Medical Devices Surface Active Coatings Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
Medical Devices Surface Active Coatings Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Medical Devices Surface Active Coatings Market Forecast and Outlook 2026 to 2036
The medical devices surface active coatings market is projected at USD 2.3 billion in 2026 and USD 4.4 billion by 2036, reflecting a 6.80% CAGR. Adoption is guided by regulatory requirements, device material compatibility, and infection control protocols rather than aesthetics. Coatings are selected based on adhesion, durability, and biocompatibility. Once a formulation is validated for a device family, substitution requires full regulatory review and stability testing. Hospitals and device manufacturers assess application uniformity, handling safety, and shelf life. Supply planning aligns batches with production schedules. Service and technical support influence supplier selection. Training, labeling, and documentation affect rollout speed.
Market growth follows procedural adoption, device launches, and product line expansion rather than promotional activity. Production schedules coordinate surface preparation, coating deposition, and curing to ensure reproducibility. Release depends on adhesion testing, biocompatibility checks, and uniformity assessment. Warehouses stage coated components near device assembly lines to maintain integration timing. Quality teams monitor batch traceability, surface integrity, and compliance documentation. Supplier evaluation emphasizes consistency, audit readiness, and responsiveness. Profitability follows process yield, adherence to specifications, and predictable delivery cycles rather than catalog variety or discounting practices.
Quick Stats for Medical Devices Surface Active Coatings Market
- Medical Devices Surface Active Coatings Market Value (2026): USD 2.3 billion
- Medical Devices Surface Active Coatings Market Forecast Value (2036): USD 4.4 billion
- Medical Devices Surface Active Coatings Market Forecast CAGR (2026-2036): 6.8%
- Leading Type in Medical Devices Surface Active Coatings Market: Antimicrobial coatings
- Key Growth Regions in Medical Devices Surface Active Coatings Market: Asia Pacific, North America, Europe
- Top Players in Medical Devices Surface Active Coatings Market: SurModics, DSM Biomedical, Biocoat, Hydromer, AST Products
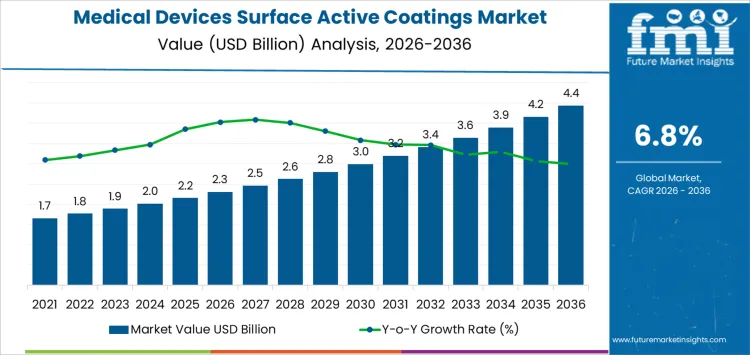
What Is the Growth Forecast for the Medical Devices Surface Active Coatings Market through 2036?
Regulatory compliance and device material compatibility drive demand in the medical devices surface active coatings market more than overall device production. In 2026, at about USD 2.3 billion, consumption is concentrated in cardiovascular implants, orthopedic devices, and minimally invasive tools where surface coatings reduce thrombosis, improve biocompatibility, and enhance wear resistance. Coating selection is fixed during device validation and remains in place across long production cycles, which ties material volumes to platform lifecycles rather than short term device output. Growth comes from new device introductions, expanded indications, and multi component assemblies requiring coated surfaces. The upward trend reflects gradual integration across device lines rather than replacement of existing platforms.
Process control and application consistency guide the later phase of the medical devices surface active coatings market. As value approaches roughly USD 4.4 billion by 2036, buyers focus on coating uniformity, adhesion performance, and reproducible surface energy across production lots. Manufacturers invest in precision deposition, in line inspection, and environmental control to meet regulatory and quality standards. The move from the low two billion into the high four billion reflects cumulative adoption across multiple device categories and production sites rather than a single technological shift. Suppliers that maintain controlled application processes, predictable performance, and reliable delivery tend to secure long term positions in approved material supply chains.
Medical Devices Surface Active Coatings Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 2.3 billion |
| Forecast Value (2036) | USD 4.4 billion |
| Forecast CAGR (2026-2036) | 6.8% |
How Is the Medical Devices Surface Active Coatings Market Enhancing Device Safety and Functionality?
Medical device surface active coatings are applied to implants, surgical instruments, and catheters to reduce friction, prevent biofilm formation, and improve biocompatibility. This specification behavior aligns with the broader medical coatings landscape, where validated performance and sterilization tolerance drive long production runs. Earlier devices used uncoated metals or polymers, which increased thrombosis, infection risk, and insertion difficulty. Surface active coatings modify material interfaces at a molecular level, controlling wettability, antimicrobial activity, or lubricity. R&D teams also evaluate antimicrobial nanocoatings when they need durable activity at very low coating thickness without affecting device dimensions. Engineers specify coatings by thickness uniformity, adhesion strength, chemical stability, and sterilization compatibility. Device teams often benchmark against medical grade coatings that already have established documentation packages and repeatable process controls. Adoption is concentrated in cardiovascular, orthopedic, and urology devices where device interaction with tissue or fluids critically affects outcomes. Manufacturers evaluate reproducibility, batch consistency, and regulatory documentation because coating performance directly impacts clinical reliability. Procurement decisions consider coating supplier support, integration with existing device assembly, and process validation requirements.
Commercial selection is led by R&D, quality, and regulatory teams because any change in coating can trigger new validation or approval filings. Earlier sourcing allowed multiple coating vendors, which increased variability and complicated compliance audits. Current programs favor standardized coating platforms with documented process control and lot traceability. Suppliers compete on adhesion stability, chemical resistance, and coating uniformity across volumes rather than headline antimicrobial claims. Process discipline is especially strict in peripheral vascular stents, where coating defects can translate into restenosis risk and post-market scrutiny. Device producers evaluate total cost through reduced failure rates, improved insertion performance, and lower rework, not coating price per unit alone. Distributors add value through technical training, post-coating inspection support, and supply continuity. Over time, demand follows growth in implantable and minimally invasive devices, where consistent surface properties influence both patient safety and manufacturing reproducibility across high-volume production lines.
What Factors Are Shaping the Demand for Medical Devices Surface Active Coatings Market in 2026?
Demand in Medical Devices Surface Active Coatings Market is driven by device performance requirements, regulatory compliance, and infection control protocols rather than short-term equipment or material procurement cycles. Selection of coating type is integrated into device design and validation programs, affecting manufacturing, sterilization processes, and clinical efficacy studies. Segment structure reflects differences in surface chemistry, durability under sterilization, and interaction with biological tissues or fluids. Buyers prioritize reproducibility, regulatory documentation, and compatibility with device materials. Suppliers that provide consistent quality, scalable application methods, and regulatory support are better positioned to capture high-value segments. Volume growth follows device adoption in hospitals and interventional suites rather than temporary market trends.
How Does Product Type Selection Affect Manufacturing Integration and Supplier Engagement in the Medical Devices Surface Active Coatings Market?
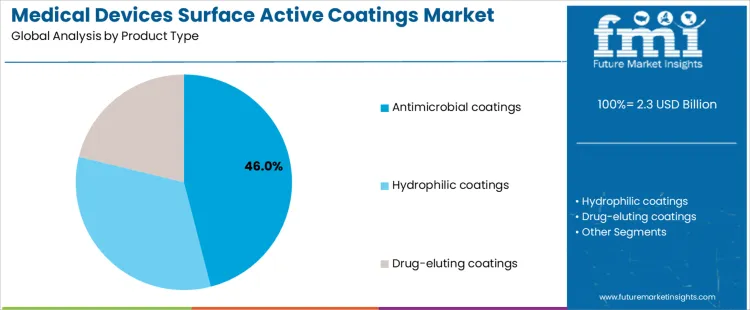
Antimicrobial coatings account for approximately 46% of demand due to their proven ability to reduce device-related infections, particularly on high-risk devices such as catheters and guidewires. Procurement teams often cross-reference performance claims with trends in antimicrobial-coated medical devices to validate infection-control value across comparable use cases. Manufacturers of these coatings must ensure uniform application, adhesion, and sterilization stability, which increases qualification requirements. Hydrophilic coatings are applied to improve device maneuverability and lubricity, especially for implants, which requires supplier collaboration on testing and material compatibility. Many catheter and guidewire programs treat hydrophilic coating selection as a locked design input because small formulation changes can shift friction and particulate behavior. Drug-eluting coatings support controlled local therapeutic delivery on surgical instruments, creating high technical entry barriers for suppliers. This capability overlaps with implantable drug eluting devices, where release kinetics and stability data become central to regulatory files. Each product type establishes specific process controls, inspection standards, and supplier performance expectations, influencing production planning and commercial agreements.
From a manufacturing and commercial perspective, coating selection defines quality control intensity and service requirements. Antimicrobial coatings demand verification of microbial inhibition and sterilization resistance. Hydrophilic coatings require assessment of surface wetting behavior and consistency over batches. Drug-eluting coatings involve long-term release profiling, stability testing, and regulatory validation. Once a coating type is approved for a device, substitutions are limited because changes require extensive regulatory submissions and revalidation. Demand by product type therefore aligns with device development pipelines, clinical adoption rates, and regulatory frameworks rather than short-term material pricing or availability.
Why Does Application Mix Determine Market Volume and Technical Complexity in the Medical Devices Surface Active Coatings Market?
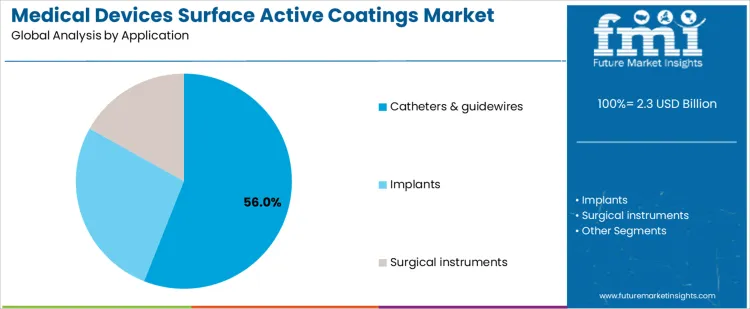
Catheters and guidewires represent approximately 56% of demand in the Medical Devices Surface Active Coatings Market because high procedural frequency and infection risk drive widespread adoption of antimicrobial solutions. Implants require coatings that ensure biocompatibility and durability, with moderate volumes but stringent regulatory and technical requirements. Surgical instruments utilize drug-eluting or lubricious coatings, where precision and controlled release properties define specification and supplier selection. These application differences inform procurement priorities, technical support needs, and service agreements, emphasizing the importance of specialized coatings aligned with device type and clinical context.
End-use segmentation also shapes supplier strategy and commercial opportunity. High-volume catheter programs favor suppliers capable of consistent batch quality, validated application processes, and scalable production. Implant manufacturers require extensive documentation, material traceability, and regulatory support. Surgical instrument coatings involve limited volumes but high-value margins, requiring strong R&D collaboration and technical support. The resulting market structure concentrates revenue in high-volume, high-frequency devices while maintaining higher technical complexity in implants and instruments. Segment shares therefore move with device adoption, procedural prevalence, and regulatory stringency rather than short-term shifts in medical device output.
How Is the Medical Devices Surface Active Coatings Market Being Defined By Device Performance And Patient Safety?
The Medical Devices Surface Active Coatings Market is shaped by how device manufacturers manage biocompatibility, surface interaction, and infection prevention. Coatings influence blood contact, wear resistance, and microbial adhesion, making them critical to device performance and regulatory compliance. Adoption patterns are also visible in bioactive coating devices, where surface function is tied directly to clinical risk management and validation burden. Buyers focus on coating consistency, adhesion strength, and chemical compatibility with base materials. Selection decisions involve design engineers, quality assurance teams, and regulatory specialists. This positions surface coatings as integral components that affect device approval and clinical outcomes rather than optional enhancements, emphasizing supplier reliability and documented performance.
How Are Manufacturing Consistency and Standard Device Platforms Changing Expectations in The Medical Devices Surface Active Coatings Market?
Medical device companies increasingly standardize platforms and components across multiple product lines to reduce variability and streamline regulatory approvals. This shifts expectations in the Medical Devices Surface Active Coatings Market toward coatings that perform reliably across different substrates and production lines. Buyers prioritize repeatable coating thickness, uniform surface energy, and minimal impact on device dimensions or functionality. Suppliers are evaluated on quality control, batch documentation, and long-term reproducibility. Consistent coatings that integrate seamlessly into standardized device platforms are more likely to be adopted for multiple products and long-term production runs.
Where Is Recurring Demand Being Built in The Medical Devices Surface Active Coatings Market?
Recurring demand arises from high-volume medical devices such as catheters, implants, surgical instruments, and cardiovascular products that require protective or functional coatings. Each production lot requires precise application to maintain compliance and device performance. The Medical Devices Surface Active Coatings Market also benefits from suppliers who can support multiple facilities or product lines with consistent quality. There is opportunity for companies that provide documentation, validation support, and training to internal teams. Being embedded in standard coating processes ensures predictable repeat orders and long-term revenue for coating suppliers.
Why Do Regulatory Requirements and Validation Constraints Limit Faster Adoption in The Medical Devices Surface Active Coatings Market?
New or alternative surface coatings require extensive testing, process validation, and regulatory review before they can be applied to approved devices. This makes device manufacturers cautious about switching suppliers or introducing new formulations. The Medical Devices Surface Active Coatings Market also faces cost and timeline pressure because validation delays can impact product launches. Even when performance benefits are clear, adoption may be limited to high-risk or high-value devices. These factors slow wider application and keep usage concentrated in devices with critical performance requirements or strict clinical standards.
What is the Demand for Medical Devices Surface Active Coatings by Country?
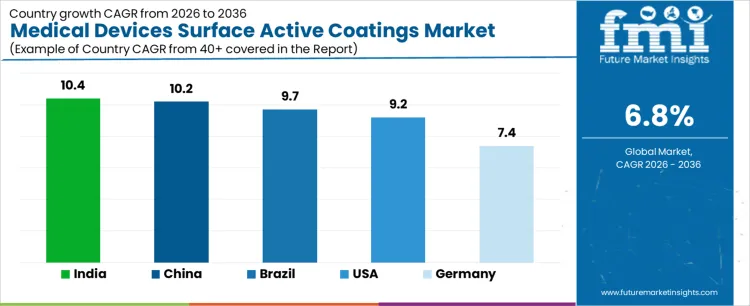
| Country | CAGR |
|---|---|
| USA | 9.2% |
| Brazil | 9.7% |
| China | 10.2% |
| India | 10.4% |
| Germany | 7.4% |
Demand for medical devices surface active coatings is rising as manufacturers aim to improve biocompatibility, reduce bacterial adhesion, and enhance durability of surgical instruments, implants, and diagnostic devices. India leads with a 10.4% CAGR, supported by expansion of medical device production, rising export oriented manufacturing, and growing adoption of coated devices in hospitals and clinics. China follows at 10.2%, driven by domestic medical device scale up, hospital modernization, and regulatory push for quality improvements. Brazil records 9.7%, reflecting increased use in implantables and surgical tools. The USA grows at 9.2%, shaped by replacement of legacy devices and adoption of advanced coatings. Germany, at 7.4%, reflects a mature device manufacturing market with steady innovation uptake.
How Are Infection Control Protocols Driving Demand in the Medical Devices Surface Active Coatings Market in the United States?
Rising hospital emphasis on infection prevention supports a 9.2% CAGR for medical devices surface active coatings demand in United States. Procurement authority sits with sterile processing teams, surgical committees, and device evaluation boards. Adoption focuses on catheters, implants, and surgical instruments requiring antimicrobial or hydrophilic coatings. Device selection emphasizes durability, biocompatibility, and ease of sterilization. Once validated, coating technologies are standardized across hospital networks and OEM platforms. Regulatory compliance and FDA guidance influence acceptance. Domestic suppliers provide coatings for routine devices, while import products serve higher performance applications. Capital allocation aligns with hospital equipment upgrades and new product launches. Replacement cycles track usage. Commercial success depends on inclusion in hospital device lists and OEM programs rather than isolated single unit sales.
Why Are Hospital Network Expansion Programs Influencing Demand in the Medical Devices Surface Active Coatings Market in Brazil?
Expansion of private and public hospital chains supports a 9.7% CAGR for medical devices surface active coatings demand in Brazil. Central procurement offices and clinical engineering teams control most adoption decisions. Coatings are applied to devices for infection prevention, hydrophilic performance, and patient safety. Import systems dominate high specification instruments, while local suppliers cover standard instruments. Once a product is approved, deployment spans multiple hospitals within the network. Training and documentation influence adoption. Regulatory requirements, procedural guidelines, and sterilization compatibility shape equipment choice. Capital spending follows network expansion and refurbishment schedules. Replacement cycles track device life and usage intensity. Commercial success depends on inclusion in network framework agreements and reference installations rather than ad hoc clinic purchases or individual instrument orders.
What Are Institutional Safety Standards Driving in the Medical Devices Surface Active Coatings Market in China?
Hospital safety and infection prevention standards support a 10.2% CAGR for medical devices surface active coatings demand in China. Major tertiary hospitals, specialty clinics, and medical device manufacturers control most procurement. Adoption targets catheters, surgical instruments, and implantable devices requiring antimicrobial or lubricious coatings. Domestic suppliers dominate standard devices, while international brands retain positions in premium or complex systems. Regulatory approval and tender processes shape selection. Once a technology is approved, replication across multiple facilities is rapid. Training programs, sterilization workflow, and reporting influence acceptance. Capital allocation aligns with hospital expansion and replacement schedules. Replacement cycles track procedure volume and device wear. Commercial success depends on inclusion in institutional procurement lists and OEM qualification frameworks rather than sales to fragmented smaller facilities or single departments.
Why Are Device Standardization and Hospital Program Expansion Driving Growth in the Medical Devices Surface Active Coatings Market in India?
Growing hospital networks and device standardization policies support a 10.4% CAGR for medical devices surface active coatings demand in India. Adoption is led by tertiary care hospitals, private chains, and specialty clinics. Coatings are applied to implants, catheters, and surgical instruments to enhance infection control and device performance. Import products serve high specification applications, while domestic suppliers cover routine instruments. Procurement follows central committee approval, and once validated, deployment spreads across multiple sites. Capital planning aligns with hospital expansions and new device rollouts. Training, documentation, and sterilization compatibility influence acceptance. Replacement cycles track device usage. Commercial success depends on inclusion in hospital standard equipment lists and OEM qualification programs rather than selling to individual clinics or isolated operating rooms.
How Are Regulatory Guidance and Sterilization Protocols Shaping Demand in the Medical Devices Surface Active Coatings Market in Germany?
Regulatory guidance and sterilization compliance programs establish a 7.4% CAGR baseline for medical devices surface active coatings demand in Germany. University hospitals, specialty clinics, and medical device manufacturers control most adoption. Evaluation focuses on durability, biocompatibility, and antimicrobial performance. Procurement follows health technology assessment and quality assurance processes. Import suppliers dominate premium instruments, while domestic suppliers cover routine devices. Once approved, coatings are integrated across multiple departments and hospital networks. Training and workflow compatibility influence adoption. Capital planning follows hospital refurbishment and new device schedules. Replacement cycles track device life and usage intensity. Commercial success depends on inclusion in approved device lists, OEM frameworks, and hospital standardization programs rather than one off clinic purchases.
How Do Suppliers Compete for Placement in the Medical Devices Surface Active Coatings Market?
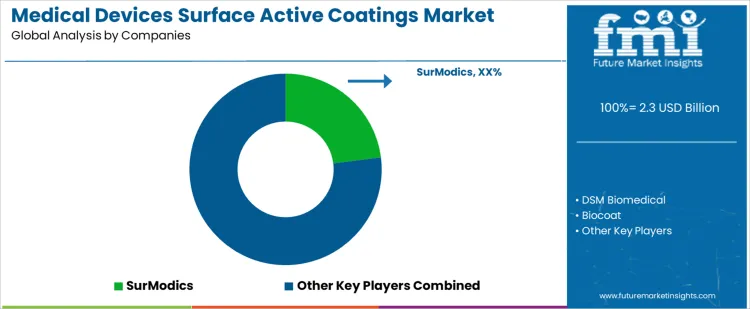
Medical device surface active coating adoption follows device design, regulatory pathway, and performance validation rather than simple material selection. Engineers define wettability targets, protein adsorption limits, lubricity, and sterilization compatibility before approving coatings. SurModics, DSM Biomedical, Biocoat, Hydromer, AST Products compete within those specification programs. Once a coating enters device master files, requalification and regulatory submission requirements anchor that choice. SurModics focuses on hydrophilic coatings for catheters and stents with established clinical data. DSM Biomedical supplies polymer chemistries tailored to vascular and orthopedic implants. Biocoat emphasizes anti thrombogenic and lubricious layers for small bore devices. Hydromer targets custom formulations for combination products and controlled release applications. AST Products provides contract coating services across varied medical platforms.
Competitive outcomes reflect process reproducibility, regulatory compliance, and supply consistency rather than price alone. Quality teams examine coating thickness uniformity, surface energy stability, sterilization tolerance, and lot to lot variability during evaluation. Manufacturing departments value suppliers capable of scaling coating processes without disrupting device assembly. SurModics benefits from documented clinical performance and long term regulatory track record. DSM Biomedical secures positions where polymer customization aligns with device mechanics. Biocoat holds share in programs requiring specialized anti thrombogenic properties. Hydromer competes through rapid formulation support and adaptation to device geometry. AST Products gains placement through contract services that reduce internal process burden. Market positions vary by device type, regulatory class, and internal validation protocols across manufacturers.
Key Players in the Medical Devices Surface Active Coatings Market
- SurModics
- DSM Biomedical
- Biocoat
- Hydromer
- AST Products
- Others
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD Billion |
| Product Type | Antimicrobial coatings; Hydrophilic coatings; Drug-eluting coatings |
| Application | Catheters & guidewires; Implants; Surgical instruments |
| End User | OEM partnerships; Contract coating services; Aftermarket |
| Region | Asia Pacific; Europe; North America; Latin America; Middle East & Africa |
| Countries Covered | United States, Canada, Mexico, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Brazil, Chile, Saudi Arabia, Turkey, South Africa, Rest of regions |
| Key Companies Profiled | SurModics; DSM Biomedical; Biocoat; Hydromer; AST Products |
| Additional Attributes | Dollar by sales by product and application, coating thickness control, adhesion and durability performance, sterilization compatibility, regulatory validation burden, OEM qualification cycles, lot traceability, yield and scrap rates, batch reproducibility, integration with device assembly lines |
Medical Devices Surface Active Coatings Market Segmentation
Product Type:
- Antimicrobial coatings
- Hydrophilic coatings
- Drug-eluting coatings
Application:
- Catheters & guidewires
- Implants
- Surgical instruments
End User:
- OEM partnerships
- Contract coating services
- Aftermarket
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- Cassa, M. A. (2024). Smart self‑defensive coatings with bacteria‑triggered antimicrobial responses for medical devices. Biomaterials Science, 12(8), 1234–1250.
- Negut, I., Albu, C., & Bita, B. (2024). Advances in antimicrobial coatings for preventing infections of head‑related implantable medical devices. Coatings, 14(3), 256.
- International Journal of Medical Science and Clinical Research. (2025). The impact of hydrophilic coatings on medical device performance: A comprehensive review, 5(11), 1916–1921.
Frequently Asked Questions
How big is the medical devices surface active coatings market in 2026?
The global medical devices surface active coatings market is estimated to be valued at USD 2.3 billion in 2026.
What will be the size of medical devices surface active coatings market in 2036?
The market size for the medical devices surface active coatings market is projected to reach USD 4.4 billion by 2036.
How much will be the medical devices surface active coatings market growth between 2026 and 2036?
The medical devices surface active coatings market is expected to grow at a 6.8% CAGR between 2026 and 2036.
What are the key product types in the medical devices surface active coatings market?
The key product types in medical devices surface active coatings market are antimicrobial coatings, hydrophilic coatings and drug-eluting coatings.
Which application segment to contribute significant share in the medical devices surface active coatings market in 2026?
In terms of application, catheters & guidewires segment to command 56.0% share in the medical devices surface active coatings market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Antimicrobial coatings
- Hydrophilic coatings
- Drug-eluting coatings
- Antimicrobial coatings
- Y-o-Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Catheters & guidewires
- Implants
- Surgical instruments
- Catheters & guidewires
- Y-o-Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- OEM partnerships
- Contract coating services
- Aftermarket
- OEM partnerships
- Y-o-Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Application
- By End User
- Competition Analysis
- Competition Deep Dive
- SurModics
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- DSM Biomedical
- Biocoat
- Hydromer
- AST Products
- Others
- SurModics
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 10: Global Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by End User
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y to o to Y Growth Comparison by Region, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 24: North America Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Product Type
- Figure 26: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 27: North America Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Application
- Figure 29: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 30: North America Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by End User
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 34: Latin America Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 35: Latin America Market Attractiveness Analysis by Product Type
- Figure 36: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 37: Latin America Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 38: Latin America Market Attractiveness Analysis by Application
- Figure 39: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 40: Latin America Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by End User
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 44: Western Europe Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 45: Western Europe Market Attractiveness Analysis by Product Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 47: Western Europe Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 48: Western Europe Market Attractiveness Analysis by Application
- Figure 49: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 50: Western Europe Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 51: Western Europe Market Attractiveness Analysis by End User
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 54: Eastern Europe Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 57: Eastern Europe Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Application
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 60: Eastern Europe Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End User
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 64: East Asia Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 65: East Asia Market Attractiveness Analysis by Product Type
- Figure 66: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 67: East Asia Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 68: East Asia Market Attractiveness Analysis by Application
- Figure 69: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 70: East Asia Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 71: East Asia Market Attractiveness Analysis by End User
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 84: Middle East & Africa Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 87: Middle East & Africa Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 90: Middle East & Africa Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

