Exoskeleton and Rehabilitation Robot Safety and Performance Test Equipment Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
The exoskeleton and rehabilitation robot safety and performance test equipment market is segmented by Test Type (Mechanical Safety, Functional Performance, Usability Validation, Sensor Calibration, Endurance Testing), Equipment Format (Benchtop Rigs, Gait Labs, Wearable Simulators, Force Platforms, Integrated Suites), Robot Type (Lower-limb Systems, Upper-limb Systems, Gait Trainers, Balance Trainers, Pediatric Systems), End User (OEMs, Contract Laboratories, Rehabilitation Hospitals, Universities, Certification Support Teams), Sales Channel (Direct Sales, Channel Partners, Service Contracts), and Region. Forecasts cover the period from 2026 to 2036.
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Exoskeleton and Rehabilitation Robot Safety and Performance Test Equipment Market Size, Market Forecast and Outlook by FMI
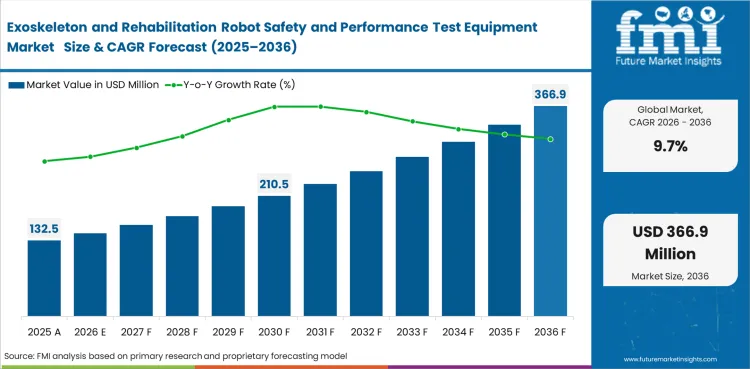
The exoskeleton and rehabilitation robot safety and performance test equipment market was valued at USD 120.8 million in 2025 and is expected to reach USD 132.5 million by 2026. It is projected to expand at a 9.7% CAGR from 2026 to 2036 and reach USD 333.0 million by 2036. Mechanical safety testing is expected to lead with a 31.0% share in 2026, while lower-limb systems are projected to account for 38.0% of the robot type segment.
Summary of Exoskeleton and Rehabilitation Robot Safety and Performance Test Equipment Market
- Demand and Growth Drivers
- Rising use of rehabilitation robots is expected to support demand for specialized test equipment as manufacturers need stronger proof of safety and repeatable performance before wider deployment.
- Tighter documentation requirements are likely to remain an important market driver, especially as software checks and revalidation work gain importance across device updates.
- Interest from hospitals and rehabilitation centers is expected to support adoption of integrated testing platforms designed to assess gait response and calibration stability.
- Product and Segment View
- Mechanical safety testing is expected to remain the leading test segment, supported by the need to verify load tolerance, fatigue resistance, and safe operation in human-contact devices.
- Lower-limb systems are projected to lead the robot type segment, driven by continued focus on gait recovery and mobility restoration programs.
- OEM validation is expected to lead the main end-use segment since most qualification spending stays concentrated in product development, release preparation, and post-modification checks.
- Geography and Competitive Outlook
- China is expected to stay a high-growth market due to stronger standards activity and rising investment in medical robotics development.
- Japan is likely to stay a key market, supported by aging-related rehabilitation demand and continued public support for care and mobility robotics.
- Suppliers with strong application support and the ability to adapt testing systems to specific device designs are expected to improve their market position through the forecast period.
- Analyst Opinion
- Sabyasachi Ghosh, Associate Vice President at FMI says, “Demand for exoskeleton and rehabilitation robot safety and performance test equipment is expected to rise as developers face tighter validation requirements and greater pressure to demonstrate repeatable device performance. Companies that can support both mechanical verification and software-linked testing workflows are likely to strengthen their position as clinical robotics programs move toward wider adoption.”
- Exoskeleton and Rehabilitation Robot Safety and Performance Test Equipment Market Value Analysis
- The exoskeleton and rehabilitation robot safety and performance test equipment market is moving from a narrow validation niche toward a more established medical device support category.
- Demand is being supported by rising use of lower-limb rehabilitation systems and the need to verify safe human-device interaction before wider placement.
- The market is gaining from stronger focus on software checks, repeatability, and documented performance across product updates.
- Spending is supported by the need for test platforms that can help manufacturers meet safety expectations, quality requirements, and clinical-use standards over time.
Exoskeleton and Rehabilitation Robot Safety and Performance Test Equipment Market Definition
This market covers equipment and software used to test the safety, performance, and repeatability of powered exoskeletons and rehabilitation robots before commercial release, during design updates, and across service life verification.
Exoskeleton and Rehabilitation Robot Safety and Performance Test Equipment Market Inclusions
Included in scope are benchtop and floor-mounted test benches, force platforms, motion analysis hardware, wearable simulators, calibration tools, environmental and endurance fixtures, data acquisition modules, validation software, and bundled engineering services delivered specifically for exoskeleton and rehabilitation robot qualification.
Exoskeleton and Rehabilitation Robot Safety and Performance Test Equipment Market Exclusions
Excluded from scope are the exoskeletons and rehabilitation robots themselves, routine physiotherapy devices without a verification function, generic laboratory instruments sold without market-specific adaptation, and broad hospital diagnostics that are not procured for robotics testing.
Exoskeleton and Rehabilitation Robot Safety and Performance Test Equipment Market Research Methodology
- Primary Research: Research design for this assessment relied on triangulation across public health demand indicators, rehabilitation burden statistics, medical device quality requirements, and current company activity in exoskeleton and rehabilitation robotics.
- Desk Research: Desk research combined official public health data, standards and regulatory updates, public company press releases, and active vendor verification from current websites.
- Market Sizing and Forecasting: Sizing was built from a bottom-up view of validation spend attached to exoskeleton and rehabilitation robot development, release testing, and service requalification.
- Data Validation and Update Cycle: Cross-checks were applied against rehabilitation need, stroke burden, reimbursement progress, standards activity, and the current footprint of leading suppliers.
Why is the Exoskeleton and Rehabilitation Robot Safety and Performance Test Equipment Market Growing?
- Clinical rehabilitation demand is expanding and pushes robot makers to prove safer and more repeatable patient interaction.
- Regulators and payers now expect stronger documentation around device quality, software control, and revalidation after updates.
- Hospital buyers increasingly prefer integrated proof of gait response, calibration stability, and service readiness before adoption.
Market expansion is tied to the steady shift of rehabilitation robotics from pilot use into broader clinical evaluation and hospital adoption. As exoskeleton and rehabilitation robot programs move closer to routine use, manufacturers need better evidence on load performance and repeat behavior under continued operation. This is raising demand for specialized systems used to test structural safety, gait response, and calibration stability before release or wider placement.
Regulatory pressure is adding another layer of support. Quality systems now place greater emphasis on traceable verification, especially for devices that combine hardware with software-driven control. That is increasing the value of equipment used for fatigue checks, control testing, and documented validation after design changes. Spending is supported by the need to maintain reliable records across product development and field updates.
Market Segmental Analysis
- Mechanical safety testing is expected to account for 31.0% of the market in 2026, reflecting the importance of load checks, fatigue validation, and safe operation in human-contact rehabilitation devices.
- Lower-limb systems are projected to lead demand with a 38.0% share, supported by continued use in gait recovery and mobility restoration programs.
- Direct sales remain the leading channel, which reflects the technical nature of procurement and the need for application support during installation and validation.
The exoskeleton and rehabilitation robot safety and performance test equipment market is segmented by test type, equipment format, robot type, end user, and sales channel. Test type includes mechanical safety, functional performance, usability validation, sensor calibration, and endurance testing. By equipment format, the market includes benchtop rigs, gait labs, wearable simulators, force platforms, and integrated suites. Robot type covers lower-limb systems, upper-limb systems, gait trainers, balance trainers, and pediatric systems.
Insights into the test type segment
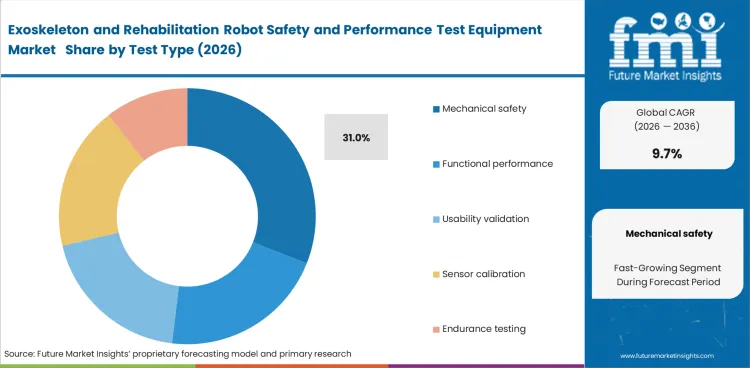
- Mechanical safety is expected to account for 31.0% share in 2026. Wearable robots and rehabilitation platforms operate in direct physical contact with vulnerable users. This makes structural proof, joint loading evidence, and fatigue behavior the first requirement before broader functional claims gain relevance.
- Load cells and torque verification stay core purchases even for manufacturers that later add more advanced gait replay or software validation layers.
Insights into the equipment format segment
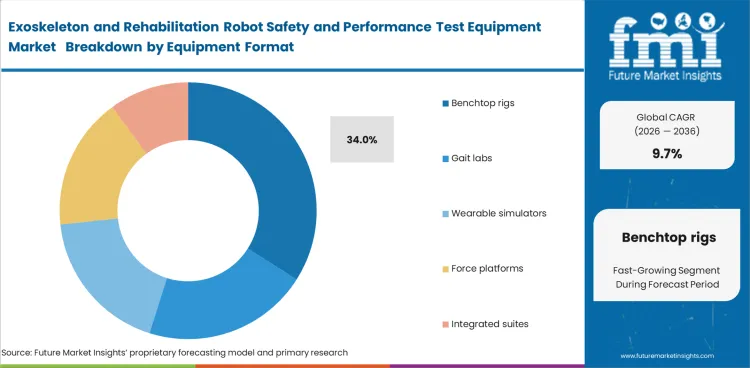
- Benchtop rigs are projected to contribute 34.0% of total market share in 2026. Their lead reflects how many manufacturers still need compact platforms for actuator checks and joint movement validation before shifting to larger integrated environments.
- Benchtop formats fit earlier development budgets and can be used more often during firmware updates, spare-part qualification, and supplier change review.
Insights into the robot type segment
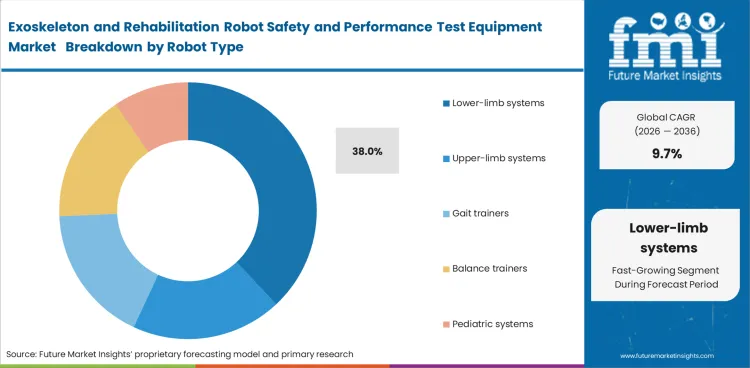
- Lower-limb systems are anticipated to represent 38.0% of the market in 2026. Gait restoration is the most visible and commercially established use case in rehabilitation robotics.
- Test equipment spending follows the device class with the greatest need for body-weight support validation and repeated cycle endurance.
Insights into the end user segment
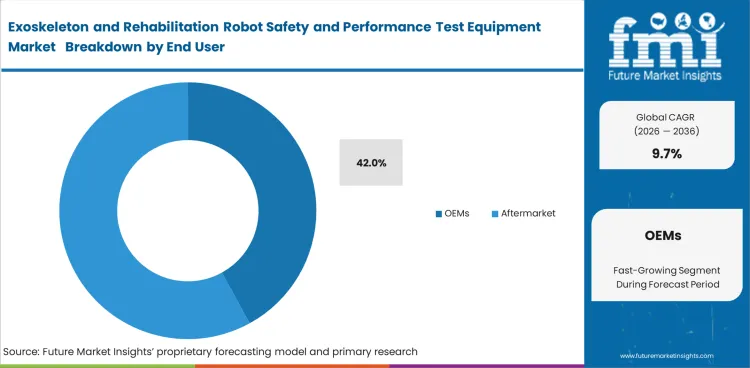
- OEMs are likely to secure 42.0% share in 2026. The leading budget center remains inside manufacturers because safety and performance evidence is needed before release, during international expansion, and after hardware or software modifications.
- Rehabilitation hospitals buy validation tools. They usually prefer training and service-focused systems over full engineering suites.
Insights into the sales channel segment
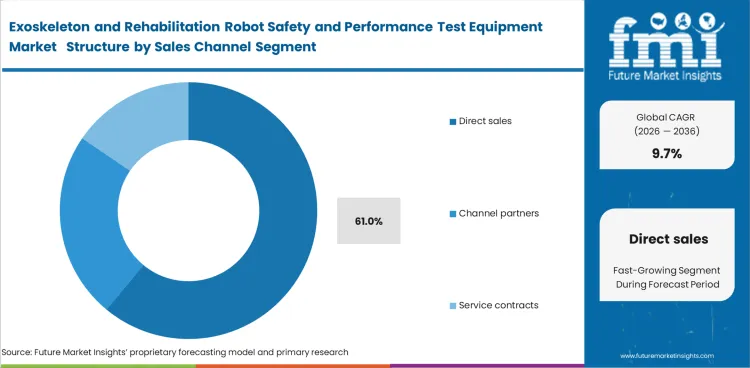
- Direct sales are set to make up 61.0% of the market in 2026. Manufacturers want test sequences adapted to device architecture and documentation expected by regulators or notified bodies. This makes standard catalog selling less effective than engineering-led selling.
- Direct relationships help suppliers add calibration and method development revenue around the hardware base.
Exoskeleton and Rehabilitation Robot Safety and Performance Test Equipment Market Drivers, Restraints, and Opportunities
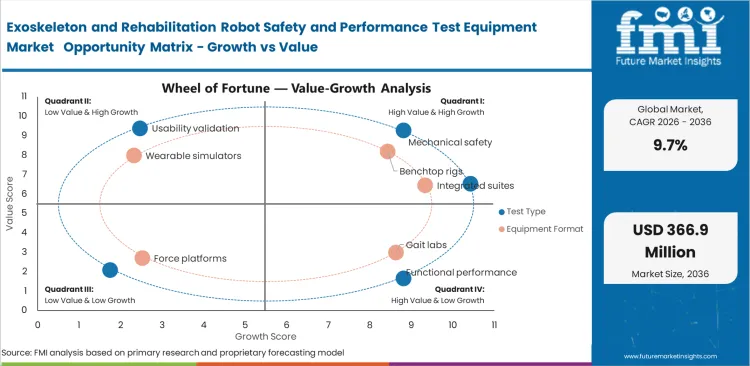
- Rising rehabilitation demand and tighter quality requirements are increasing the need for test systems used to verify safety, repeatability, and device readiness before wider clinical use.
- High system cost and long consultative selling cycles continue to limit faster adoption, especially among smaller developers with staged validation budgets.
- Greater focus on software-linked verification is opening new opportunity for suppliers offering integrated platforms for mechanical checks, motion analysis, and control validation.
The market is advancing through clinical need and documentation pressure across hospitals and specialist centers. Demand continues to build as manufacturers seek stronger proof of safe human-device interaction and more reliable evidence for performance under repeated use. Even with a relatively small installed base, opportunity stays visible in gait systems and revalidation after product updates.
Clinical Rehabilitation Expansion
A larger rehabilitation pipeline is pushing demand for more structured validation methods. As exoskeletons and rehabilitation robots move closer to routine use, manufacturers need tools that can measure load response and motion repeatability in realistic operating conditions. This keeps demand firm for systems used during development, release preparation, and periodic requalification.
Cost and Procurement Constraints
Market expansion is held back by the high cost of specialized validation equipment and the narrow customer base for advanced systems. Smaller developers often begin with compact test setups or outsourced validation work before moving to full integrated platforms. This slows conversion and keeps purchase timing closely tied to funding cycles and product maturity.
Integrated Validation Opportunity
A clear opportunity is forming around systems that combine structural testing and control analysis. Manufacturers increasingly want platforms that can evaluate movement quality and software behavior within one workflow. Suppliers able to support this broader validation need are likely to gain from rising demand for more connected and application-specific test environments.
Analysis of Exoskeleton and Rehabilitation Robot Safety and Performance Test Equipment Market by Key Countries
.webp)
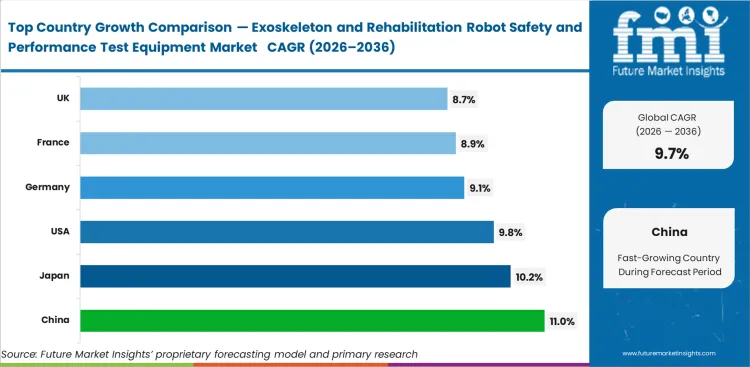
| Country | CAGR |
|---|---|
| China | 11.0% |
| Japan | 10.2% |
| United States | 9.8% |
| Germany | 9.1% |
| France | 8.9% |
| United Kingdom | 8.7% |
Source: Future Market Insights competitive analysis, 2026.
Analysis of Exoskeleton and Rehabilitation Robot Safety and Performance Test Equipment Market CAGR Analysis by Countries
- China leads with 11.0% CAGR, while Japan follows at 10.2% CAGR due to stronger robotics and standards activity.
- The United States is projected to expand at 9.8% CAGR, supported by reimbursement progress and deeper rehabilitation infrastructure.
- Germany is expected to grow at 9.1% CAGR, while France records 8.9% CAGR through the forecast period.
- The United Kingdom posts 8.7% CAGR, against the global market growth rate of 9.7% CAGR through 2036.
The global exoskeleton and rehabilitation robot safety and performance test equipment market is expected to grow at a 9.7% CAGR from 2026 to 2036. The study tracks demand across key countries, with the leading markets outlined below.
Demand Outlook for Exoskeleton and Rehabilitation Robot Safety and Performance Test Equipment Market in China
China is projected to expand at a 11.0% CAGR through 2036. Demand is rising as investment in medical robots grows standards activity moves faster across active medical devices. Local manufacturers are investing earlier in traceable test methods, which supports spending on safety and performance validation systems. A large rehabilitation need base and growing domestic device innovation continue to strengthen the market outlook.
- Faster standards activity is supporting earlier investment in validation tools.
- Domestic medical robotics programs are widening the need for proof-based testing.
- Policy support for high-end devices is improving long-term demand visibility.
Demand Outlook for Exoskeleton and Rehabilitation Robot Safety and Performance Test Equipment Market in Japan
Japan is expected to register a 10.2% CAGR over the forecast period. Demand is supported by an aging population, strong engineering capability, and continued adoption of eldercare assistive robots across care settings. Companies and institutions in Japan place high value on repeatable performance records. This favors test equipment used for documentation, calibration, and safe device behavior. Public support for robotics adoption is also helping create a stronger setting for rehabilitation robot validation demand.
- Aging-related rehabilitation demand is supporting device development activity.
- Funding support for care robotics is improving market familiarity.
- Reliable performance documentation remains important in procurement decisions.
Demand Outlook for Exoskeleton and Rehabilitation Robot Safety and Performance Test Equipment Market in the United States
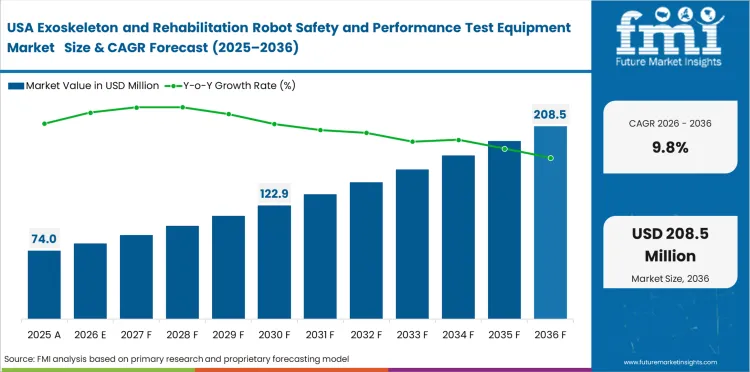
The United States is forecast to grow at a 9.8% CAGR through 2036. The market benefits from a deeper rehabilitation infrastructure, broader reimbursement progress, and stronger access to specialist hospitals working with wearable medical devices. Demand for validation systems is supported by tighter process expectations under current quality requirements. These conditions are encouraging manufacturers to invest in equipment that can document safe operation, repeatability, and performance consistency before wider deployment.
- Reimbursement progress is improving confidence in exoskeleton adoption.
- Specialist hospitals and OEMs support a deeper validation spending base.
- Quality-system pressure is strengthening demand for traceable test workflows.
Demand Outlook for Exoskeleton and Rehabilitation Robot Safety and Performance Test Equipment Market in Germany
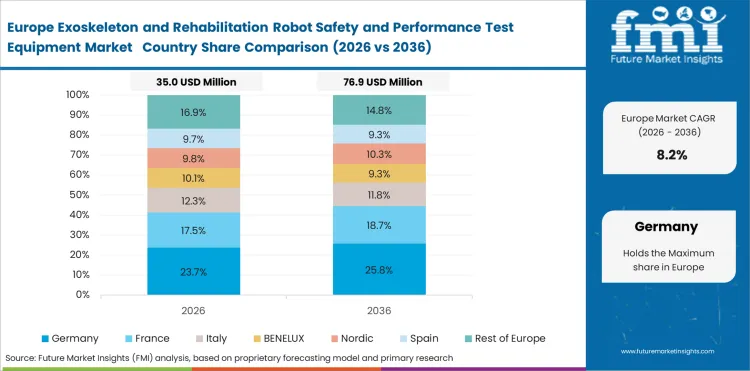
Germany is projected to grow at a 9.1% CAGR through 2036. Demand is supported by a mature rehabilitation structure and a healthcare environment that values documented device performance. The country benefits from more visible payer movement in exoskeleton access. This is encouraging stronger evidence generation before scale-up. Test equipment demand is likely to depend on safety verification, repeatability assessment, and clinical-readiness checks across wearable medical robots.
- Structured rehabilitation pathways support consistent device evaluation activity.
- Payer movement is increasing pressure for stronger validation evidence.
- Engineering-led procurement supports demand for repeatable test methods.
Demand Outlook for Exoskeleton and Rehabilitation Robot Safety and Performance Test Equipment Market in France
France is expected to record 8.9% CAGR during the forecast period. Demand is supported by a sizeable rehabilitation care burden and steady interest from hospitals and research centers evaluating mobility recovery technologies. The market is likely to stay more measured than Germany. Clinical need continues to support investment in validation systems linked to gait recovery and assisted rehabilitation. Suppliers with practical testing workflows and application support are likely to gain better traction in a healthcare setting shaped by broader use of physiotherapy equipment.
- Rehabilitation demand continues to support evaluation of gait-focused platforms.
- Hospital and research use remains more visible than local supplier concentration.
- Practical validation support is likely to matter in supplier selection.
Demand Outlook for Exoskeleton and Rehabilitation Robot Safety and Performance Test Equipment Market in the United Kingdom
The United Kingdom is projected to expand at an 8.7% CAGR through 2036. Demand is supported by organized stroke care pathways and growing interest in technologies that can show safe and measurable rehabilitation outcomes. This increases the importance of validation systems tied to clinician use, training support, and dependable performance records. The market is likely to progress steadily as rehabilitation centers evaluate more evidence-backed programs connected with wearable healthcare devices.
- Stroke pathway reform supports interest in measurable rehabilitation technologies.
- Procurement remains selective and favors well-supported validation systems.
- Clinician-linked training support can improve equipment acceptance.
Competitive Landscape and Strategic Positioning
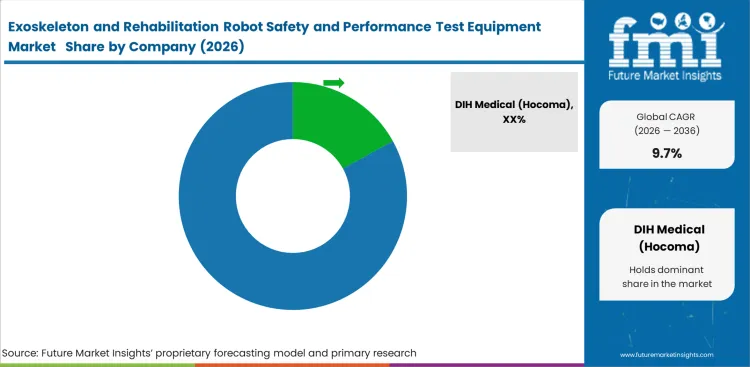
- DIH Medical holds a leading position due to its broad rehabilitation technology exposure and stronger alignment with clinical validation workflows.
- Tyromotion, Ekso Bionics, Lifeward, and CYBERDYNE remain important participants with visible positions in rehabilitation robotics and exoskeleton-centered applications.
- Smaller players are focusing on specialized validation needs such as custom fixtures, pediatric systems, and mixed hardware-software testing environments.
DIH Medical benefits from broad rehabilitation technology exposure and stronger workflow familiarity across therapy environments. Tyromotion holds a solid market position through rehabilitation platform adjacency and international commercial reach. Ekso Bionics and Lifeward remain well placed in exoskeleton-centered applications, while CYBERDYNE brings strength through its medical HAL platform and related validation needs.
Barriers include a relatively small installed base, long consultative selling cycles, strict documentation requirements, and the need for application-specific validation methods. Strategic priorities across the market include improving workflow integration, expanding software-linked verification, and strengthening service support around calibration, training, and requalification.
Key Companies in Exoskeleton and Rehabilitation Robot Safety and Performance Test Equipment Market
The market includes global rehabilitation technology vendors, focused exoskeleton manufacturers, and specialist suppliers that sit close to clinical robotics development.
- DIH Medical holds a leading position due to its broad rehabilitation technology exposure and stronger alignment with clinical validation workflows.
- Tyromotion, Ekso Bionics, Lifeward, and CYBERDYNE remain important participants with visible positions in rehabilitation robotics and exoskeleton-centered applications.
- Smaller players are focusing on specialized validation needs such as custom fixtures, pediatric systems, and mixed hardware-software testing environments.
DIH Medical benefits from broad rehabilitation technology exposure and stronger workflow familiarity across therapy environments. Tyromotion holds a solid market position through rehabilitation platform adjacency and international commercial reach. Ekso Bionics and Lifeward remain well placed in exoskeleton-centered applications, while CYBERDYNE brings strength through its medical HAL platform and related validation needs.
Barriers include a relatively small installed base, long consultative selling cycles, strict documentation requirements, and the need for application-specific validation methods. Strategic priorities across the market include improving workflow integration, expanding software-linked verification, and strengthening service support around calibration, training, and requalification.
Competitive Benchmarking: Exoskeleton and Rehabilitation Robot Safety and Performance Test Equipment Market
| Company | Application Breadth | Validation Support | Portfolio Depth | Geographic Footprint |
|---|---|---|---|---|
| DIH Medical (Hocoma) | High | High | Strong | Global |
| Tyromotion | High | Medium | Strong | Global |
| Ekso Bionics | Medium | High | Strong | Global |
| Lifeward | Medium | High | Moderate | Global |
| CYBERDYNE | Medium | High | Moderate | Multi-region |
| Fourier Intelligence | High | Medium | Moderate | Global |
| Rex Bionics | Low | Medium | Low | Regional |
| MediTouch | Low | Medium | Low | Regional |
| Bioness | Low | Medium | Low | Multi-region |
| Motek | Medium | High | Moderate | Global |
Source: Future Market Insights competitive analysis, 2026.
Key Developments in Exoskeleton and Rehabilitation Robot Safety and Performance Test Equipment Market
- In February 2025, Ekso Bionics named NSM the exclusive U.S. CRT distributor for Ekso Indego Personal, broadening reimbursement-facing channel access.
- In February 2025, Lifeward finalized a reimbursement agreement with BARMER, adding 8.5 million covered lives in Germany for personal exoskeleton access.
Key players in the Exoskeleton and Rehabilitation Robot Safety and Performance Test Equipment Market

Major global players
- DIH Medical (Hocoma)
- Tyromotion
- Ekso Bionics
- Lifeward
- CYBERDYNE
Key emerging players/startups
- Fourier Intelligence
- Rex Bionics
Report Scope and Coverage
| Parameter | Details |
|---|---|
| Quantitative Units | USD 132.5 million to USD 333.0 million, at a CAGR of 9.7% |
| Market Definition | The exoskeleton and rehabilitation robot safety and performance test equipment market covers equipment and software used to test safety, performance, repeatability, and release readiness of powered exoskeletons, gait trainers, and rehabilitation robots. |
| Regions Covered | North America, Europe, East Asia, Asia Pacific |
| Countries Covered | United States, Germany, France, United Kingdom, China, Japan, Australia |
| Key Companies Profiled | DIH Medical (Hocoma), Tyromotion, Ekso Bionics, Lifeward, CYBERDYNE, Fourier Intelligence, Rex Bionics |
| Forecast Period | 2026 to 2036 |
| Approach | Bottom-up market sizing built on rehabilitation burden, standards activity, reimbursement progress, and current supplier positioning across key countries and end-use settings. |
Source: Future Market Insights competitive analysis, 2026.
Exoskeleton and Rehabilitation Robot Safety and Performance Test Equipment Market by Segments
Exoskeleton and Rehabilitation Robot Safety and Performance Test Equipment Market segmented as Test Type
- Mechanical safety
- Functional performance
- Usability validation
- Sensor calibration
- Endurance testing
Exoskeleton and Rehabilitation Robot Safety and Performance Test Equipment Market classified by Equipment Format
- Benchtop rigs
- Gait labs
- Wearable simulators
- Force platforms
- Integrated suites
Exoskeleton and Rehabilitation Robot Safety and Performance Test Equipment Market analyzed by Robot Type
- Lower-limb systems
- Upper-limb systems
- Gait trainers
- Balance trainers
- Pediatric systems
Exoskeleton and Rehabilitation Robot Safety and Performance Test Equipment Market classified by Sales Channel
- Direct sales
- Channel partners
- Service contracts
Exoskeleton and Rehabilitation Robot Safety and Performance Test Equipment Market by Region and Country
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Research Sources and Bibliography
- Ekso Bionics Holdings, Inc. (2025, February 10). NSM named exclusive distributor of Ekso Indego® Personal within the CRT industry.
- Lifeward Ltd. (2025, February 5). Lifeward finalizes personal exoskeleton reimbursement agreement with BARMER, adding 8.5 million covered lives in Germany.
- World Health Organization. (2024, January 2). Assistive technology.
- Centers for Disease Control and Prevention. (2024, October 24). Stroke facts.
The bibliography is provided for reader reference.
This Report Answers
- Current market sizing and forecast for specialized safety and performance test equipment.
- Segment demand by test type, equipment format, robot type, end user, and channel.
- Country analysis across seven priority geographies with modeled CAGR ranges.
- Competitive assessment of active companies and positioning across the workflow.
- Regulatory, standards, and reimbursement factors shaping procurement timing.
- Whitespace in mixed hardware and software verification environments.
- Supply-side implications of broader rehabilitation robotics adoption.
- Decision-useful evidence for vendors, investors, and strategy teams.
Frequently Asked Questions
What is the estimated size of the market in 2026?
The market is estimated at USD 132.5 million in 2026 based on modeled demand for qualification and requalification tools.
What is the projected value by 2036?
The market is projected to reach USD 333.0 million by 2036 as validation workflows deepen across clinical robotics programs.
What CAGR is expected from 2026 to 2036?
The market is expected to expand at a CAGR of 9.7% during the 2026 to 2036 period.
Which test segment leads the market?
Mechanical safety testing leads the market because structural proof and fatigue evidence remain the first buying gate.
Which end user leads demand?
OEMs lead demand since design verification and release testing are still concentrated before wider clinical deployment.
Which countries are rising fastest?
China and Japan are rising fastest due to stronger standards activity, robotics support, and aging-linked rehabilitation demand.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Test Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Test Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Test Type , 2026 to 2036
- Mechanical safety
- Functional performance
- Usability validation
- Sensor calibration
- Endurance testing
- Mechanical safety
- Y to o to Y Growth Trend Analysis By Test Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Test Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Equipment Format
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Equipment Format, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Equipment Format, 2026 to 2036
- Benchtop rigs
- Gait labs
- Wearable simulators
- Force platforms
- Integrated suites
- Benchtop rigs
- Y to o to Y Growth Trend Analysis By Equipment Format, 2021 to 2025
- Absolute $ Opportunity Analysis By Equipment Format, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Robot Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Robot Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Robot Type, 2026 to 2036
- Lower-limb systems
- Upper-limb systems
- Gait trainers
- Balance trainers
- Pediatric systems
- Lower-limb systems
- Y to o to Y Growth Trend Analysis By Robot Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Robot Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- OEMs
- Aftermarket
- OEMs
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Sales Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Sales Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Sales Channel, 2026 to 2036
- Direct sales
- Channel partners
- Service contracts
- Direct sales
- Y to o to Y Growth Trend Analysis By Sales Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Sales Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Test Type
- By Equipment Format
- By Robot Type
- By End User
- By Sales Channel
- Competition Analysis
- Competition Deep Dive
- DIH Medical (Hocoma)
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Tyromotion
- Ekso Bionics
- Lifeward
- CYBERDYNE
- Fourier Intelligence
- Rex Bionics
- DIH Medical (Hocoma)
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Equipment Format, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Robot Type, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Equipment Format, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Robot Type, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Equipment Format, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Robot Type, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Equipment Format, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Robot Type, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Equipment Format, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Robot Type, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Equipment Format, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Robot Type, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Equipment Format, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Robot Type, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Equipment Format, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Robot Type, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Test Type
- Figure 6: Global Market Value Share and BPS Analysis by Equipment Format, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Equipment Format, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Equipment Format
- Figure 9: Global Market Value Share and BPS Analysis by Robot Type, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Robot Type, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Robot Type
- Figure 12: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by End User
- Figure 15: Global Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Sales Channel
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Test Type
- Figure 32: North America Market Value Share and BPS Analysis by Equipment Format, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Equipment Format, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Equipment Format
- Figure 35: North America Market Value Share and BPS Analysis by Robot Type, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Robot Type, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Robot Type
- Figure 38: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by End User
- Figure 41: North America Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Sales Channel
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Test Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Equipment Format, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Equipment Format, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Equipment Format
- Figure 51: Latin America Market Value Share and BPS Analysis by Robot Type, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Robot Type, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Robot Type
- Figure 54: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by End User
- Figure 57: Latin America Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Sales Channel
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Test Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Equipment Format, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Equipment Format, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Equipment Format
- Figure 67: Western Europe Market Value Share and BPS Analysis by Robot Type, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Robot Type, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Robot Type
- Figure 70: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by End User
- Figure 73: Western Europe Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Sales Channel
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Test Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Equipment Format, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Equipment Format, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Equipment Format
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Robot Type, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Robot Type, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Robot Type
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by End User
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Sales Channel
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Test Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Equipment Format, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Equipment Format, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Equipment Format
- Figure 99: East Asia Market Value Share and BPS Analysis by Robot Type, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Robot Type, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Robot Type
- Figure 102: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by End User
- Figure 105: East Asia Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Sales Channel
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Test Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Equipment Format, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Equipment Format, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Equipment Format
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Robot Type, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Robot Type, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Robot Type
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Sales Channel
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Test Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Equipment Format, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Equipment Format, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Equipment Format
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Robot Type, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Robot Type, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Robot Type
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Sales Channel
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

