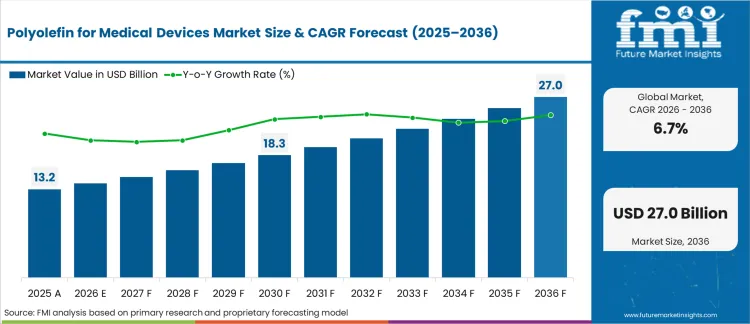
In 2025, the polyolefin for medical devices market was valued at USD 13,174.2 million. Based on Future Market Insights analysis, demand for polyolefin materials in medical devices is estimated to grow to USD 14,094.6 million in 2026 and USD 26,941.3 million by 2036. FMI projects a CAGR of 6.7% during the forecast period.
The market is expected to generate an absolute dollar growth of USD 12,846.7 million between 2026 and 2036. This reflects strong structural expansion driven by rising demand for disposable medical devices, drug delivery systems, and home-use healthcare products. Growth is supported by increasing preference for lightweight, sterilization-compatible, and cost-efficient materials such as PE, PP, EVA, and advanced olefin copolymers. Expanding use in syringes, IV components, diagnostic casings, and wearable injectors reinforces long-term momentum. However, regulatory scrutiny, raw material price volatility, and competition from specialty engineering polymers moderate pricing flexibility.
“Many people aren’t aware that countless plastics products we use every day come from fossil-based components,” said Infinium CEO Robert Schuetzle. “With Infinium eNaphtha, Borealis will create plastics with an ultra-low carbon footprint for customers and end consumers seeking more sustainable, environmentally friendly alternatives.”
Polyolefin for Medical Devices Market Definition
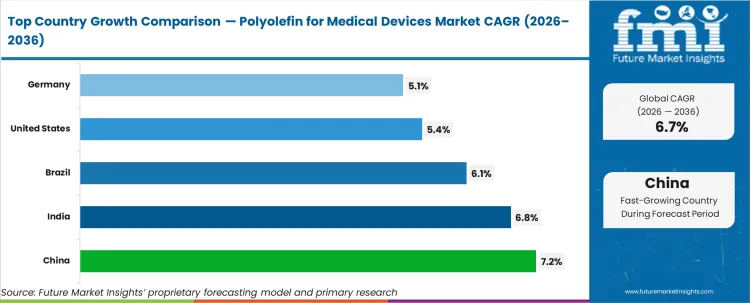
China (7.2%) is projected to record the fastest growth, supported by rapid medical device manufacturing expansion and export capacity. India (6.8%) follows, driven by domestic healthcare infrastructure growth and rising disposable device production. Brazil (6.1%) benefits from increasing diagnostic and hospital equipment demand. The United States (5.4%) and Germany (5.1%) represent mature markets where growth is steady but primarily innovation-led, constrained by regulatory compliance costs and established supplier ecosystems.
Polyolefin for medical devices market refers to the production and supply of polyolefin-based polymers used in the manufacturing of medical components and devices. Polyolefins include polyethylene (PE), polypropylene (PP), ethylene vinyl acetate (EVA), metallocene polypropylene, olefin block copolymers, and other specialty olefin materials. These materials are used because of their chemical resistance, flexibility, durability, and compatibility with sterilization processes. Their primary function is to provide safe, biocompatible, and processable materials for disposable and semi-durable medical devices. Key end uses include syringes, IV bags, blood bags, catheter components, inhaler housings, insulin pens, diagnostic casings, and wearable drug delivery systems.
Our report provides global and regional market size estimates for 2025 and 2026, with forecasts through 2036 and CAGR analysis. It includes segmentation by medical device type, polyolefin material type, sterilization compatibility, end user, and region. The study covers value-based sizing, regulatory influence assessment, sterilization trend analysis, material innovation developments, and demand patterns across OEMs and contract manufacturers.
The scope excludes non-polyolefin engineering plastics such as polycarbonate, ABS, PEEK, and PVC unless blended within defined polyolefin systems. It does not include finished medical devices or pharmaceutical formulations. The analysis focuses strictly on polyolefin raw materials and compounds used in medical device manufacturing rather than downstream healthcare products.
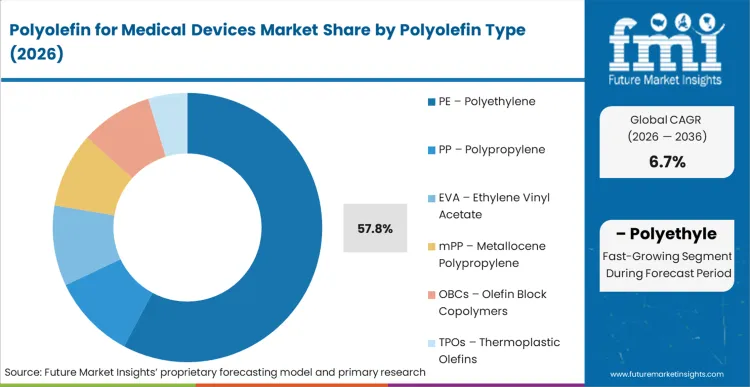
Based on FMI's Polyolefin for Medical Devices market report, consumption of PE - Polyethylene is estimated to hold 57.8% share in 2026. Polyethylene dominates due to its biocompatibility, chemical resistance, flexibility, and ease of sterilization. It addresses critical manufacturer requirements including low extractables, durability under repeated sterilization cycles, and compatibility with tubing, packaging films, and fluid-handling components. PE is widely used in medical tubing, IV bags, squeeze bottles, and protective packaging where flexibility and safety compliance are essential.
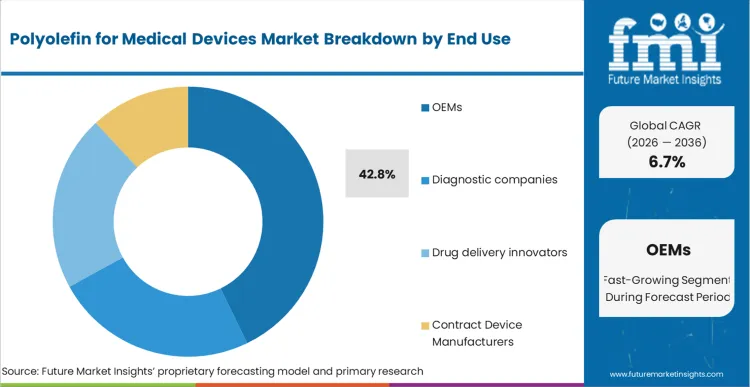
Based on FMI's Polyolefin for Medical Devices market report, consumption by OEMs is estimated to hold 42.8% share in 2026. OEMs lead due to their direct role in large-scale medical device manufacturing, including tubing systems, housings, diagnostic components, and sterile packaging. Polyolefins address OEM priorities related to regulatory compliance, material consistency, lightweight design, and cost-effective scalability.
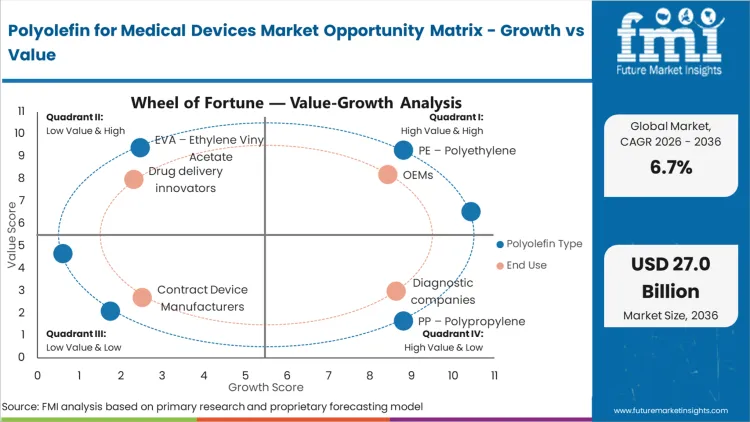
Future Market Insights analysis indicates that the polyolefin for medical devices market is structurally anchored to the global shift toward disposable, lightweight, and chemically stable medical components. Polyolefins such as polyethylene (PE) and polypropylene (PP) are widely used in syringes, IV components, diagnostic housings, packaging films, and non-implantable device parts due to their biocompatibility, sterilization compatibility, and cost efficiency. The market exists at its current scale because healthcare systems increasingly rely on high-volume, single-use medical devices to reduce cross-contamination risks and support infection control protocols. Regulatory frameworks governing medical device safety and material performance reinforce sustained demand for compliant polymer grades.
FMI analysts observe a transition from conventional commodity-grade polyolefins toward medical-grade, high-purity, and specialty formulations. While standard PE and PP continue to dominate volume in basic disposables, growth is increasingly concentrated in clarified polypropylene, metallocene-based polyolefins, and impact-modified grades designed for improved transparency, chemical resistance, and dimensional stability. These advanced materials carry higher per-kilogram pricing due to tighter quality controls and traceability requirements. However, improved performance, compatibility with gamma and ethylene oxide sterilization, and regulatory documentation justify premium positioning, supporting steady value growth even where device unit volumes fluctuate.
.webp)
The global polyolefin for medical devices market is segmented across Asia Pacific, North America, Europe, and Latin America. Regional performance is influenced by healthcare infrastructure expansion, medical device manufacturing capacity, regulatory compliance standards, and domestic polymer production. The full report provides comprehensive regional market attractiveness analysis.
| Country | CAGR (%) |
|---|---|
| China | 7.2% |
| India | 6.8% |
| Brazil | 6.1% |
| United States | 5.4% |
| Germany | 5.1% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Asia Pacific serves as the fastest-growing manufacturing base for medical devices and healthcare consumables. The region benefits from expanding hospital infrastructure and rising domestic device production. Key regional players include China Petrochemical Corporation, PetroChina Company Limited, and Reliance Industries Limited, all supplying medical-grade polyolefins. Competitive positioning is shaped by healthcare investment, polymer capacity expansion, and regulatory strengthening.
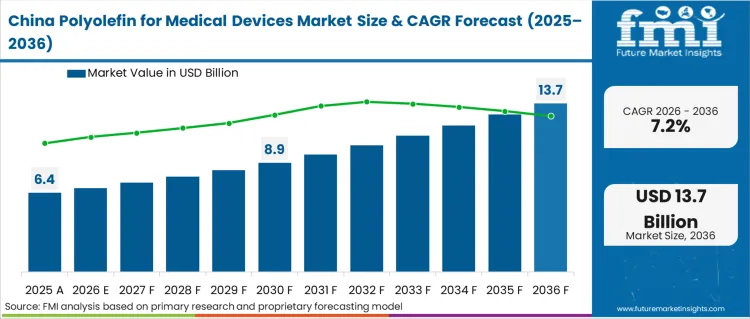
Demand for polyolefin for medical devices in China is projected to rise at 7.2% CAGR through 2036. Growth is driven by rapid expansion of domestic medical device manufacturing and hospital capacity. In 2024, the National Medical Products Administration continued strengthening Good Manufacturing Practice compliance for medical devices, reinforcing requirements for high-purity and traceable polymer materials. China’s Healthy China 2030 initiative continues to expand public healthcare infrastructure, increasing demand for syringes, IV components, and diagnostic device housings made from polypropylene and polyethylene. In 2024, domestic petrochemical producers announced capacity upgrades in high-performance polyolefins to serve medical and hygiene sectors. Rising exports of disposable medical consumables further strengthen demand for compliant polymer grades.
Demand for polyolefin for medical devices in India is projected to rise at 6.8% CAGR through 2036. Growth is linked to expansion of domestic medical device production supported by the Production Linked Incentive scheme for medical devices, extended through 2024. The Central Drugs Standard Control Organization continues enforcement of Medical Device Rules, strengthening traceability and packaging compliance requirements that indirectly drive use of certified medical-grade polymers. In 2024, investments in new medical device parks under government initiatives increased domestic manufacturing of syringes, catheters, and IV sets, all reliant on polypropylene and polyethylene. Rising healthcare access under national public health programs also supports growth in disposable medical product demand.
FMI's analysis of polyolefin for medical devices market in Asia Pacific consists of country-wise assessment that includes China and India. Readers can find insights on healthcare expansion, domestic polymer capacity growth, and regulatory compliance trends.
North America represents a technologically advanced and regulation-driven market for medical-grade polyolefins. Companies such as Exxon Mobil Corporation, LyondellBasell Industries N.V., and Chevron Phillips Chemical Company LLC supply high-purity resin grades tailored for healthcare applications. Market positioning is defined by regulatory oversight, traceability requirements, and innovation in medical device design.
Demand for polyolefin for medical devices in the United States is projected to rise at 5.4% CAGR through 2036. Growth is influenced by strict oversight from the USA Food and Drug Administration, which enforces quality system regulations for medical device materials. In 2024, continued alignment with ISO 13485 quality management standards reinforced demand for validated, medical-grade polymer resins. Expansion in minimally invasive devices, diagnostic equipment, and single-use medical consumables supports steady polypropylene and polyethylene demand. Public funding for healthcare infrastructure upgrades and increased production of domestically manufactured medical supplies further strengthen resin consumption. Sustainability initiatives in healthcare are also encouraging development of recyclable and lower-carbon polyolefin grades.
FMI's analysis of polyolefin for medical devices market in North America consists of country-wise assessment that includes the United States. Readers can find insights on FDA compliance impact, innovation in medical consumables, and high-purity polymer demand.
Europe functions as a compliance-driven and sustainability-focused market for medical device materials. Companies such as Borealis GmbH, TotalEnergies SE, and INEOS Group Holdings S.A. maintain medical-grade polyolefin portfolios aligned with European standards. Competitive positioning is shaped by regulatory harmonization and environmental performance expectations.
Demand for polyolefin for medical devices in Germany is projected to rise at 5.1% CAGR through 2036. The market is influenced by the European Union Medical Device Regulation, which reinforces strict documentation and material traceability requirements. In 2024, continued MDR implementation reviews strengthened oversight of device material compliance and risk assessment procedures. Germany’s strong medical device manufacturing base supports sustained demand for certified polypropylene and polyethylene grades. Additionally, the European Union’s climate and circular economy objectives encourage development of lower-carbon and recyclable polymer solutions suitable for medical applications. Industrial innovation funding programs continue to support advanced polymer research and healthcare material development.
FMI's analysis of polyolefin for medical devices market in Europe consists of country-wise assessment that includes Germany. Readers can find insights on MDR compliance impact, sustainable polymer innovation, and medical device production trends.
Latin America represents a growing market driven by healthcare infrastructure expansion and domestic production of medical consumables.
Demand for polyolefin for medical devices in Brazil is projected to rise at 6.1% CAGR through 2036. Growth is influenced by healthcare system modernization and regulatory enforcement. The Brazilian Health Regulatory Agency (ANVISA) continues to oversee medical device quality standards and material compliance. In 2024, public health procurement programs increased demand for disposable syringes, IV components, and diagnostic consumables manufactured using polypropylene and polyethylene. Investments in domestic petrochemical production capacity also support regional supply of medical-grade polymers. While economic conditions may influence short-term investment cycles, structural healthcare expansion supports sustained polymer demand.
FMI's analysis of polyolefin for medical devices market in Latin America consists of country-wise assessment that includes Brazil. Readers can find insights on public procurement influence, healthcare modernization, and domestic resin production capacity.
The polyolefin for medical devices market is moderately concentrated, dominated by large global petrochemical and polymer producers with integrated upstream and downstream operations. Companies such as LyondellBasell, Exxon Mobil, SABIC, Dow, Borealis, Braskem, INEOS, TotalEnergies, and Chevron Phillips Chemical account for a substantial share of global polyolefin production capacity. Competition in this segment is driven primarily by material purity, regulatory compliance (USP Class VI, ISO 10993), supply reliability, and long-term contracts with medical device manufacturers and converters. Scale and global distribution capabilities remain key competitive differentiators.
Structural advantages are strongest among companies with vertically integrated feedstock access, advanced polymerization technologies, and dedicated medical-grade product portfolios. Major producers such as LyondellBasell, SABIC, Dow, and Borealis operate global resin manufacturing networks supported by captive ethylene and propylene production, enabling cost stability and supply security. Many of these firms offer controlled medical-grade polyolefin lines with change management protocols, regulatory documentation support, and long-term formulation stability, which create high entry barriers. Companies with regional compounding and technical service centres further strengthen their positioning in regulated healthcare markets.
Buyer leverage in this market is significant but balanced by regulatory constraints. Medical device manufacturers typically require long-term material consistency, validated formulations, and strict change notification agreements, which increase switching costs once a resin grade is qualified. While large OEMs may dual source to mitigate risk, supplier substitution is limited due to regulatory revalidation requirements. As a result, pricing power remains relatively stable for approved medical-grade polyolefin suppliers, especially during feedstock volatility cycles.
Recent Development

| Metric | Value |
|---|---|
| Quantitative Units | USD 13,174.2 million (2025); USD 14,094.6 million (2026) to USD 26,941.3 million (2036), at a CAGR of 6.7% |
| Market Definition | The polyolefin for medical devices market comprises the global production and supply of medical-grade polyethylene, polypropylene, and specialty olefin-based polymers engineered for biocompatibility, sterilization resistance, chemical stability, and regulatory compliance across disposable, semi-durable, and wearable medical device applications. |
| Form Segmentation | Disposable Devices; Semi-Durable Devices; Wearable & Home-Use Devices |
| End Use Segmentation | OEMs; Diagnostic Companies; Drug Delivery Innovators; Contract Device Manufacturers; Hospitals, Clinics & Home Care Providers |
| Application Coverage | Syringes & Needles; IV Bags & Tubing; Blood Bags; Diagnostic Casings; Catheter Components; Inhalers & DPI Housings; Insulin Pens; Auto-Injectors; Transdermal Patch Holders; Surgical Kits |
| Regions Covered | North America; Latin America; East Asia; South Asia & Pacific; Eastern Europe; Western Europe; Middle East & Africa |
| Countries Covered | United States; Germany; France; United Kingdom; China; Japan; India; South Korea; Brazil; Italy; and 40+ countries |
| Key Companies Profiled | LyondellBasell Industries N.V.; Exxon Mobil Corporation; SABIC; The Dow Chemical Company; Borealis GmbH; Others |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up market modeling validated through primary interviews with polymer producers, medical device OEMs, and contract manufacturers, supported by capacity benchmarking, sterilization compatibility assessment, regulatory compliance analysis (FDA, ISO 10993), and trade flow evaluation across global healthcare supply chains. |
The Polyolefin for Medical Devices Market is valued at 14,094.6 Mn in 2026.
The market was valued at 13,174.2 Mn in 2025.
The market is projected to reach 26,941.3 Mn by 2036.
The Polyolefin for Medical Devices Market is expected to grow at a CAGR of 6.7%.
Disposable devices lead the market with 47.4% share, followed by semi-durable devices at 36.4% and wearable/home-use devices at 16.2%.
PE – Polyethylene dominates with 57.8% share, followed by PP Polypropylene at 23.5%.
OEMs represent the largest end-user segment, accounting for 42.8% of the market, followed by contract device manufacturers at 22.9%.
China is projected to grow at the highest CAGR of 7.2%, followed by India at 6.8% and Brazil at 6.1%.
The United States is projected to grow at a CAGR of 5.4%, while Germany is expected to expand at 5.1% during the forecast period.
The market covers polyolefins compatible with gamma, ethylene oxide (EtO), steam autoclave, and e-beam sterilization methods.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.