Gene Editing Off-Target Effect Detection Test Systems Market is estimated at USD 391.8 million in 2025. Industry value is projected to reach USD 444.3 million in 2026 and USD 1,555.1 million by 2036, reflecting a 13.4% CAGR during the forecast period. The market is gaining importance as genome editing programs move deeper into translational and clinical development. Sponsors are placing more emphasis on test systems that identify candidate off-target sites and support a clearer path into regulatory review.
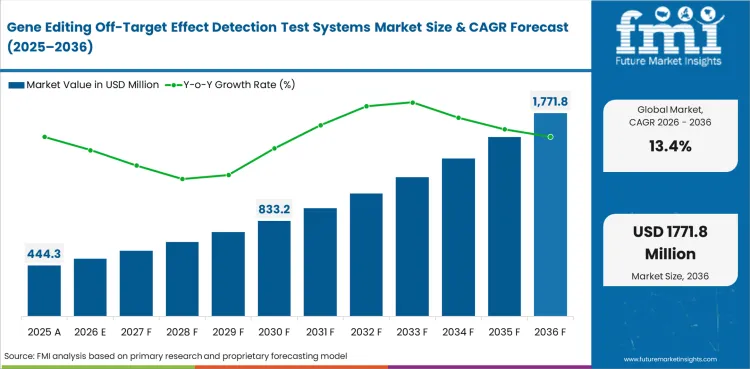
| Parameter | Details |
|---|---|
| Market value (2026) | USD 444.3 million |
| Forecast value (2036) | USD 1,555.1 million |
| CAGR (2026 to 2036) | 13.4% |
| Estimated market value (2025) | USD 391.8 million |
| Incremental opportunity | USD 1,110.8 million |
| Leading detection approach | Cell-based assays (31.2% of detection approach segment) |
| Leading product format | Services (34.1% of product format segment) |
| Leading workflow stage | Site nomination (36.4% of workflow stage segment) |
| Leading editing modality | CRISPR-Cas9 (42.3% of editing modality segment) |
| Leading readout technology | Amplicon NGS (29.1% of readout technology segment) |
| Leading end user | Biopharma companies (39.4% of end user segment) |
| Key players | Integrated DNA Technologies, MaxCyte/SeQure, SeqWell, Active Motif, Mission Bio, Azenta Life Sciences, Avance Biosciences |
Source: Future Market Insights analysis, based on proprietary forecasting model and primary research.
Off-target effect detection is becoming more important in gene editing because development groups need earlier clarity on unintended edits before programs move deeper into preclinical and clinical work. Early analytical evidence helps identify risk before it affects guide selection, safety interpretation, or regulatory preparation. System choice depends on assay sensitivity, biologic relevance, sequencing quality, and clear interpretation. Adoption improves when developers define how each result supports nomination, confirmation, genomic integrity review, and routine program decisions.
China is expected to rise at 14.6% CAGR through 2036 because expanding genome editing research and translational activity are increasing demand for stronger off-target assessment. The United States is projected to grow at 13.9% CAGR as clinical-stage gene editing work and regulatory-facing safety requirements continue supporting analytical use. South Korea is anticipated to expand at 13.2% CAGR through 2036, supported by precision medicine investment and strengthening genomic capability. The United Kingdom is likely to register 12.8% CAGR, while Germany is set to grow at 12.6% CAGR and Switzerland at 12.4% CAGR as high-value therapeutic development continues requiring stronger evidence around unintended edits and genomic integrity. Japan is expected to expand at 11.9% CAGR through 2036.
The gene editing off-target effect detection test systems market includes assay platforms, sequencing-linked workflows, software tools, and service-based analytical systems used to identify, nominate, confirm, and characterize unintended editing events generated during genome editing research, translational development, and therapeutic program advancement.
The market includes cell-based assays, biochemical assays, in silico prediction tools, orthogonal confirmation workflows, sequencing-based readout systems, analytical software, service-led off-target profiling, and related workflow components used across guide selection, preclinical development, IND-enabling studies, genomic integrity assessment, and edited-cell characterization.
The scope excludes general gene editing reagents sold without a defined off-target analysis role, broad sequencing activity not linked to genome editing safety assessment, general bioinformatics services without off-target workflow relevance, and manufacturing tools used for editing delivery or cell processing where off-target detection is not the primary analytical function.
Demand for gene editing off-target effect detection test systems is rising because genome editing programs face tighter scrutiny around unintended edits and structural genomic changes. FDA’s draft guidance now places clearer emphasis on NGS and bioinformatics for off-target and chromosomal integrity assessment. EMA guidance also points to in vitro and in silico off-target evaluation in investigational advanced therapies.
The market is expanding as off-target assessment moves from a narrow discovery task into a broader program requirement. Developers use multiple methods across nomination, confirmation, and structural follow-up, especially in programs involving CRISPR-Cas systems, base editors, and prime editors. NIST work on assay qualification, interlaboratory consistency, and metadata discipline adds further relevance to workflows that support repeatable output and cleaner interpretation across development stages.
Clinical expansion is adding another further support. CRISPR programs continue to widen across therapeutic settings, and which increases the need for robust off-target evidence before later-stage investment decisions are made. Market growth is shaped by scientific depth, regulatory expectation, and the need for better-documented analytical support across gene editing programs moving closer to patient-facing use.
The gene editing off-target effect detection test systems market is segmented by Detection Approach, Product Format, Workflow Stage, Editing Modality, Readout Technology, End User, Application, Deployment, and Region. By Detection Approach, the market is divided into Cell-based Assays, Biochemical Assays, In Silico Tools, and Orthogonal Validation. In terms of Product Format, the market is classified into Services, Assay Kits, Software, Instruments, and Reagents. Based on Workflow Stage, the market is segmented into Site Nomination, Site Confirmation, Structural Follow-up, and Release Support. By Editing Modality, the market is divided into CRISPR-Cas9, Base Editing, Prime Editing, Cas12 Systems, and Other Editors. In terms of Readout Technology, the market is classified into Amplicon NGS, Short-read WGS, Single-cell DNA, ChIP-seq Workflows, and Long-read Sequencing. Based on End User, the market is segmented into Biopharma Companies, Academic Laboratories, CROs, CDMOs, and Hospitals. By Application, the market is divided into IND-enabling Studies, Discovery Screening, Cell Therapy Quality Assessment, In vivo Profiling, and Comparability Studies. In terms of Deployment, the market is classified into Outsourced Testing, In-house Testing, and Hybrid Models.
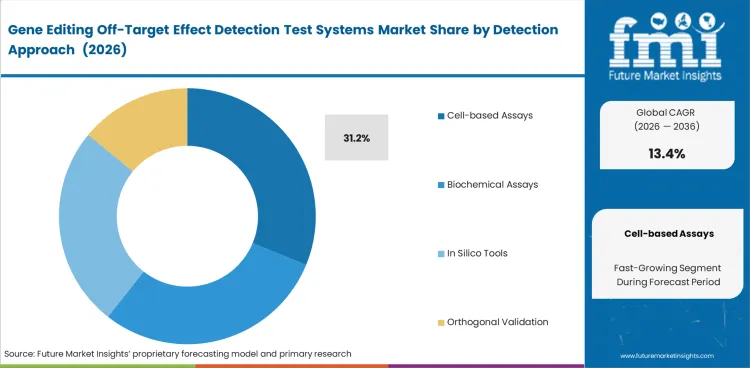
Cell-based assays lead this segment because off-target findings carry more value when they are generated in a biological setting closer edited cells. Cell-based assays are expected to account for 31.2% share in 2026, supported by the growing need for evidence that moves beyond computational prediction and strengthens confidence in follow-up review. Interest in this segment is also supported by progress in the CRISPR-based gene editing market, where therapeutic use continues to raise the bar for safety assessment.
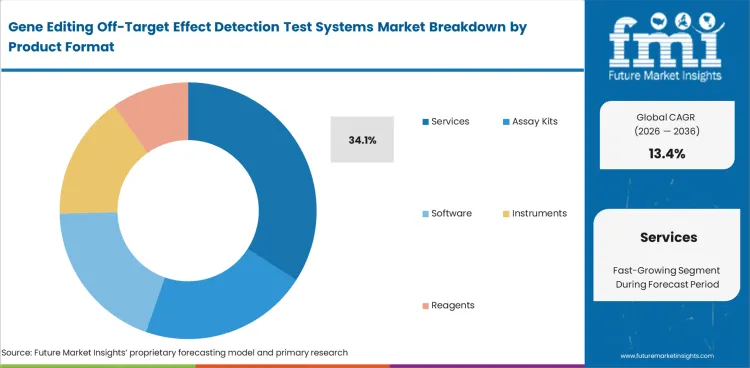
Services lead this segment because many gene editing developers still rely on external specialists for study design, assay execution, sequencing support, and interpretation of technically complex findings. Services are projected to hold 34.1% share in 2026 because off-target analysis often requires a broader analytical package than internal teams can manage with speed and consistency. This position is reinforced by activity across the bioinformatics market, where interpretation and data handling remain central to analytical value.
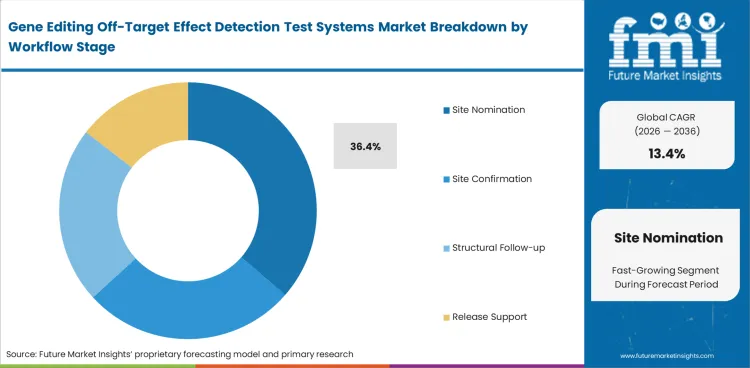
Site nomination leads because off-target assessment starts with identifying the genomic locations most likely to show unintended activity before confirmation work begins. Site nomination is likely to account for 36.4% share in 2026 because this step shapes later analytical work and determines how much confidence teams can place in downstream review. As the gene editing tools market continues expanding, this step remains one of the earliest and most important checkpoints in the workflow.
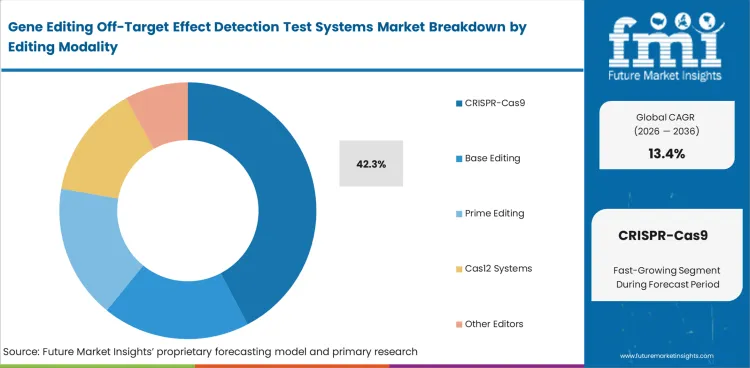
CRISPR-Cas9 leads this segment because a large share of active development work still depends on Cas9-based editing systems and the analytical needs linked to them. CRISPR-Cas9 is expected to account for 42.3% share in 2026 because the installed base of research and translational programs remains strongest in this modality even as base editing and prime editing continue to grow. This lead is closely tied to the wider CRISPR market, where therapeutic and research activity continue supporting demand for off-target review.
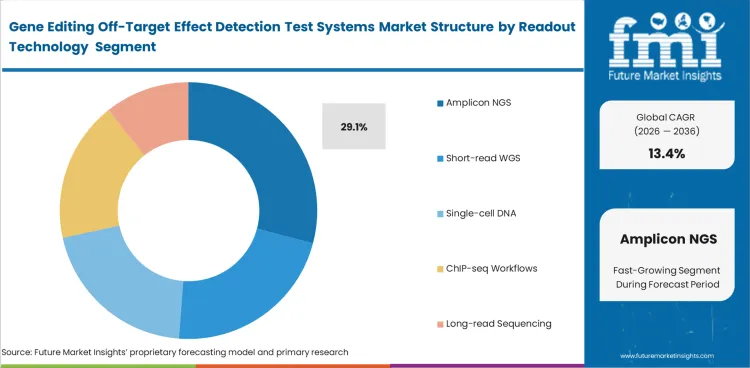
Amplicon NGS is expected to account for 29.1% share in 2026 because targeted sequencing remains a practical way to confirm nominated sites with clear and manageable output. Development groups continue to favor methods that support structured confirmation without the heavier burden linked to broader sequencing strategies, especially when turnaround time and interpretability both matter. This lead also fits with trends in the genomics market, where sequencing depth and analytical usability continue to shape adoption.
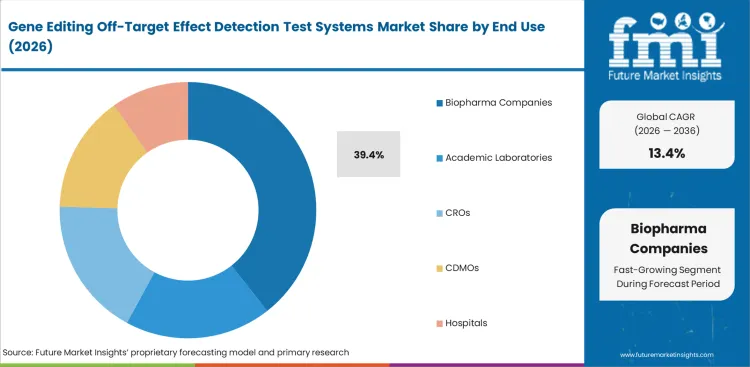
Biopharma companies are expected to account for 39.4% share in 2026 because the strongest commercial pressure for off-target analysis sits with organizations moving programs through preclinical and clinical development. These companies need stronger analytical evidence as programs move towards higher-cost and higher-risk development stages, which keeps demand concentrated in this group. The same pattern is visible across the genome editing market, where therapeutic progression continues to drive deeper analytical requirements.
The gene editing off-target effect detection test systems market is expanding as development groups place more weight on stronger evidence around unintended edits, genomic integrity, and regulatory readiness. Adoption depends on whether analytical output supports clear interpretation, repeatable use across workflows, and stronger confidence during program review. Growth remains strong, though uptake is still shaped by method qualification burden, workflow complexity, and the challenge of aligning results from multiple assay types into one usable safety view.
Demand is rising because gene editing programs need earlier clarity on unintended edits before they move deeper into preclinical and clinical development. Development groups need systems that identify likely off-target sites, support confirmation with stronger evidence, and help interpret broader genomic disruption when safety questions become more complex. This is increasing the relevance of off-target test systems across guide selection, translational work, and IND-enabling studies. The same trend aligns with expansion in the gene editing tools market, where development-stage use continues to broaden.
Wider uptake remains constrained by the effort required to qualify methods and interpret results across different assay classes. Development teams need more than sensitive detection. They need a clear understanding of how nomination methods, confirmation workflows, and sequencing readouts connect before results can support structured safety review. This slows adoption, especially in programs where low-frequency events and structural changes create added interpretation burden. It also increases dependence on capabilities associated with the bioinformatics market.
Market direction is moving toward systems that support biologically relevant evidence, cleaner sequencing-backed confirmation, and stronger continuity from nomination through follow-up analysis. Development teams are giving more weight to tools and services that support routine analytical execution rather than one-off technical use, especially when programs move toward regulatory-facing stages. This shift also draws support from advances in the genomics market, where sequencing capability and analytical depth continue improving the usefulness of more structured off-target workflow.
Based on the regional analysis, the Gene Editing Off-Target Effect Detection Test Systems Market is segmented into North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, and Middle East & Africa across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 14.6% |
| United States | 13.9% |
| South Korea | 13.2% |
| United Kingdom | 12.8% |
| Germany | 12.6% |
| Switzerland | 12.4% |
| Japan | 11.9% |
Source: Future Market Insights analysis, based on proprietary forecasting model and primary research
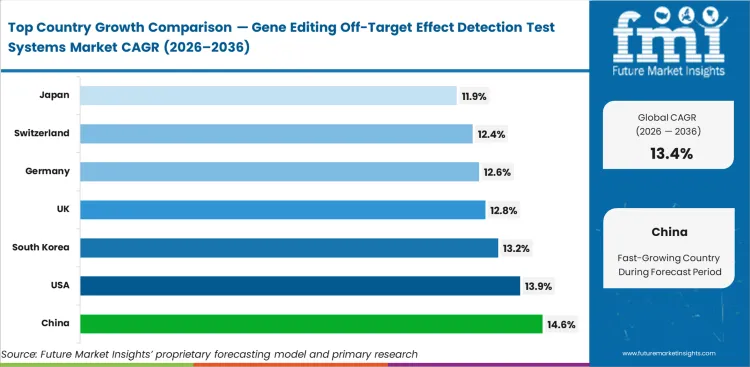
China is projected to expand at 14.6% CAGR through 2036. Strong research activity in genome editing, rising translational work, and broader use of advanced sequencing workflows are support demand for off-target detection systems. Development teams are placing greater emphasis on methods that generate clearer evidence on unintended edits before programs move into more demanding stages of review.
The United States is expected to grow at 13.9% CAGR through 2036. A strong base of gene editing companies, clinical development activity, and specialist analytical providers support market expansion. Demand also benefits from growing regulatory attention on unintended edits and genomic integrity. Companies active in this market are giving more weight to assay reliability, sequencing quality, and interpretive support as programs move toward higher-stakes development decisions. This pattern aligns with the wider CRISPR-based gene editing market.
South Korea is projected to record 13.2% CAGR by 2036. The market benefits from continued investment in genomic medicine, growing interest in advanced therapies, and improving analytical capability across research and development settings. Demand is building around systems that support more dependable safety interpretation without adding unnecessary complexity to program execution. Companies with stronger technical support and cleaner workflow delivery are likely to perform well in this market, especially as the country builds further capability across single cell analysis system applications.
The United Kingdom is projected to expand at 12.8% CAGR from 2026 to 2036. Strong academic science, translational research depth, and continued interest in cell and gene therapy development support this market. Off-target detection systems are gaining more attention because development groups need clearer evidence around unintended edits as programs mature. Market progress depends on scientific credibility, analytical consistency, and the ability to support structured development work with dependable results.
Germany is projected to expand at 12.6% CAGR through 2036. The market is shaped by a technically disciplined research environment and strong interest in robust analytical workflows. Development teams assess these systems on assay performance, sequencing output, documentation quality, and interpretation support, especially in programs that require stronger evidence across multiple stages of review. This keeps Germany important for suppliers that combine technical depth with dependable analytical execution, particularly as the broader genomics market continues to deepen its analytical base.
Switzerland is projected to register 12.4% CAGR by 2036. Demand comes from a high-value therapeutic development environment where evidence quality carries strong weight. The strongest interest is directed toward systems and services that support confident interpretation of unintended edits and related genomic effects. Companies active here are more likely to gain traction when they provide dependable analytical support across specialized development programs with demanding evidence expectations. This also connects well with the wider bioinformatics market.
Japan is expected to expand at 11.9% CAGR through 2036. Growth remains solid, though adoption is moving in a more measured way than in some faster-growing countries. Development groups continue to favor methods that provide repeatable output, well-structured sequencing analysis, and dependable interpretation across different stages of program work. This keeps the market attractive for companies that can support long-term analytical consistency and clear workflow execution.
The gene editing off-target effect detection test systems market includes assay specialists, sequencing-linked workflow providers, bioinformatics-supported analysis participants, and service-led companies working across genome editing research and therapeutic development. The wider CRISPR market gives useful context here because demand is widening across both research and clinical programs. Development teams compare workflow continuity, sequencing quality, interpretive support, service responsiveness, and the ability to generate results that can support internal review and later regulatory preparation.
Integrated DNA Technologies remains well positioned through its rhAmpSeq-based workflow, while MaxCyte has strengthened its position through SeQure and Mission Bio remains relevant through single-cell analysis of on-target edits, off-target events, and translocations. Active Motif, SeqWell, Azenta Life Sciences, and Avance Biosciences continue supporting the market through specialized services and workflow execution, with the single cell analysis system market adding useful context to this positioning. Stronger market positions are likely to remain with companies that combine analytical depth, dependable support, and clearer interpretive value across the broader genome editing landscape.
Key global companies leading the gene editing off-target effect detection test systems market include:
| Company | Off-Target Assay Depth | Genomic Integrity Coverage | Workflow Breadth | Documentation / Application Support |
|---|---|---|---|---|
| Integrated DNA Technologies | High | Medium | Strong | Strong |
| MaxCyte / SeQure | High | High | Strong | Strong |
| Mission Bio | High | High | Moderate | Strong |
| Active Motif | Medium | Medium | Moderate | Strong |
| SeqWell | Medium | Medium | Moderate | Moderate |
| Azenta Life Sciences | Medium | Medium | Moderate | Moderate |
| Avance Biosciences | Medium | Medium | Moderate | Moderate |
Source: Future Market Insights competitive analysis, 2026.
Recent Developments in Gene Editing Off-Target Effect Detection Test Systems Market
Major Global Players
Emerging Players / Specialized Participants
| Metric | Value |
|---|---|
| Quantitative Units | USD 391.8 million (2025) / USD 444.3 million (2026) to USD 1,555.1 million (2036), at a CAGR of 13.4% |
| Market Definition | The gene editing off-target effect detection test systems market includes assay platforms, sequencing-linked workflows, software tools, and service-based analytical systems used to identify, nominate, confirm, and characterize unintended editing events generated during genome editing research, translational development, and therapeutic program advancement. |
| Segmentation |
|
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, and Middle East & Africa |
| Countries Covered | China, United States, South Korea, United Kingdom, Germany, Switzerland, Japan, and 40 plus countries |
| Key Companies Profiled | Integrated DNA Technologies, MaxCyte / SeQure, Mission Bio, Azenta Life Sciences, Active Motif, SeqWell, Avance Biosciences |
| Forecast Period | 2026 to 2036 |
| Approach | Bottom-up review of assay use and workflow-based demand modeling, supported by primary interviews with genome editing researchers, assay developers, and regulatory consultants, alongside secondary research including FDA/EMA guidance, NIST material, peer-reviewed studies, and public company updates |
Source: Future Market Insights analysis, based on proprietary forecasting model and primary research.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is the Gene Editing Off-Target Effect Detection Test Systems Market in 2026?
The market is expected to reach USD 444.3 million in 2026.
What will the market be worth by 2036?
FMI estimates market value will reach USD 1,555.1 million by 2036.
What CAGR is projected for the market during 2026 to 2036?
The market is projected to expand at a 13.4% CAGR during the forecast period.
Which detection approach segment leads the market?
Cell-based assays lead the detection approach segment, with 31.2% share expected in 2026.
Which product format segment holds the leading position?
Services are projected to lead product format demand, accounting for 34.1% share in 2026.
Which workflow stage remains the most commercially important?
Site nomination is expected to hold 36.4% share in 2026 because candidate unintended edit sites need to be identified before deeper confirmation work proceeds.
Which end user segment leads the market?
Biopharma companies are expected to account for 39.4% share of end user demand in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.