 17.00% |
Source: Future Market Insights, 2026
Reprocessed Medical Devices Market Definition
Reprocessed medical devices encompass single-use medical instruments that undergo validated cleaning, functional testing, sterilization, and repackaging procedures to restore them to original equipment manufacturer performance specifications for subsequent clinical use. These devices span interventional cardiology catheters, laparoscopic trocars and graspers, orthopedic surgical tools, gastroenterology accessories, and general surgical instruments processed through FDA-cleared or CE-marked reprocessing facilities.
Reprocessed Medical Devices Market Inclusions
Market scope includes third-party reprocessed single-use devices, original equipment manufacturer take-back reprocessing programmes, hospital in-house reprocessing operations, and contract reprocessing services. Regulatory clearance tracking across FDA 510(k) reprocessing submissions and EU MDR reprocessing pathway validations falls within defined boundaries.
Reprocessed Medical Devices Market Exclusions
Capital sterilization equipment, standalone autoclave machinery, and original single-use device manufacturing are excluded. Reusable surgical instrument refurbishment services and general hospital central sterile supply department operations fall outside analytical parameters.
Reprocessed Medical Devices Market Research Methodology
- Primary Research: Analysts engaged with hospital value analysis committee directors, group purchasing organization procurement leads, and reprocessing facility operations managers to map the decision gates triggering device reprocessing contract renewals.
- Desk Research: Data collection phases aggregated FDA 510(k) reprocessing clearance databases, EU MDR notified body reprocessing validation schedules, and state-level medical waste diversion reporting.
- Market-Sizing and Forecasting: Baseline values derive from a bottom-up aggregation of reprocessed device unit volumes multiplied by per-unit reprocessing fees, applying region-specific regulatory clearance expansion curves to project future adoption velocity.
- Data Validation and Update Cycle: Projections are tested against publicly reported hospital supply chain expenditure guidance from leading integrated delivery networks and group purchasing organizations.
Why is the Reprocessed Medical Devices Market Growing?
The Reprocessed Medical Devices market is experiencing robust growth, driven by the increasing need to reduce healthcare costs while maintaining high standards of patient care. Rising demand for sustainable medical practices, coupled with budgetary constraints in hospitals and healthcare facilities, has accelerated the adoption of reprocessed devices. These devices, which are sterilized and refurbished for safe reuse, offer significant cost advantages without compromising quality or compliance.
Growth is further supported by advancements in sterilization techniques, quality control processes, and regulatory frameworks that ensure safety and efficacy. Hospitals and healthcare providers are increasingly integrating reprocessed devices into surgical and diagnostic workflows, optimizing operational efficiency and reducing environmental impact.
The market is also benefiting from growing awareness among clinicians and procurement teams regarding the economic and ecological advantages of device reprocessing As healthcare systems focus on sustainable resource utilization, regulatory compliance, and cost containment, the Reprocessed Medical Devices market is expected to sustain long-term expansion, supported by continued technological innovation and widespread adoption in clinical settings.
Segmental Analysis
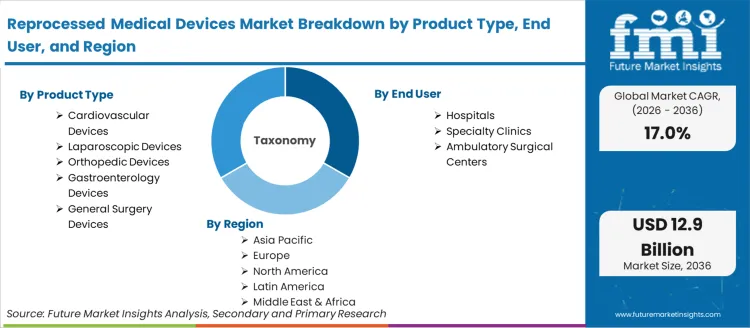
The reprocessed medical devices market is segmented by product type, end user, and geographic regions. By product type, reprocessed medical devices market is divided into Cardiovascular Devices, Laparoscopic Devices, Orthopedic Devices, Gastroenterology Devices, and General Surgery Devices. In terms of end user, reprocessed medical devices market is classified into Hospitals, Specialty Clinics, and Ambulatory Surgical Centers. Regionally, the reprocessed medical devices industry is classified into North America, Latin America, Western Europe, Eastern Europe, Balkan & Baltic Countries, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific, and the Middle East & Africa.
Insights into the Cardiovascular Devices Product Type Segment
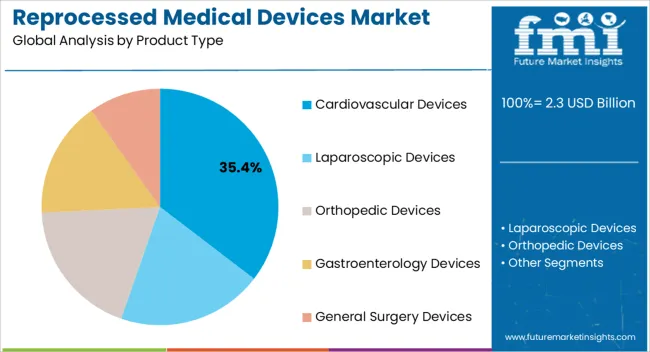
The cardiovascular devices segment is projected to hold 35.4% of the market revenue in 2026, establishing it as the leading product type. Growth in this segment is being driven by the high utilization of cardiovascular instruments in surgeries, interventional procedures, and diagnostic applications, combined with the cost savings achieved through reprocessing. Advanced sterilization and refurbishment processes ensure that device performance and patient safety are not compromised, increasing acceptance among hospitals and healthcare providers.
The segment benefits from rising cardiovascular disease prevalence and the growing number of procedures performed globally, which create a consistent demand for reprocessed devices. Hospitals prefer reprocessed cardiovascular instruments due to their ability to reduce operational expenses while maintaining quality standards.
Continuous improvements in regulatory oversight, quality assurance, and device tracking have further strengthened market confidence As cost efficiency and sustainability remain priorities for healthcare facilities, the cardiovascular devices segment is expected to maintain its leadership, supported by ongoing adoption and technological innovation in reprocessing methods.
Insights into the Hospitals End User Segment
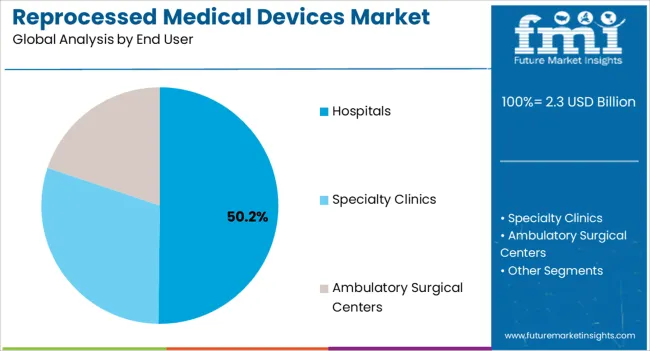
The hospitals segment is anticipated to account for 50.2% of the market revenue in 2026, making it the leading end-user category. Growth is being driven by hospitals’ need to manage operational costs effectively while ensuring patient safety and compliance with medical regulations. Reprocessed medical devices allow hospitals to achieve significant cost savings, particularly for high-volume and expensive devices such as cardiovascular, orthopedic, and surgical instruments.
The integration of reprocessing programs enables hospitals to optimize device utilization, reduce procurement expenses, and improve inventory management. Adoption is further supported by advancements in sterilization, quality control, and device tracking technologies, ensuring safety, reliability, and regulatory adherence.
Hospitals are increasingly recognizing the sustainability benefits of device reprocessing, which reduces medical waste and promotes environmental responsibility As healthcare facilities continue to balance budget constraints with patient care quality, the hospitals segment is expected to remain the largest revenue contributor, driven by ongoing implementation of reprocessing strategies and technological improvements in device refurbishment and monitoring.
Market Overview
Reprocessed Medical Devices Market Overview
Reprocessed medical devices are those devices are those devices that are refurbished and put to reuse again. The reprocessing of medical devices involves disinfection, cleaning, remanufacturing, testing, sterilization, packaging and labelling of the devices in order to put to service again.
The reprocessing of any medical device must follow a validated process which renders it fit for use. In order to reprocess the device factors like the efficacy, life cycle, potential risk to the patient and components used to manufacture the device are taken into consideration. With the increasing healthcare cost and high price of medical devices then demand for reprocessed medical devices is expected to witness high growth over the forecast period.
Analysis of Reprocessed Medical Devices Market By Key Countries
.webp)
| Country | CAGR |
|---|---|
| China | 22.9% |
| India | 21.2% |
| Germany | 19.5% |
| France | 17.8% |
| UK | 16.1% |
| USA | 14.4% |
| Brazil | 12.7% |
Country-wise Analysis
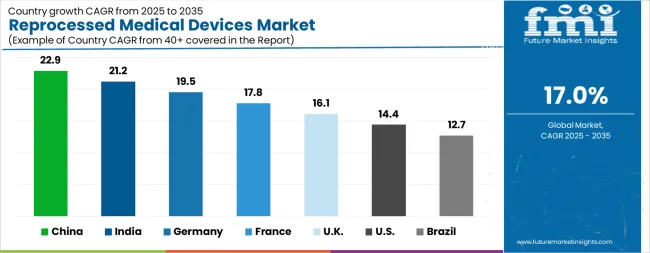
The Reprocessed Medical Devices Market is expected to register a CAGR of 17.0% during the forecast period, exhibiting varied country level momentum. China leads with the highest CAGR of 22.9%, followed by India at 21.2%. Developed markets such as Germany, France, and the UK continue to expand steadily, while the USA is likely to grow at consistent rates. Brazil posts the lowest CAGR at 12.7%, yet still underscores a broadly positive trajectory for the global Reprocessed Medical Devices Market. In 2025, Germany held a dominant revenue in the Western Europe market and is expected to grow with a CAGR of 19.5%. The USA Reprocessed Medical Devices Market is estimated to be valued at USD 834.5 million in 2026 and is anticipated to reach a valuation of USD 3.2 billion by 2036. Sales are projected to rise at a CAGR of 14.4% over the forecast period between 2026 and 2036. While Japan and South Korea markets are estimated to be valued at USD 111.9 million and USD 63.3 million respectively in 2026.
Key Players in the Reprocessed Medical Devices Market
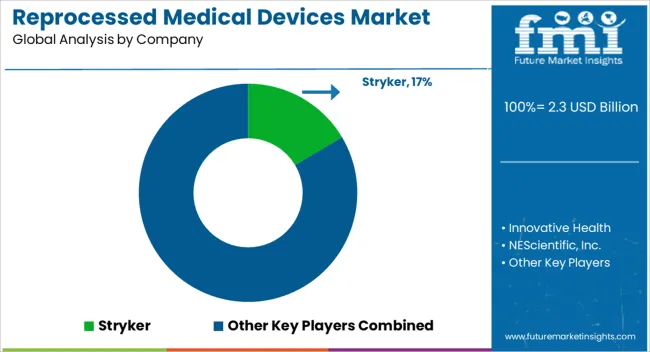
- Stryker
- Innovative Health
- NEScientific, Inc.
- Medline Industries, LP.
- Arjo
- Cardinal Health
- SureTek Medical
- Soma Tech Intl
- Johnson & Johnson MedTech
- GE Healthcare
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 2.69 Billion to USD 12.94 Billion, at a CAGR of 17.00% |
| Market Definition | Reprocessed medical devices are single-use instruments restored through validated reprocessing procedures to meet original performance standards, spanning cardiovascular, laparoscopic, orthopedic, and general surgical device categories. |
| Segmentation | Product Type: Cardiovascular Devices, Laparoscopic Devices, Orthopedic Devices, Gastroenterology Devices, and General Surgery Devices; End User: Hospitals, Specialty Clinics, and Ambulatory Surgical Centers |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | China, India, USA, Germany, UK, Brazil, Japan, and 40 plus countries |
| Key Companies Profiled | Stryker, Innovative Health, NEScientific, Inc., Medline Industries, LP., Arjo, Cardinal Health, SureTek Medical, Soma Tech Intl, Johnson & Johnson MedTech, GE Healthcare |
| Forecast Period | 2026 to 2036 |
| Approach | Forecasting models apply a bottom-up methodology starting with installed base metrics and projecting conversion rates to advanced analytics and technology adoption. |
Reprocessed Medical Devices Market by Segments
Product Type:
- Cardiovascular Devices
- Laparoscopic Devices
- Orthopedic Devices
- Gastroenterology Devices
- General Surgery Devices
End User:
- Hospitals
- Specialty Clinics
- Ambulatory Surgical Centers
Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- 1. USA Food and Drug Administration. (2024). Reprocessing of Single-Use Devices: Guidance for Industry and FDA Staff. FDA.
- 2. European Commission. (2024). Medical Devices Regulation (EU) 2017/745: Reprocessing Provisions Update. European Commission.
- 3. Association of Medical Device Reprocessors. (2024). Annual Industry Survey: Reprocessed Device Volumes and Savings Metrics. AMDR.
- 4. World Health Organization. (2024). Medical Device Reuse and Reprocessing: Global Regulatory Landscape Assessment. WHO.
- 5. National Health Service England. (2024). NHS Green Plan: Medical Device Sustainability Targets 2024-2028. NHS.
- 6. Agencia Nacional de Vigilancia Sanitaria. (2024). RDC 156/2024: Updated Framework for Medical Device Reprocessing in Brazil. Anvisa.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary research documentation.
Frequently Asked Questions
How large is the demand for Reprocessed Medical Devices in the global market in 2026?
Demand for reprocessed medical devices in the global market is estimated to be valued at USD 2.69 billion in 2026.
What will be the market size of Reprocessed Medical Devices by 2036?
Market size for reprocessed medical devices is projected to reach USD 12.94 billion by 2036.
What is the expected CAGR for the Reprocessed Medical Devices Market between 2026 and 2036?
Demand for reprocessed medical devices is expected to grow at a CAGR of 17.00% between 2026 and 2036.
Which Product Type is poised to lead global sales by 2026?
Cardiovascular Devices accounts for 35.4% share in 2026 as high unit-cost electrophysiology catheters and ablation devices generate the largest per-unit reprocessing savings across hospital procurement cycles.
How is Hospitals positioned as an End User segment in 2026?
Hospitals represents 50.2% of end user share in 2026, as integrated delivery networks consolidate reprocessing contracts under system-wide group purchasing agreements.
What is driving demand in China?
National hospital procurement reform campaigns mandating cost reduction benchmarks for Class II medical consumables fuel the fastest growth at 22.9% CAGR through 2036.
What is included in the scope of this report?
The market covers third-party reprocessed single-use devices, OEM take-back reprocessing programmes, hospital in-house reprocessing operations, and contract reprocessing services across cardiovascular, laparoscopic, orthopedic, gastroenterology, and general surgery device categories.
What is excluded from the scope of this report?
Capital sterilization equipment, standalone autoclave machinery, original single-use device manufacturing, and reusable surgical instrument refurbishment services are excluded.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Cardiovascular Devices
- Laparoscopic Devices
- Orthopedic Devices
- Gastroenterology Devices
- General Surgery Devices
- Cardiovascular Devices
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospitals
- Specialty Clinics
- Ambulatory Surgical Centers
- Hospitals
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Company
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Company, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Company, 2026 to 2036
- Stryker
- Innovative Health
- NEScientific, Inc.
- Medline Industries, LP.
- Arjo
- Cardinal Health
- SureTek Medical
- Soma Tech Intl
- Johnson & Johnson MedTech
- GE Healthcare
- Y to o to Y Growth Trend Analysis By Company, 2021 to 2025
- Absolute $ Opportunity Analysis By Company, 2026 to 2036
- Stryker
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By End User
- By Company
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End User
- By Company
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By End User
- By Company
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End User
- By Company
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By End User
- By Company
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End User
- By Company
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By End User
- By Company
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End User
- By Company
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By End User
- By Company
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End User
- By Company
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By End User
- By Company
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End User
- By Company
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By End User
- By Company
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End User
- By Company
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- By Company
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- By Company
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- By Company
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- By Company
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- By Company
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- By Company
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- By Company
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- By Company
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- By Company
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- By Company
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- By Company
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- By Company
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- By Company
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- By Company
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- By Company
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- By Company
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- By Company
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- By Company
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- By Company
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- By Company
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- By Company
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- By Company
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By End User
- By Company
- Competition Analysis
- Competition Deep Dive
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Company, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Company, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Company, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Company, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by Company, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by Company, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by Company, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by Company, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by End User
- Figure 9: Global Market Value Share and BPS Analysis by Company, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Company, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Company
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Product Type
- Figure 26: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by End User
- Figure 29: North America Market Value Share and BPS Analysis by Company, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Company, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Company
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Product Type
- Figure 36: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by End User
- Figure 39: Latin America Market Value Share and BPS Analysis by Company, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Company, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Company
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Product Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by End User
- Figure 49: Western Europe Market Value Share and BPS Analysis by Company, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by Company, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by Company
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by End User
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by Company, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by Company, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by Company
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Product Type
- Figure 66: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by End User
- Figure 69: East Asia Market Value Share and BPS Analysis by Company, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by Company, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by Company
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by Company, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by Company, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by Company
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by Company, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by Company, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by Company
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

