Surgical Robotics Performance and Safety Validation Test Platforms Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
The Surgical Robotics Performance And Safety Validation Test Platforms Market Is Segmented By Test Type (Electrical Safety, EMC, Software V&V, Usability, Cybersecurity), Platform Type (Integrated Rigs, Motion Benches, Sensor Arrays, Simulation Suites, Hybrid Cells), Robot Type (Multi-Port, Single-Port, Orthopedic, Spine, Microsurgery), End User (OEM Labs, Test Labs, Cros, Hospitals, Research Institutes), And Validation Stage (Design Verification, System Validation, Instrument Qualification, Preclinical Validation, Post-Market Testing) And Region. Forecast For 2026 To 2036.
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Surgical Robotics Performance and Safety Validation Test Platforms Market Size, Market Forecast and Outlook By FMI
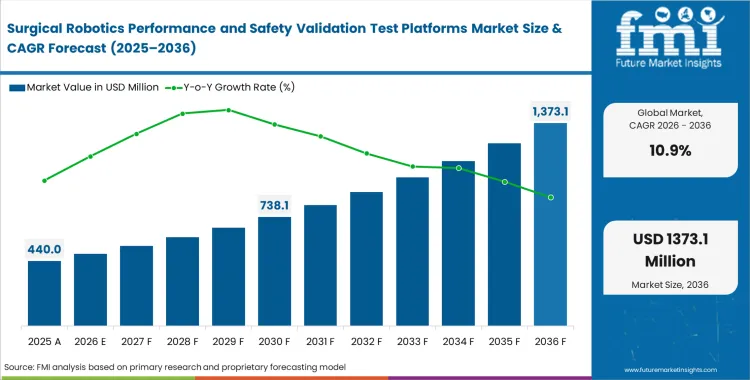
In 2025, the surgical robotics performance and safety validation test platforms market was valued at USD 400 million, with rising demand, it is now set to cross USD 440 million in 2026 at a CAGR of 10.9% during the forecast period. Revenue expansion propels total valuation to USD 1,240 million through 2036 as regulatory agencies mandate continuous software verification alongside physical hardware checks.
Summary of Surgical Robotics Performance and Safety Validation Test Platforms Market
- Market Snapshot
- The surgical robotics performance and safety validation test platforms market was valued at USD 400 million in 2025 and is projected to reach USD 1,240 million by 2036.
- The market is expected to expand at a 10.9% CAGR from 2026 to 2036, creating an incremental opportunity of about USD 800 million over the period.
- This is a compliance-driven Medtech infrastructure category centered on test rigs, integrated validation benches, and verification software used to prove safety and essential performance of robotically assisted surgical systems.
- The category is structurally shaped by IEC 80601-2-77, FDA device-review expectations, and the widening validation burden around software, usability, EMC, cybersecurity, and clinical-performance confirmation.
- Demand and Growth Drivers
- Demand is rising because surgical robotics submissions now require broader validation evidence across design verification, system validation, and safety-performance testing than earlier-generation surgical equipment.
- Adoption of dedicated validation platforms is increasing because FDA-cleared systems increasingly combine robotic motion control, navigation, imaging, software, accessories, and connected components that cannot be qualified through simple bench testing alone.
- Installed-base expansion is reinforcing spend, with Intuitive reporting 12,000+ systems in hospitals globally and 3.2M+ procedures in 2025, which supports more upgrade, revalidation, accessory-qualification, and post-change testing activity.
- Projected market development for the leading countries include, India at an anticipated 13.1% CAGR, likely followed by China at an expected 12.6%, the United States at 11.8%, South Korea at 10.4%, Germany at 9.8%, the United Kingdom at 9.3%, and Japan at 8.9%.
- Growth is moderated by high capital intensity, accreditation needs, long OEM qualification cycles, and the fact that many buyers still combine in-house labs with outsourced third-party validation programs.
- Product and Segment View
- The market covers electrical safety, EMC, software verification and validation, usability, cybersecurity, motion and force measurement, instrument qualification, and hybrid test environments built specifically for surgical robotic systems and subsystems.
- These platforms are used across multi-port laparoscopy, single-port surgery, orthopedic robotics, spine robotics, and emerging microsurgical robotic programs.
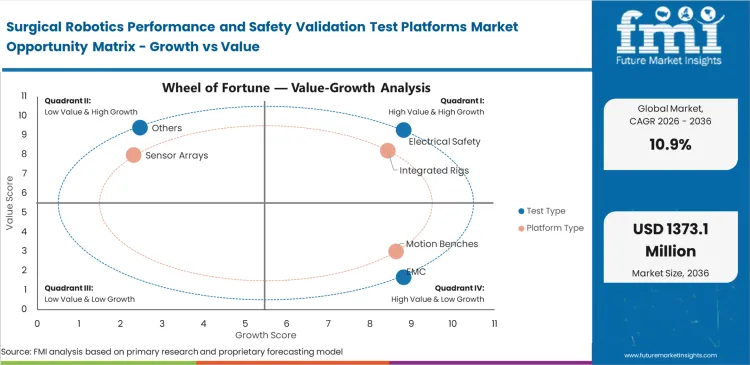
- Supported by mandatory core safety compliance under the IEC 60601 / 80601 framework, electrical safety is set to lead the test type segment with an estimated 26.0% share.
- Integrated rigs are poised to lead the platform type segment with an assessed 31.0% share, because OEMs prefer single-workflow benches that combine motion, electrical, environmental, software, and data-acquisition functions.
- Multi-port systems are predicted to lead the robot type segment with an anticipated 49.0% share, reflecting the still-dominant installed base of legacy and current da Vinci-class surgical platforms.
- As original manufacturers still keep first-pass verification and change-control validation close to product engineering teams, OEM labs are projected to lead the end user segment with an expected 43.0% share.
- Design verification is expected to lead the validation stage segment with an estimated 34.0% share, as it is the first high-spend point where motion, software, EMC, electrical, and instrument interfaces are locked down before broader system validation.
- The scope includes standalone and integrated validation platforms for surgical robotic systems and accessories, but excludes clinical robotic procedures revenue, routine sterilization equipment, and generic hospital biomedical test gear with no surgical-robot specialization.
- Geography and Competitive Outlook
- China, India, and South Korea are the fastest-expanding build-out markets, while the United States remains the deepest current revenue base for advanced validation platforms.
- Competition is shaped by regulatory breadth, accredited lab networks, robotics-specific standards expertise, and the ability to combine mechanical, electrical, software, EMC, usability, and AI-related assessments in one program.
- Key companies include Intertek, TÜV SÜD, SGS, Element Materials Technology, and NAMSA, with Intertek leading due to visible positioning in medical robots testing and certification.
- The market remains moderately fragmented because OEMs split spend across specialized third-party partners, internal engineering labs, and multi-service certification groups rather than relying on a single dominant global vendor.
R&D directors at equipment companies face intense pressure to qualify new sensor arrays quickly using compliant IEC 80601-2-77 testing platforms. Delaying validation cycles means missing crucial submission windows for new product launches. Evaluating these robot-assisted surgery validation systems requires more than simple stress testing. Engineering teams need accurate surgical robot procedures evaluation to ensure their equipment behaves predictably under real operating room conditions. Purchasing officers sourcing outsourced surgical robot testing services often discover that standard labs lack the specific fixtures needed for joint articulation checks.
Once a facility combines a motion analysis platform for surgical robots with concurrent software debugging, the entire qualification timeline shrinks from months to weeks. System architects trigger this shift when they stop evaluating hardware and software in isolation. Unified testing environments allow engineers to catch calibration errors before they force a complete redesign.
India is forecasted to lead global expansion at an estimated 13.1% as major equipment builders move their preclinical work to specialized testing centers in the country. China is likely to follow closely with a scale of 12.6% growth rate driven by aggressive domestic manufacturing initiatives. United States test labs poised to record an estimated 11.8% CAGR while managing complex FDA submission protocols. South Korea set to track an inclination of 10.4% as local technology companies invest in custom motion benches. Germany predicted to post a 9.8% rise by focusing on strict European compliance standards. United Kingdom facilities expected to manage an estimated 9.3% rate through focused surgical robot software V&V tools programs. Japan is projected to witness at 8.9%, characterized by heavy reliance on legacy safety assessment protocols.
Segmental Analysis
Surgical Robotics Performance and Safety Validation Test Platforms Market Analysis by Test Type
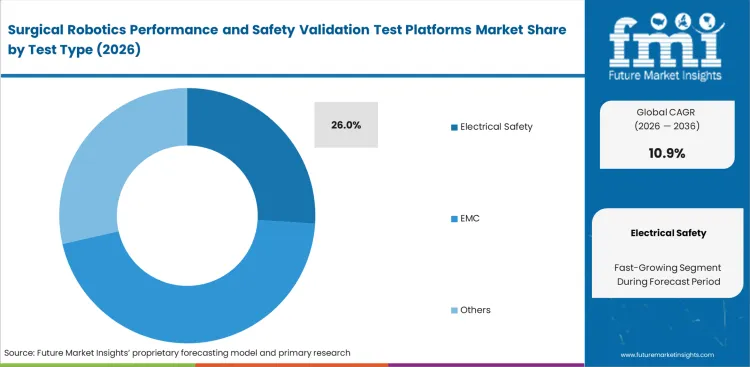
Regulatory compliance prioritize baseline electrical checks before any mechanical testing begins. Confirming power stability prevents sudden arm movements during active procedures. The electrical safety category is expected to hold 26.0% share in 2026 as this baseline stability remains a strict prerequisite for clearance. Quality managers rely on a dedicated surgical robot electrical safety test bench to catch voltage drops early in the design phase. Passing a standard internal electrical test guarantees almost nothing about how the equipment will behave when plugged into an older hospital's noisy power grid. Engineers must use surgical robot EMC testing systems to replicate these unpredictable environments. Missing these external interference checks forces companies to redesign their power supplies right before a planned product launch. Adopting AI based surgical robots requires even stricter power testing because their heavy computational loads create distinct electrical vulnerabilities.
- Interference Identification: Advanced power analyzers detect tiny voltage drops that could disrupt motor function. Quality managers avoid costly redesigns by finding these vulnerabilities early.
- Simulated Hospital Grids: Testing environments mimic the unstable electricity found in older medical facilities. Engineers ensure the equipment maintains accuracy despite power fluctuations.
- Continuous Monitoring: Teams run automated scripts that test electrical limits over thousands of hours. Using a pre-clinical imaging system alongside these tests confirms that sensors remain stable under stress.
Surgical Robotics Performance and Safety Validation Test Platforms Market Analysis by Platform Type
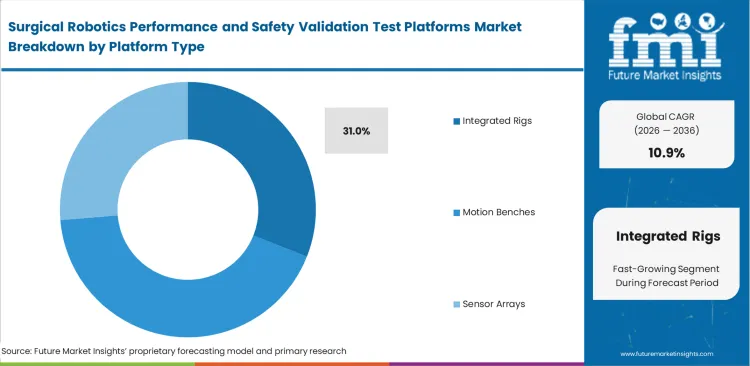
Engineering teams are no longer setting up test floors around hardware and software as separate validation tasks. That shift keeps demand firm for integrated rigs, which are estimated to account for 31.0% of market share in 2026. These systems let developers run control code against real arm resistance in the same environment, making calibration faults visible before they move into later-stage validation. The buying decision, though, extends well beyond mechanical precision. Test operators must capture and align every micro-movement, response signal, and control adjustment in real time. That creates a substantial data-management load, especially for smaller engineering firms that lack strong internal IT support. When storage architecture and traceability workflows are poorly planned, debugging slows quickly and teams can spend weeks isolating the source of a single software fault. The requirement is even stricter in robotics assisted telesurgery, where remote-latency measurement depends on tightly synchronized data logging across the full testing sequence.
- Hardware Synchronization: Rigs feed physical sensor data directly into software debugging tools. Software developers fix code errors faster when they can see the physical result immediately.
- Latency Measurement: Specialized fixtures calculate the delay between a command and the actual arm movement. Regulatory reviewers demand this specific metric before approving remote capabilities.
- Data Logging Automation: Servers capture every physical and digital event during long test runs. Implementing preclinical imaging data alongside motion logs gives investigators a complete picture of system failures.
Surgical Robotics Performance and Safety Validation Test Platforms Market Analysis by Robot Type
General surgery applications continue to drive the highest volume of equipment orders across testing facilities. Facility directors configure their largest motion benches primarily to handle these traditional four-arm setups. Testing multiple moving arms simultaneously requires extensive physical space and highly complex collision-avoidance software checks. Adapting these standard tests for a robot assisted surgical microscope requires entirely different optical measurement fixtures, pushing the multi-port category as anticipated to capture 49.0% of the market in 2026. This dominant position will persist until device makers resolve the severe heat dissipation challenges currently limiting single-port system approvals.
- Collision Simulation: Software algorithms force robotic arms into potential crash scenarios. Test engineers verify that built-in safety limits prevent physical impact between instruments.
- Cable Friction Analysis: Repeated motion tests measure how quickly internal wires degrade inside tight spaces. Maintenance planners use this data to set mandatory part replacement schedules.
- Heat Dissipation Tracking: Thermal sensors monitor temperature buildup during continuous operation. Designers adjust cooling mechanisms based on these extended endurance runs.
Surgical Robotics Performance and Safety Validation Test Platforms Market Analysis by End User
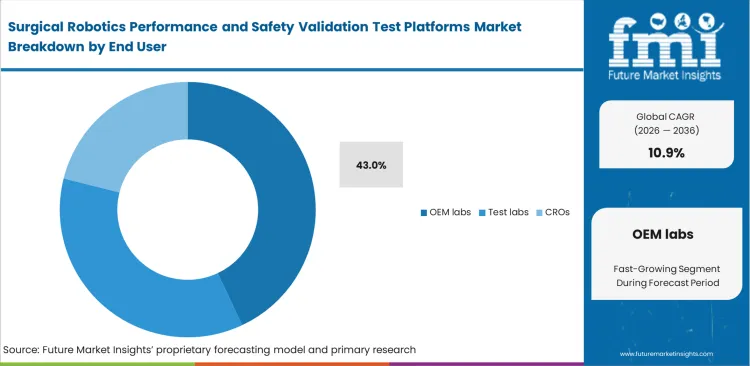
Large device manufacturers fiercely protect their unreleased designs by keeping early-stage testing strictly behind closed doors. R&D directors build massive internal testing floors to hide their upcoming prototypes from competitors and independent contractors. Driven by this need for absolute secrecy, OEM labs are projected to secure 43.0% share in 2026. Maintaining these private facilities requires an ongoing capital investment in expensive new measurement technology. Most of these private labs actually rely on an external surgical robot cybersecurity testing platform to handle their network security audits. Contracting with a specialized preclinical CRO allows these equipment builders to meet strict FDA hacking-resistance guidelines without hiring full-time security experts. Testing radically new swarm robotics microsurgery systems routinely pushes these internal labs past their technical limits.
- Proprietary Design Protection: Internal labs prevent external contractors from viewing unreleased product features. R&D directors maintain total control over their intellectual property.
- Cybersecurity Delegation: Teams hire outside experts to attempt authorized hacks on the equipment software. Compliance officers require these external audits to satisfy regulatory security demands.
- Continuous Hardware Iteration: Engineers walk prototypes directly from the design desk to the test bench. Project managers eliminate the shipping delays associated with external laboratories.
Surgical Robotics Performance and Safety Validation Test Platforms Market Analysis by Validation Stage
Catching structural flaws on a computer screen costs a fraction of fixing a fully assembled machine. Engineering managers allocate large initial budgets to software modeling to confirm the basic architecture works before purchasing any raw materials. In 2026, the design verification stage is estimated to represent 34.0% of total market share based on these heavy upfront software investments. Project leaders frequently mistake excellent digital simulation results as proof that physical testing will pass smoothly. Regulatory bodies ignore perfect digital blueprints and demand rigorous real-world checks using an instrument qualification platform for robotic surgery. Companies that drain their entire testing budget on digital verification often scramble to fund their final compliance runs. Approving advanced robotic assisted endovascular systems demands extensive physical endurance data regardless of early simulation success.
- Blueprint Modeling: Digital environments run mathematical tests on proposed equipment designs. Engineering managers catch structural weaknesses before purchasing any raw materials.
- Algorithm Verification: Coders use automated scripts to verify that basic software logic operates correctly. Development teams resolve programming conflicts early in the project timeline.
- Physical Compliance Transition: Planners must allocate sufficient time and money for real-world testing. Regulatory directors ensure that digital success translates into physical safety documentation.
Surgical Robotics Performance and Safety Validation Test Platforms Market Drivers, Restraints, and Opportunities
Strict medical software regulations compel equipment builders to upgrade their testing capacity immediately. Quality assurance teams must run complete regression tests for every minor code patch to prove the update breaks nothing in the existing system. Delaying these mandatory checks stops hospitals from getting necessary security fixes and operational improvements. Operating a specialized medical device tester lets engineering teams automate these repetitive code checks overnight. Manual testing methods simply take too long. Companies relying on human testers fall months behind competitors who automate their software validation. The commercial stakes rise because a delayed software patch can pause a company's entire sales cycle. Device manufacturers now view rapid software testing as a core business requirement rather than just a basic compliance step. Without continuous testing environments, engineering teams spend more time documenting changes than writing actual code.
A severe shortage of engineers who understand both mechanical stress testing and medical compliance rules slows down laboratory expansion. Finding professionals who can interpret physical test data and write FDA-compliant reports remains incredibly difficult. Facility managers spend years teaching traditional mechanical engineers how to handle strict medical regulatory paperwork. New software scripts handle basic daily electrical checks just fine. Diagnosing why a specific robotic joint failed under pressure still requires an experienced human engineer. This specific talent gap creates long waiting lines for testing slots at major laboratories. Companies wanting to launch new equipment must book their physical testing time months in advance. The lack of qualified testing personnel acts as a hard speed limit on how fast the medical robotics industry can introduce new hardware.
- Simulation Software Licensing: Startups can verify their early robotic designs on rented digital platforms instead of buying expensive physical motion benches. R&D directors save millions in initial capital costs while still gathering enough solid performance data to satisfy potential investors and internal engineering reviews.
- Dedicated Hacking Services: Medical device builders willingly pay external security experts to find software weaknesses in their operating systems. Compliance officers need these independent audit reports to pass strict new network safety rules and prevent costly data breaches in connected hospital environments.
- Automated Compliance Paperwork: Software that turns raw motion data into formal regulatory documents eliminates hundreds of hours of manual data entry. Working with a specialized preclinical CRO that uses these automated reporting tools helps regulatory directors avoid formatting errors and accelerate final government submissions.
Regional Analysis
Device makers must align their testing budgets with the distinct approval rules of their target geographies. Every health authority demands different proof before allowing new equipment into operating rooms. Testing facility directors map their validation capabilities directly to these varying national standards to ensure a smooth clearance process. Based on regional analysis, Surgical Robotics Performance and Safety Validation Test Platforms is segmented into North America, Asia Pacific, and Europe across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 13.1% |
| China | 12.6% |
| United States | 11.8% |
| South Korea | 10.4% |
| Germany | 9.8% |
| United Kingdom | 9.3% |
| Japan | 8.9% |
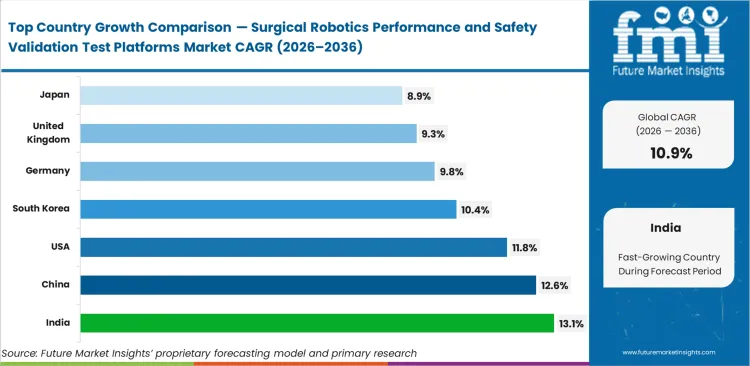
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Surgical Robotics Performance and Safety Validation Test Platforms Market Analysis
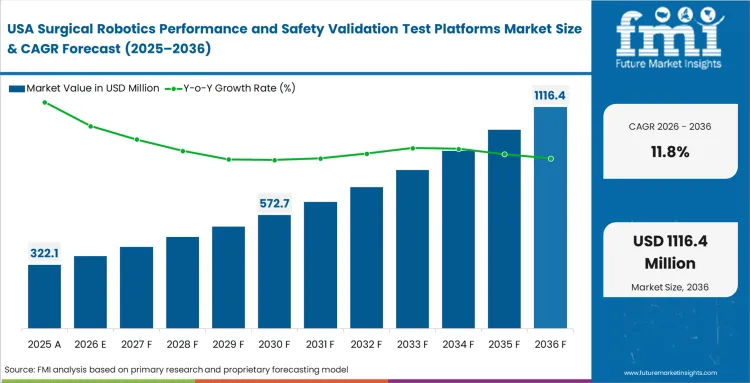
Strict FDA approval rules dictate how testing happens across this geography. Medical tech companies structure their entire testing timeline around the specific data the FDA requires for its 510(k)-clearance process. Building a dedicated facility here means working closely with local compliance experts. Engineering teams spend heavily on human factors testing to prove that surgeons will not make critical errors under high-pressure conditions.
- United States: High expectations for software safety force local companies to spend heavily on automated testing environments. Demand for testing platforms in the United States is anticipated to rise at a CAGR of 11.8% through 2036, as test lab directors constantly upgrade their computer simulations to handle smarter, faster equipment updates. Gaining early access to an advanced medical device tester gives early-stage companies a significant edge when competing against established device makers.
FMI's report includes extensive data on testing lab expansion in Canada and Mexico. Cross-border agreements now enable device builders to run distinct software checks simultaneously across allied facilities, saving valuable time.
Asia Pacific Surgical Robotics Performance and Safety Validation Test Platforms Market Analysis
Pressure to lower costs and expand local manufacturing shapes the testing environment across these countries. Western companies actively move their physical testing work to Asian labs to reduce overhead. Local governments provide significant funding to build specialized medical testing infrastructure. Analyzing the region reveals a sharp divide between countries focusing on high-volume mechanical stress tests and those building advanced hubs for checking software reliability.
- India: The country is projected to witness 13.1% CAGR in the testing platforms sector through 2036, due to western brands sending their repetitive mechanical stress tests to local centers to keep development budgets under control. Managing these high-volume outsourced agreements requires rock-solid data transfer security so proprietary designs stay safe. Finalizing reliable India preclinical medical device testing services contracts opens up massive revenue streams for regional testing providers.
- China: Heavy government funding for homegrown medical technology fuels the rapid construction of new testing centers, propelling the adoption of testing platforms in China expected to move ahead at a CAGR of 12.6% from 2026 to 2036. Local labs scale their operations quickly by securing high-volume China preclinical medical device testing services agreements. This rapid scaling puts these facilities on track to handle global testing overflow within the next five years.
- South Korea: South Korea is set to record a CAGR of 10.4% in testing platforms during the assessment period, with regard to the local tech firms use their deep background in consumer electronics to construct extremely accurate sensor-checking stations. Engineering managers here concentrate intensely on verifying the tiniest physical movements. Achieving this level of precision allows these labs to seamlessly support advanced robot assisted surgical microscope qualification processes.
- Japan: Cautious local regulators insist on massive amounts of physical endurance data before letting any new device enter a hospital. Sales of testing platforms in Japan are poised to grow at a CAGR of 8.9% during the assessment period as facility managers keep huge rows of physical testing benches running constantly to meet these older, stricter rules. This heavy reliance on physical testing creates a reality where digital simulation alone will not clear a device for local use.
FMI's report includes extensive data on testing lab expansion in Australia and emerging Southeast Asian hubs. Examining these smaller geographies highlights a growing trend of renting digital simulation access instead of building expensive physical labs from the ground up.
Europe Surgical Robotics Performance and Safety Validation Test Platforms Market Analysis
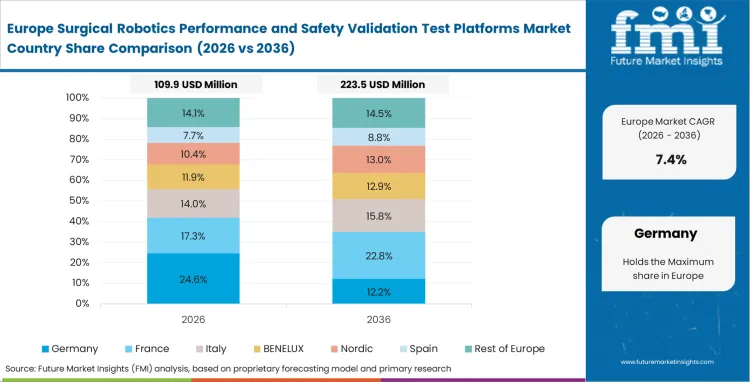
Moving to the new Medical Device Regulation rules forces massive updates to how labs test equipment here. Quality managers now have to show much more clinical proof and technical paperwork than they did a few years ago. Labs are upgrading their data tracking systems so they can trace every single test back to its exact moment of execution. Developing complex internal validation tools often occurs alongside these hardware checks.
- Germany: Exact engineering standards compel local businesses to build the most accurate motion-tracking benches possible, leading the demand in Germany expected to register a CAGR of 9.8% through 2036. Combining robotic handling tests with preclinical antibody development checks demands very specific, clean testing rooms. Setting up these specialized rooms allows domestic testing providers to secure high-value contracts from neighboring countries.
- United Kingdom: Deep ties with university research centers give local labs access to the newest ways to check complex software. Testing platforms adoption in the United Kingdom is likely to advance at a CAGR of 9.3% by 2036, as offering specialized United Kingdom preclinical medical device testing services helps smaller tech startups figure out the tricky local rules. Mastering these local rules completely changes how fast a new device moves from the lab to the operating room.
FMI's report includes extensive data on testing lab expansion in France, Italy, and Spain. Investigating these distinct national setups shows that regulators increasingly want usability tests done locally to prove that different hospital work cultures do not compromise safety.
Competitive Aligners for Market Players
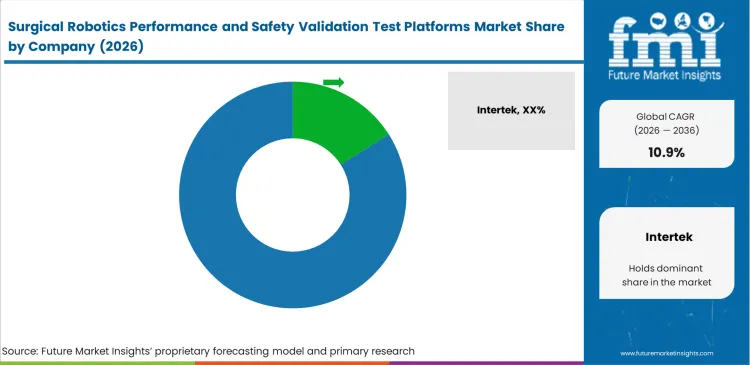
Primary validation work continues to favor global testing firms with established regulatory standing. Intertek and TÜV SÜD benefit from certified laboratory networks that hospital procurement teams and device manufacturers already know, which reduces internal resistance during vendor approval and helps procurement move faster. Smaller laboratories find it difficult to enter this tier of work because manufacturers are reluctant to attach a regulatory submission to an unproven testing partner. A failed or delayed filing can push commercialization back by months and disrupt launch schedules. Preclinical medical device testing services share analysis therefore tends to favor firms with recognized certifications, mature quality systems, and a demonstrated record in regulated device programs.
SGS has accumulated years of equipment failure data, wear patterns, and performance deviations across a broad mix of medical devices. That record helps the company build more precise validation routines and identify likely problem areas earlier in the testing cycle. New entrants lack that depth of physical test history, so many try to compete through specialized software verification or narrower digital compliance support. Some also rely on healthcare regulatory affairs outsourcing to understand how leading device manufacturers structure documentation for software-related clearances.
Splitting work across multiple providers gives manufacturers tighter control over turnaround time, pricing, and technical accountability. Independent healthcare analytical testing services remain relevant when companies want external confirmation before moving a submission forward. Demand is expected to shift further toward continuous validation of software revisions, since device code is being updated more frequently than the underlying hardware platform.
Key Players in Surgical Robotics Performance and Safety Validation Test Platforms Market
- Intertek
- TÜV SÜD
- SGS
- Element Materials Technology
- NAMSA
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 440 million in 2026 to USD 1,240 billion in 2036, at a CAGR of 10.9% |
| Market Definition | Validation test platforms encompass the specialized hardware fixtures and software simulators required to ensure automated surgical equipment operates safely and accurately before clinical use. |
| Segmentation | Test Type, Platform Type, Robot Type, End User, Validation Stage, and Region |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, Asia Pacific, Middle East and Africa |
| Countries Covered | United States, Canada, Brazil, Mexico, Germany, United Kingdom, France, Spain, Italy, Russia, China, Japan, India, South Korea, Australia, GCC Countries, South Africa |
| Key Companies Profiled | Intertek, TÜV SÜD, SGS, Element Materials Technology, NAMSA |
| Forecast Period | 2026 to 2036 |
| Approach | Annual capital expenditure on validation equipment by top medical device OEMs anchors the baseline valuation. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Segments
Test Type:
- Electrical safety
- EMC
- Software V&V
- Usability
- Cybersecurity
Platform Type:
- Integrated rigs
- Motion benches
- Sensor arrays
- Simulation suites
- Hybrid cells
Robot Type:
- Multi-port
- Single-port
- Orthopedic
- Spine
- Microsurgery
End User:
- OEM labs
- Test labs
- CROs
- Hospitals
- Research institutes
Validation Stage:
- Design verification
- System validation
- Instrument qualification
- Preclinical validation
- Post-market testing
Regions:
- Asia Pacific
- India
- China
- Japan
- South Korea
- Indonesia
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- Italy
- France
- United Kingdom
- Spain
- Benelux
- Nordics
- Central & Eastern Europe
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Turkey
- Rest of Middle East & Africa
Frequently Asked Questions
what standards apply to surgical robot safety testing?
Testing procedures follow strict guidelines outlined by regulatory bodies such as the FDA and the comprehensive IEC 80601-2-77 standard.
how are surgical robots validated before clearance?
Engineers subject the equipment to exhaustive electrical, mechanical, and software checks before compiling the data for formal regulatory submission.
what is IEC 80601-2-77?
This specific standard dictates the exact essential safety and performance requirements for all medical robots used in surgical environments.
what tests are required for surgical robots?
Regulatory authorities demand complete verification covering electrical safety, electromagnetic compatibility, usability, software reliability, and cybersecurity.
how much does surgical robot testing cost?
Securing full testing and certification services often requires substantial capital expenditure depending on the complexity of the hardware and algorithms.
who provides surgical robot certification services?
Established global testing laboratories like Intertek, TÜV SÜD, and SGS handle the majority of official certification protocols.
surgical robot verification vs validation?
Verification ensures the equipment was built exactly according to design specifications, while validation proves the equipment safely meets the user's actual clinical needs.
explain the surgical robotics validation stack from EMC to usability?
The complete stack begins with baseline electrical safety and electromagnetic compatibility checks before progressing through software debugging and final human factors testing.
best way to validate a robotically assisted surgical system for FDA submission?
Equipment builders must utilize a combination of digital simulation suites and physical instrument qualification platforms to generate comprehensive submission data.
compare third-party and in-house surgical robot validation platforms?
In-house setups protect proprietary designs during early development, whereas third-party labs provide the independent verification data that regulators ultimately demand.
what should a surgical robot OEM test before 510k submission?
Manufacturers must thoroughly test and document all software algorithms, motion precision limits, and cybersecurity defenses to satisfy submission requirements.
how fast is the surgical robotics validation platform market growing?
Sales for testing equipment and services are poised to expand at a CAGR of 10.9% during the assessment period.
list vendors for surgical robot performance testing and certification?
Leading suppliers in this specialized category include Intertek, TÜV SÜD, SGS, Element Materials Technology, and NAMSA.
buy surgical robot test bench?
Procurement directors can source specialized motion benches and integrated rigs directly from specialized testing equipment manufacturers.
request quote surgical robot validation lab?
Smaller startups frequently contact independent contract research organizations to receive customized pricing for full-system testing services.
surgical robot test platform price?
Pricing for these advanced systems varies significantly based on whether the buyer needs simple modular benches or fully integrated hybrid testing cells.
Intertek vs TÜV SÜD surgical robot testing?
Both global giants provide excellent certification services, but equipment builders often choose between them based on specific regional regulatory familiarity.
in-house vs outsourced surgical robot validation?
Major brands keep early mechanical testing internal to protect trade secrets while outsourcing complex cybersecurity checks to specialized external experts.
integrated rig vs modular test bench for surgical robots?
Integrated rigs allow engineers to debug software and hardware simultaneously, while modular benches offer cheaper, focused testing for single components.
best surgical robot validation platform?
The ideal platform depends entirely on whether the engineering team is currently focused on early design verification or final preclinical endurance testing.
alternatives to generic medical device testers for surgical robots?
Advanced surgical systems require highly customized motion arrays and specialized simulation software that generic electronic testers cannot provide.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Test Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Test Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Test Type , 2026 to 2036
- Electrical Safety
- EMC
- Others
- Electrical Safety
- Y to o to Y Growth Trend Analysis By Test Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Test Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Platform Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Platform Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Platform Type, 2026 to 2036
- Integrated Rigs
- Motion Benches
- Sensor Arrays
- Integrated Rigs
- Y to o to Y Growth Trend Analysis By Platform Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Platform Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Robot Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Robot Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Robot Type, 2026 to 2036
- Multi-port
- Single-port
- Orthopedic
- Multi-port
- Y to o to Y Growth Trend Analysis By Robot Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Robot Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- OEM labs
- Test labs
- CROs
- OEM labs
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Validation Stage
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Validation Stage, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Validation Stage, 2026 to 2036
- Design Verification
- System Validation
- Instrument Qualification
- Design Verification
- Y to o to Y Growth Trend Analysis By Validation Stage, 2021 to 2025
- Absolute $ Opportunity Analysis By Validation Stage, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Test Type
- By Platform Type
- By Robot Type
- By End User
- By Validation Stage
- Competition Analysis
- Competition Deep Dive
- Intertek
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- TÜV SÜD
- SGS
- Element Materials Technology
- NAMSA
- Intertek
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Platform Type, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Robot Type, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Validation Stage, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Platform Type, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Robot Type, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Validation Stage, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Platform Type, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Robot Type, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Validation Stage, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Platform Type, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Robot Type, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Validation Stage, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Platform Type, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Robot Type, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Validation Stage, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Platform Type, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Robot Type, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Validation Stage, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Platform Type, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Robot Type, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Validation Stage, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Platform Type, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Robot Type, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Validation Stage, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Test Type
- Figure 6: Global Market Value Share and BPS Analysis by Platform Type, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Platform Type, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Platform Type
- Figure 9: Global Market Value Share and BPS Analysis by Robot Type, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Robot Type, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Robot Type
- Figure 12: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by End User
- Figure 15: Global Market Value Share and BPS Analysis by Validation Stage, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Validation Stage, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Validation Stage
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Test Type
- Figure 32: North America Market Value Share and BPS Analysis by Platform Type, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Platform Type, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Platform Type
- Figure 35: North America Market Value Share and BPS Analysis by Robot Type, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Robot Type, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Robot Type
- Figure 38: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by End User
- Figure 41: North America Market Value Share and BPS Analysis by Validation Stage, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Validation Stage, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Validation Stage
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Test Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Platform Type, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Platform Type, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Platform Type
- Figure 51: Latin America Market Value Share and BPS Analysis by Robot Type, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Robot Type, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Robot Type
- Figure 54: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by End User
- Figure 57: Latin America Market Value Share and BPS Analysis by Validation Stage, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Validation Stage, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Validation Stage
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Test Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Platform Type, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Platform Type, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Platform Type
- Figure 67: Western Europe Market Value Share and BPS Analysis by Robot Type, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Robot Type, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Robot Type
- Figure 70: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by End User
- Figure 73: Western Europe Market Value Share and BPS Analysis by Validation Stage, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Validation Stage, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Validation Stage
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Test Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Platform Type, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Platform Type, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Platform Type
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Robot Type, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Robot Type, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Robot Type
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by End User
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Validation Stage, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Validation Stage, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Validation Stage
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Test Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Platform Type, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Platform Type, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Platform Type
- Figure 99: East Asia Market Value Share and BPS Analysis by Robot Type, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Robot Type, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Robot Type
- Figure 102: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by End User
- Figure 105: East Asia Market Value Share and BPS Analysis by Validation Stage, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Validation Stage, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Validation Stage
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Test Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Platform Type, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Platform Type, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Platform Type
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Robot Type, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Robot Type, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Robot Type
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Validation Stage, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Validation Stage, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Validation Stage
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Test Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Platform Type, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Platform Type, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Platform Type
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Robot Type, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Robot Type, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Robot Type
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Validation Stage, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Validation Stage, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Validation Stage
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

