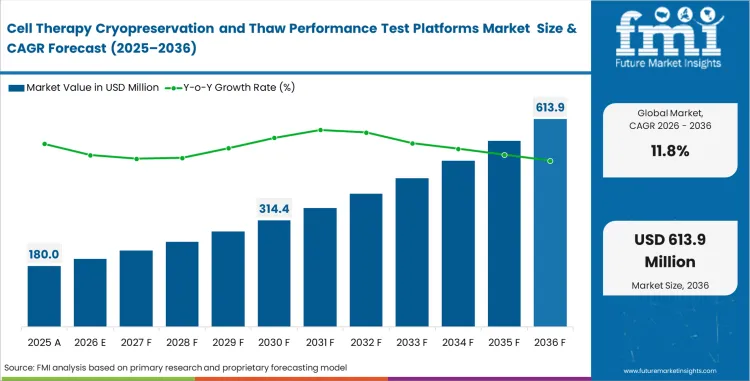
Cell therapy cryopreservation and thaw performance test platforms market crossed a valuation of USD 180 million in 2026 and expand at a CAGR of 11.8% from 2026 to 2036. Market value is projected to rise to USD 549 million by 2036. Growth is driven by the need to control thawing, assess post-thaw viability and improve handling consistency before release or administration.
| Parameter | Details |
|---|---|
| Market value (2026) | USD 180 million |
| Forecast value (2036) | USD 549 million |
| CAGR (2026 to 2036) | 11.8% |
| Estimated market value (2025) | USD 161 million |
| Leading platform type | Post-thaw analyzers |
| Platform share (2026) | 32% |
| Leading assay readout | Viability assays |
| Assay share (2026) | 29% |
| Leading cell type | CAR-T cells |
| Cell type share (2026) | 31% |
| Leading preservation format | Cryobags |
| Format share (2026) | 38% |
| Leading automation level | Semi-automated systems |
| Automation share (2026) | 41% |
| Leading end user | Biopharma developers |
| End-user share (2026) | 33% |
| Leading workflow stage | Process development |
| Workflow share (2026) | 28% |
Source: Future Market Insights, 2026.
Manufacturing and quality teams are replacing manual thawing and isolated lab routines. Cell therapy batches move across various sites operators and delivery points, where variation in thaw timing and handling can create data gaps and review issues. Stronger testing platforms can reduce this risk.
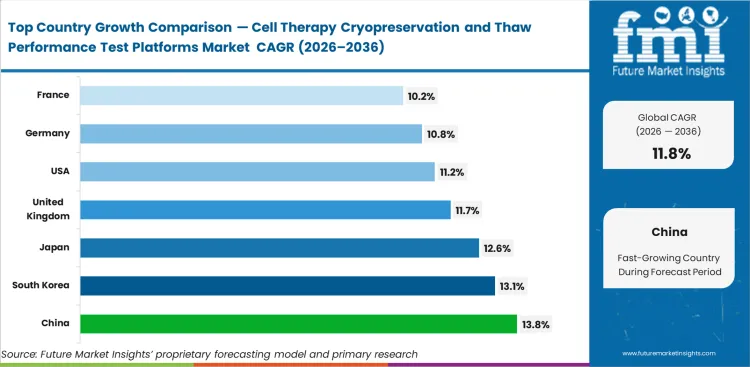
China is expected to record 13.8% CAGR through 2036, followed by South Korea at 13.1% and Japan at 12.6%. The United Kingdom is projected at 11.7%, the United States at 11.2%, Germany at 10.8%, and France at 10.2% by the forecast period.
Cell therapy cryopreservation and thaw performance test platforms market covers tools used to check how frozen cell therapy products behave during freezing, storage and thawing. Scope includes instruments and workflow tools that measure viability, recovery and temperature control.
This market includes post-thaw analyzers, automated thawing units, controlled-rate freezing systems, thermal mapping tools, and assay platforms used in cell therapy development and quality control. Method-transfer support and comparability testing tied to those platforms also fall within scope when they support freeze-thaw performance assessment. Related platform demand also connects with freeze thaw system.
General bio storage freezers, passive cold-chain packaging, transport-only shippers, and broad laboratory instruments are excluded when they have no cryopreservation or thaw-testing role. Cell expansion systems, fill-finish lines, and therapy revenue are excluded from this market because the report focuses on testing platforms. Storage-only tools are relevant to adjacent areas such as cryopreservation systems.
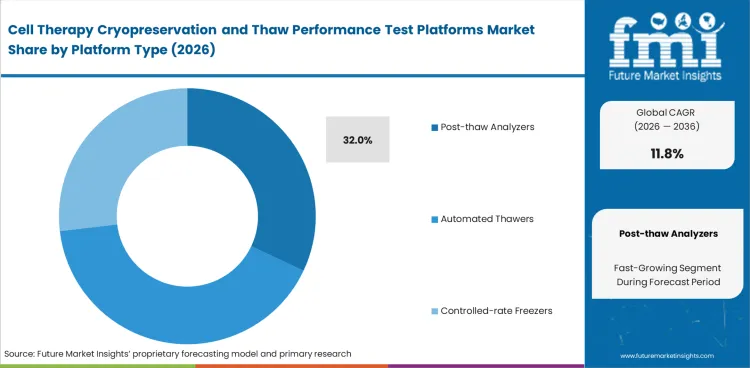
Release teams need more than visual checks when thaw results must remain consistent across sites, operators, and batches. Post-thaw analyzers lead because they measure viability and recovery after thaw and short hold periods. In 2026, post-thaw analyzers are expected to account for 32% share. Automated thawers and controlled-rate freezers are part of the workflow, through analyzers play the clearest role in release review. They bring measurement and control to a sensitive handling step.
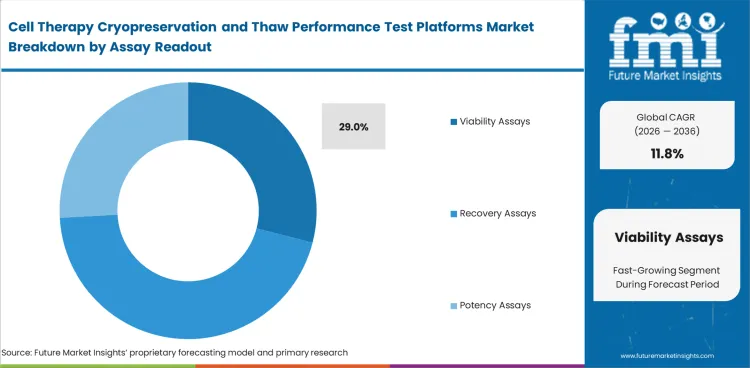
Viability assays are likely to represent 29% of the market in 2026. Post-thaw work starts by checking whether enough cells remain viable for the next step. Viability assays stay in front because they answer that question faster than more detailed methods. Recovery, phenotype, potency, and stress testing provide deeper analytical insight. Viability usually comes first because it serves as the initial check before further evaluation. Preference in this category centers on methods that are easy to repeat and compare across runs.
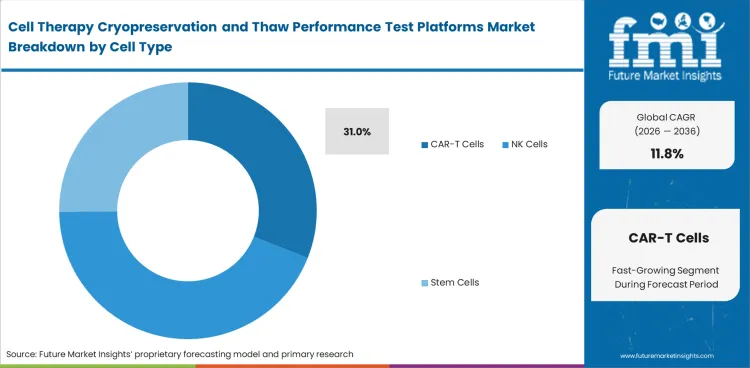
Commercial cell therapy work raises the importance of freeze-thaw control when cell types are harder to recover after storage. CAR-T cells lead platform demand because handling quality can affect dose availability and later comparability work. CAR-T cells are projected to hold 31% share in 2026. NK cells, stem cells, and MSC products expand the broader application base, though early platform selection often follows CAR-T requirements. This area depends on strong alignment between thaw method, measured recovery, and downstream use.
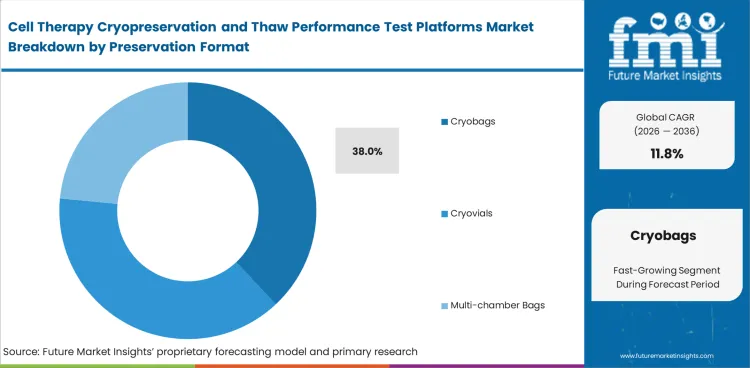
Cryobags are projected to secure 38% share in 2026. Container choice affects more than storage convenience. Cryobags lead because many cell therapy workflows use larger fill volumes and need closed handling. Bag-based formats force teams to pay closer attention to thaw uniformity, temperature control, and downstream handling than small-vial routines usually require. Cryovials still remain useful in early lab work and smaller lots. Bag use grows once programs move closer to repeatable manufacturing and administration readiness, where handling discipline matters more.
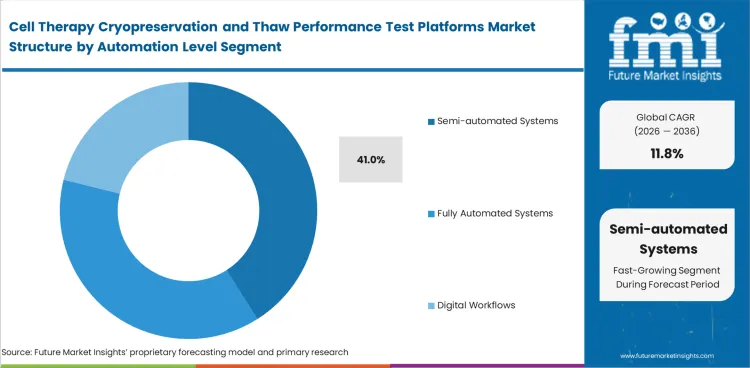
Many laboratories move beyond manual thawing before they are ready for a fully automated line. Semi-automated systems fit that stage because they improve timing control and repeatability without requiring a full process rebuild. Semi-automated systems are projected to hold 41% share in 2026. Manual setups remain common where throughput is low. Fully automated platforms gain more attention where scale is higher and workflow standardization is broader. Semi-automated tools appeal to buyers who need a measurable improvement now while still working within familiar lab routines and tighter capital limits.
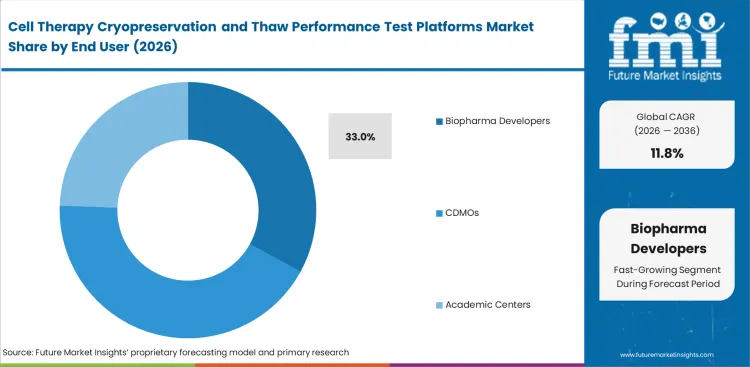
Biopharma developers are closest to the platform decision that shape later evidence quality. They define thaw methods, decide which checks matter, and set the handling rules others may later follow. Biopharma developers are anticipated to emerge with 33% market share in 2026. CDMOs buy actively and often work within methods first defined by their clients. Academic centers and hospital laboratories contribute to demand though platform selection in those settings is usually narrower and tied to specific programs. Lead position comes from direct ownership of process decisions and from the cost of carrying weak thaw logic too far into development, which also keeps this group closely linked with broader work in automated and closed cell therapy processing systems.
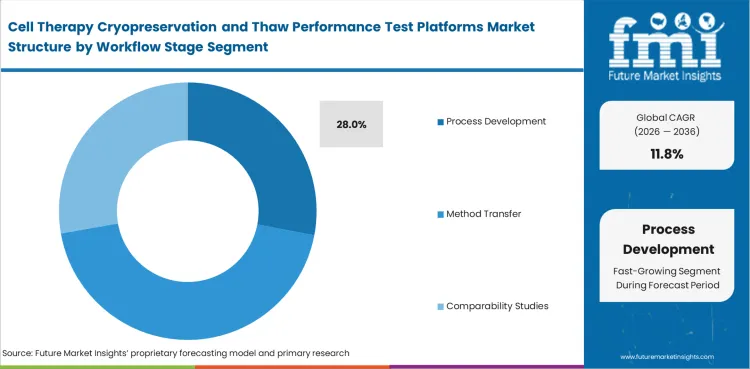
Process development leads because it is still the stage where thaw conditions, assay logic, and handling routines can be compared before later adjustment becomes difficult. By 2026, process development is expected to contribute 28% of total market share. Method transfer, lot release, and pre-infusion checks become more important later, yet many of those needs are shaped by earlier decisions. Weak setup at this stage tends to reappear in comparability work and deviation review.
Buyers asking whether thaw-performance platforms still sit at the edge of quality control should recalibrate. FDA’s 2025 regulatory action for WASKYRA states that post-thaw drug-product data were used to demonstrate post-thaw stability. FDA still required an additional in-use drug-product stability study that included the administration set filter and assessed viability under the administration conditions described in the BLA. FDA’s 2025 Q1 draft guideline for advanced therapy medicinal products also states that, for live cell-based products stored frozen, viability should be measured after thaw as part of stability studies, and that changes in viability and cell concentration should be considered for subsequent processing or dosing.[1]
Based on the regional analysis, the Cell Therapy Cryopreservation and Thaw Performance Test Platforms market is segmented into North America, Europe, Asia Pacific, Latin America, and Middle East & Africa across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 13.8% |
| South Korea | 13.1% |
| Japan | 12.6% |
| United Kingdom | 11.7% |
| United States | 11.2% |
| Germany | 10.8% |
| France | 10.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
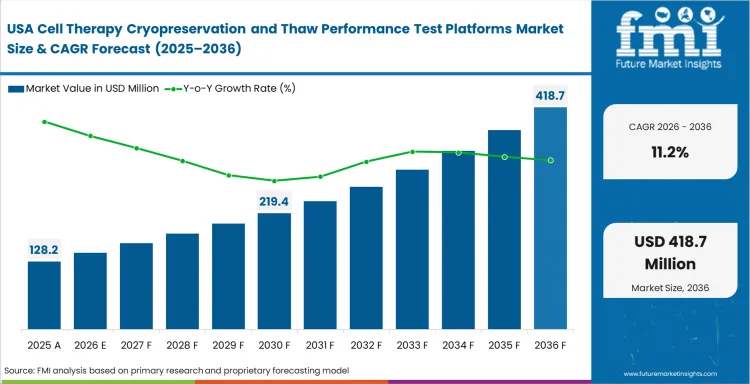
North America stays important because commercial cell therapy handling is already advanced enough for thaw-performance control to matter beyond early lab work. Buyers here are usually deciding which platform setup can support cleaner handoffs across process development, QC and manufacturing.
Report coverage also includes Canada and other North American countries. Activity there generally follows the United States, though the installed base is smaller. Interest rises as advanced therapy work becomes more formalized and site-to-site comparability gains weight.
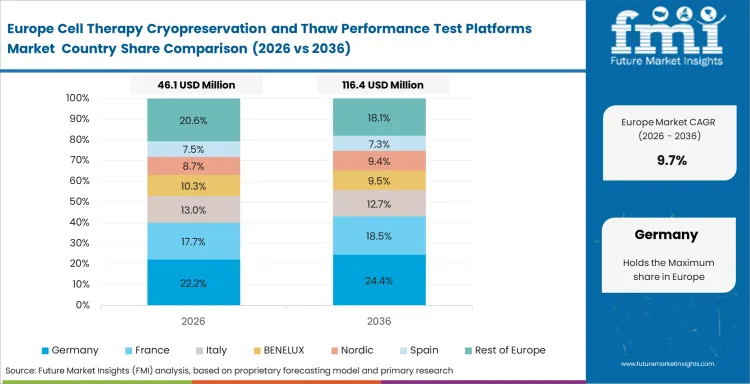
Europe follows a deliberate pace because many buying decisions are tied to qualification discipline, cross-border operating practice, and the need to fit new tools into documented workflows. Demand is established across the region, and replacement cycles carry more importance than in faster-rising parts of Asia.
The FMI report coverage also includes Italy, Spain, Switzerland, the Netherlands, Belgium, and Nordic countries. Adoption across those areas usually rises when advanced therapy work moves from academic concentration toward more repeatable manufacturing and release-oriented handling.
Asia Pacific is growing faster because cell therapy activity is expanding across several countries, while many programs are still building workflow discipline and choosing platform setups. Buyers in this region often place more value on methods that can standardize quickly across growing teams and new facilities. Better thaw control at an early stage can selection more strongly than in regions with deeper legacy setup.
Report coverage also includes Australia, Singapore, India, and other Asia Pacific countries. Markets across that group usually differ by how fast trial activity turns into documented manufacturing practice and how quickly post-thaw checks move from optional use into routine work.
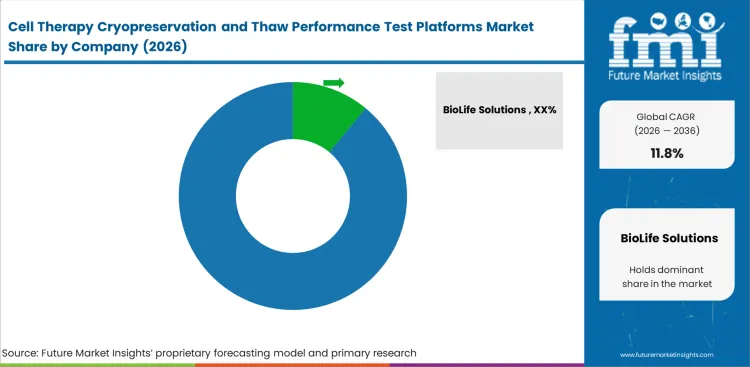
Competition in this space centers on thaw control, freezing discipline, post-thaw cell measurement, and data traceability meet. BioLife Solutions, Thermo Fisher Scientific, Cytiva, Beckman Coulter Life Sciences, ChemoMetec, PHC Corporation, and Sartorius approach the opportunity from different technical positions. Platform selection depends on whether a tool can produce repeatable thaw or viability evidence, fit existing laboratory routines, and support method adoption without triggering a long requalification process. Commercial strength depends on easier validation and stable day-to-day use.
Developers gain more influence when they want to avoid one narrow method path from development through release. Large therapy programs compare how well platforms export data, support method transfer, and fit more than one container or assay logic. Vendors want early placement so their tools remain embedded through later stages. Concentration is likely to remain moderate through 2036 because thawing, freezing, counting, and support analytics shape platform selection from different directions. Related investment in high-density cell banking also keeps adjacent suppliers in view..
| Metric | Value |
|---|---|
| Quantitative Units | USD 180 million to USD 549 million, at a CAGR of 11.8% |
| Market Definition | Cell Therapy Cryopreservation and Thaw Performance Test Platforms Market covers tools used to test how cryopreserved cell therapy products perform during freezing, storage, thawing, and immediate post-thaw handling. Scope includes platforms tied to viability, recovery, thermal behavior, method repeatability, and readiness for downstream use. Storage-only hardware without a test role is excluded. |
| Platform Type Segmentation | Post-thaw Analyzers, Automated Thawers, Controlled-rate Freezers, Thermal Mappers, Integrated Workstations, Cold-chain Simulators |
| Assay Readout Segmentation | Viability Assays, Recovery Assays, Potency Assays, Phenotype Assays, Sterility Assays, Stress Assays |
| Cell Type Segmentation | CAR-T Cells, NK Cells, Stem Cells, MSC Products, iPSC Products, Dendritic Cells |
| Preservation Format Segmentation | Cryobags, Cryovials, Multi-chamber Bags, Cartridges, Plates |
| Automation Level Segmentation | Manual Setups, Semi-automated Systems, Fully Automated Systems, Digital Workflows |
| End User Segmentation | Biopharma Developers, CDMOs, Academic Centers, Hospital Labs, Reference Labs |
| Workflow Stage Segmentation | Process Development, Method Transfer, Comparability Studies, Lot Release, Pre-infusion Checks, Excursion Studies |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Countries Covered | United States, China, Japan, South Korea, United Kingdom, Germany, France, and 40 plus countries |
| Key Companies Profiled | BioLife Solutions, Thermo Fisher Scientific, Cytiva, Beckman Coulter Life Sciences, ChemoMetec, PHC Corporation, Sartorius |
| Forecast Period | 2026 to 2036 |
| Approach | Analysis combines primary interviews with manufacturing, QC, and process development roles; review of guidance, standards, investor disclosures, and technical literature; and forecast validation against therapy activity, platform relevance, and adjoining cell therapy workflow demand. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America
Europe
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is the Cell Therapy Cryopreservation and Thaw Performance Test Platforms Market in 2026?
Industry valuation is estimated at USD 180 million in 2026. Figure shows a focused testing market with a clear role in cell therapy development, handling, and quality work.
What will the market be worth by 2036?
Valuation is projected to reach USD 549 million by 2036. Increase reflects broader use of controlled thawing and post-thaw testing across more programs and sites.
What CAGR is expected from 2026 to 2036?
Projected CAGR for the forecast period is 11.8%. Pace suggests steady expansion rather than a short burst of equipment buying.
Which Platform Type segment leads in 2026?
Post-thaw analyzers lead Platform Type. Their share is expected to reach 32% in 2026 because they give a direct check on cell condition right after thaw.
Which Assay Readout segment leads in 2026?
Viability assays lead Assay Readout with an expected 29% share in 2026. Most labs start here because cell health is the first thing they need to confirm.
Which Cell Type segment leads in 2026?
CAR-T cells lead Cell Type in 2026 with 31% share. Commercial use and tighter handling needs keep this segment at the center of platform demand.
Which Preservation Format segment leads in 2026?
Cryobags lead Preservation Format. They are expected to represent 38% of the market in 2026 because they fit larger-volume and closed-handling workflows.
Which Automation Level segment leads in 2026?
Semi-automated systems lead this segment with 41% share in 2026. Many labs choose them because they improve control without forcing a full automation build.
Which End User segment leads in 2026?
Biopharma developers lead End User in 2026. Market share for this group is expected to reach 33% because they set many of the methods used later across the workflow.
Which Workflow Stage segment leads in 2026?
Process development leads Workflow Stage with 28% share in 2026. Early method choices often carry into transfer, comparability, and later quality work.
What is this market?
This market covers tools used to test how frozen cell therapy products behave before, during, and after thaw. It includes analyzers, thaw systems, temperature checks, and related testing tools used to judge viability, recovery, and repeatability.
Why does thaw testing matter in cell therapy?
Frozen cells can lose quality when thawing is poorly controlled. Testing helps labs see whether the product still performs as expected before it moves to the next step.
Why are post-thaw analyzers important?
They turn a sensitive handling step into a measurable check. That makes batch comparison easier and gives labs a clearer basis for review.
Why do viability assays lead this market?
Viability is often the first question after thaw. If that result is unclear, later assay results become harder to trust.
Why do cryobags matter so much?
Cryobags fit many practical cell therapy workflows. Their size and handling needs make thaw control more important than it is in a small-vial setup.
Why do semi-automated systems lead instead of fully automated systems?
Many labs want better timing control and repeatability without rebuilding the whole process. Semi-automated tools fit that need better than manual practice and cost less than full automation.
Why do biopharma developers buy the most?
They usually define the methods first. Choices made at that stage often carry into CDMO work, internal QC, and later transfer activity.
Why does process development lead Workflow Stage?
Process development is where teams still have room to test and compare methods. Weak decisions made there can create extra work later.
Which country grows fastest?
China grows fastest, with a projected CAGR of 13.8% through 2036. Local cell therapy activity is still expanding, so more first-time platform adoption is taking place.
How does the United States differ from China in this market?
United States demand comes from a deeper installed base and more replacement-led buying. China still has more room for first-use adoption while programs and facilities continue to expand.
Why does the United Kingdom outpace Germany and France?
Advanced therapy activity gives the United Kingdom a stronger base in this niche. Germany and France remain important, but adoption there often moves through a slower qualification path.
What sits outside the scope of this report?
General storage equipment, passive cold-chain packs, transport-only shippers, and therapy revenue are outside scope. Report focuses on testing platforms, not the therapies themselves.
Why is repeatability such a big issue here?
Cell therapy work often moves across sites, operators, and handling steps. A method that works only in one lab is harder to defend when product use becomes broader.
What makes this market different from general lab equipment?
General lab tools may support handling, but this market is built around checking freeze-thaw performance. That testing role is what sets it apart.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.