High-Throughput Liquid Handling Robot Performance Test Equipment Market
The High-Throughput Liquid Handling Robot Performance Test Equipment Market Is Segmented By Product Type (Gravimetric Systems, Photometric Systems, Hybrid Stations, Software Kits), Measurement Mode (Integrated Kits, Bench Stations, Service Tools), Channel Format (96-Channel, 384-Channel, 1536-Channel, Single-Channel), End Use (Pharma Biopharma, CRO Labs, Clinical Labs, Academic Labs), Application (Assay Setup, NGS Prep, Compound Screening, Sample Normalization), And Region. Forecast For 2026 To 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
High-Throughput Liquid Handling Robot Performance Test Equipment Market Size, Market Forecast and Outlook By FMI
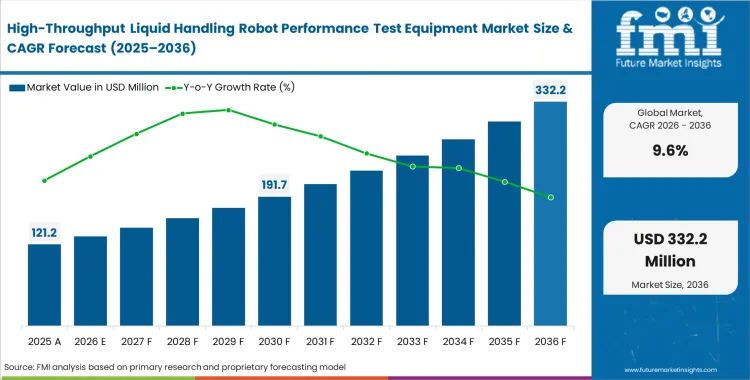
The high-throughput liquid handling robot performance test equipment market crossed a valuation of USD 110.6 million in 2025, with a steady increase in pace, the industry seems poised to surpass USD 121.2 million in 2026 at a CAGR of 9.6% during the forecast period. This pace set for revenue inclination suggests a rise to USD 303.1 million through 2036 as regulatory bodies increasingly reject assay data lacking integrated automated pipetting accuracy testing logs.
Summary of High-Throughput Liquid Handling Robot Performance Test Equipment Market
- The market is forecast to reach USD 303.1 million by 2036.
- The market is expected to grow at a CAGR of 9.6% from 2026 to 2036.
- The market was estimated at USD 110.6 million in 2025.
- The forecast period represents an incremental opportunity of USD 181.9 million.
- The market is projected to reach USD 121.2 million in 2026, indicating strong growth momentum.
- Growth is driven by increasing regulatory requirements mandating automated pipetting accuracy testing and audit-ready data logs.
- Rising adoption of high-throughput liquid handling systems in genomics, drug discovery, and clinical testing is accelerating demand for performance testing equipment.
- Integration of real-time testing tools within robotic systems is reducing downtime and improving operational efficiency in laboratories.
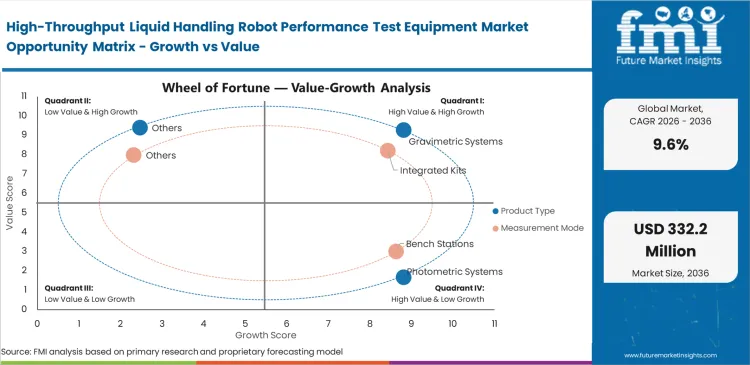
- Gravimetric systems dominate by product type with a 39.0% share due to reliability and audit acceptance.
- Integrated testing solutions lead the measurement mode segment with a 41.0% share, driven by seamless software compatibility.
- 96-channel systems dominate the format segment with a 36.0% share, aligned with standard laboratory workflows.
- Pharmaceutical and biopharmaceutical companies lead the end-use segment with a 34.0% share due to strict regulatory requirements.
- India, China, and South Korea are the fastest-growing markets, supported by expanding laboratory infrastructure and automation adoption.
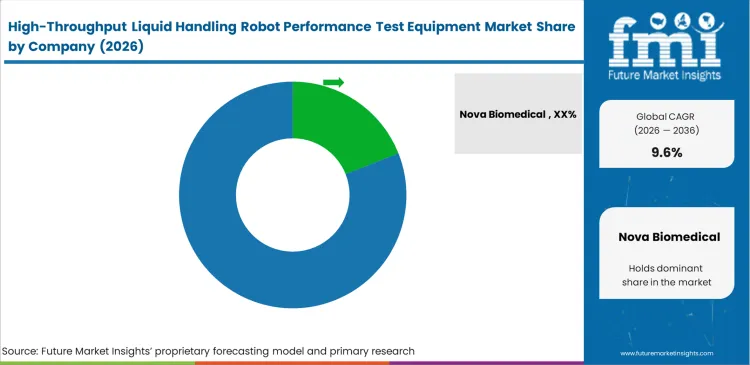
- Key companies in the market include Nova Biomedical, Hamilton Company, Tecan Group, Beckman Coulter Life Sciences, and Analytik Jena.
A modern testing organization operates under a strict zero-tolerance mandate for fluid transfer errors during high-value screening runs. Relying on manual equipment checks drains expensive chemical reserves and creates documentation gaps that trigger immediate regulatory red flags. Facilities delaying the transition to automated testing platforms eventually face complete batch losses when retrospective manual checks fail. By embedding precision metrology robots and performance sensors directly into their automated decks, these organizations purchase an active insurance policy against compliance failures, shifting the baseline of lab automation from simple movement speed to absolute dispensing accuracy.
Once a clinical laboratory integrates its daily validation data directly into central software networks, the financial burden of scaling quality control drops significantly. The facility transforms audit management from a manual, physical bottleneck into an invisible background software process. A clear signal of this operational shift occurs when an organization accelerates its procurement of molecular diagnostics software kits just before executing a complete overhaul of its legacy testing hardware.
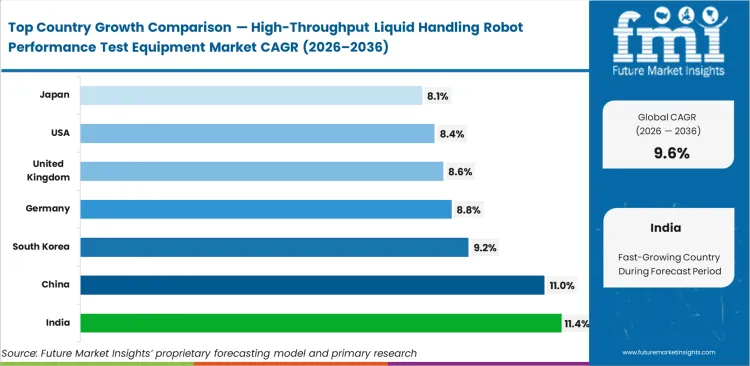
India anticipated to lead regional industry development at 11.4%, with rapid CRO infrastructure scaling demanding strict compliance with Western audit standards, while China likely to tracks at 11.0% on the back of state-sponsored genomic sequencing initiatives. South Korea predicted to follows at 9.2%, Germany at 8.8%, the United Kingdom at 8.6%, the United States at 8.4%, and Japan at 8.1%. Differences in adoption speed across these geographies come down to how aggressively local health ministry enforces NIST-traceable audit trails for clinical diagnostics.
Segmental Analysis
High-Throughput Liquid Handling Robot Performance Test Equipment Market Analysis by Product Type
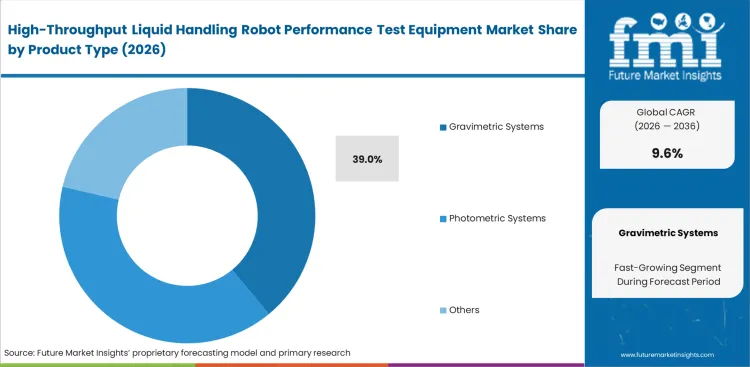
Auditors accept only specific physical proofs when validating million-dollar clinical runs. Gravimetric systems are expected to dominate this space with a 39.0% share in 2026, providing NIST-traceable absolute mass verification that regulatory bodies demand. Quality assurance managers at major clinical labs rely on these balances to establish baseline truth for their entire robotic fleet. According to FMI's estimates, this preference for gravimetric liquid handling verification systems creates a workflow bottleneck. While physical mass testing serves as the undisputed gold standard for audits, it operates slowly and reacts poorly to environmental vibrations on robotic decks. Laboratory technicians actually run photometric liquid handling validation equipment for daily checks, keeping gravimetric units strictly for scheduled compliance events. Facilities attempting to run high-speed high-throughput screening with only gravimetric verification face severe instrument downtime.
- Initial Qualification: Setting up new liquid handlers requires gravimetric tools to satisfy factory acceptance testing. Automation engineers use precise mass measurements to build primary operating profiles and establish what standards apply to liquid handling robots.
- Routine Verification: Daily operational checks shift to photometric systems to save time. Laboratory technicians process rapid dye-based tests to confirm instruments have not drifted since morning startups.
- Long-Term Expansion: Hybrid stations emerge as logical upgrade paths for expanding facilities. Operations directors invest in combined units to execute both rapid daily checks and deep regulatory audits from single deck locations.
High-Throughput Liquid Handling Robot Performance Test Equipment Market Analysis by Measurement Mode
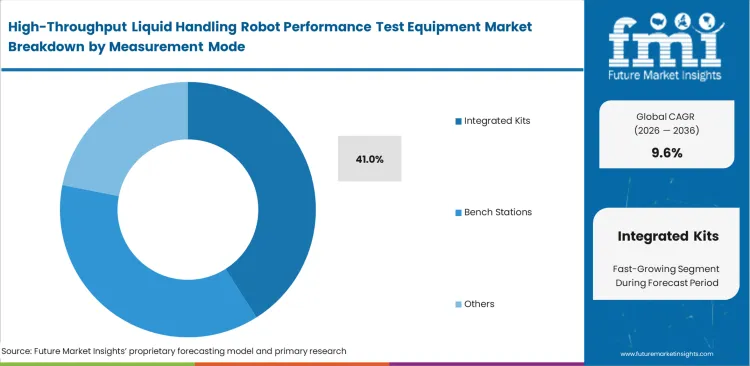
Seamless data handoff determines which tools actually survive in modern clinical settings. Informatics directors mandate built-in systems because they automatically push verification logs directly into laboratory information management systems without human intervention, pushing integrated kits anticipated to capture 41.0% of the market in 2026. FMI's analysis indicates this removes primary sources of transcription errors during compliance reporting. Buyers often view liquid handler QC kits as hardware purchases, missing that they act entirely as data governance tools designed to satisfy external auditors. Laboratory managers choosing standalone bench stations save capital upfront but end up paying hidden taxes in the form of manual data entry labor. Without integration, technicians spend hours formatting microplate instrumentation and systems data to match clinical trial submission requirements.
- Capital Acquisition: Integrated kits are approved based on their ability to reduce manual documentation hours. Return on investment is usually measured through labor savings rather than hardware longevity when evaluating the best liquid handling verification system.
- Hidden Friction: Software compatibility issues frequently stall deployment even after physical installation. IT departments struggle to map verification output data to legacy database structures.
- Lifecycle Reality: Total ownership costs drop significantly once integration is complete. Quality control managers realize audit preparation time shrinks from weeks to hours when comparing gravimetric and photometric robot verification methods that flow automatically.
High-Throughput Liquid Handling Robot Performance Test Equipment Market Analysis by Channel Format
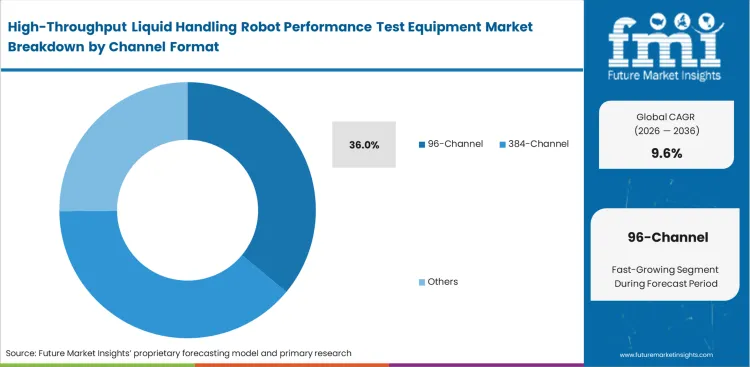
Legacy assay protocols dictate hardware geometry long after technology evolves. In 2026, the 96-channel segment is poised to account for 36.0% of market share, as this demand is sustained entirely by massive backlogs of validated standard operating procedures built around it. Clinical directors refuse to rewrite FDA-cleared workflows just to accommodate denser plate layouts. Based on FMI's assessment, loyalty to 96 channel liquid handling verification has nothing to do with technical superiority. Automation engineers know 384 well dispensing validation offers vastly superior throughput for modern laboratory benchtop automation, but pushing established clinical tests onto new formats requires complete, expensive requalification. Laboratories dragging their feet on upgrading formats eventually run out of physical floor space as they try scaling throughput with outdated spatial efficiency.
- Primary Prevention: 96-channel verification tools prevent blocked tips from ruining established diagnostic runs. Technicians rely on these checks to catch simple mechanical failures before wasting patient samples.
- Residual Vulnerability: Evaporation remains a critical risk even with perfect volumetric dispensing. Assay developers must still account for environmental loss during long incubation periods.
- Optimization Requirement: Capturing full benefits of denser formats requires completely re-optimizing reagent concentrations. Scientists spend months tweaking formulations to behave properly in microscopic 1536-well volumes.
High-Throughput Liquid Handling Robot Performance Test Equipment Market Analysis by End Use
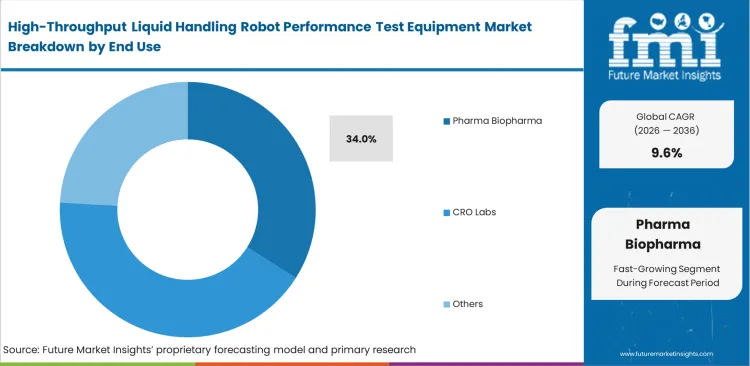
Securing international contracts requires proving absolute process control to skeptical sponsors. Outsourcing directors at massive organizations use pharmaceutical liquid handling QA tools to evaluate not just their own equipment, but the reliability of potential contract manufacturing partners. The pharma biopharma category is likely to account for 34.0% share in 2026, per FMI's view, buyers deploy biopharma automation verification equipment aggressively to qualify new suppliers of plastic consumables. Slight variations in third-party pipette tips can alter fluid dynamics enough to ruin high-content screening runs. Operations managers neglecting CRO liquid handling validation systems often discover assay failures stem from cheap plastics rather than robotic malfunction, facing millions in wasted compound libraries as a consequence.
- Tier 1 Producers: Major pharmaceutical companies possess capital to standardize verification platforms globally. They force entire supply chains to adopt identical testing metrics to ensure data harmony.
- Supply Bottlenecks: Global shortages of specific photometric dyes periodically disrupt validation schedules. Secondary suppliers are often needed to keep robotic fleets operational.
- Future Landscape: Smaller academic labs eventually inherit older, refurbished clinical diagnostics liquid handling verification systems as major players upgrade. Researchers at these institutions gain access to enterprise-grade compliance tools at fractions of original costs.
High-Throughput Liquid Handling Robot Performance Test Equipment Market Analysis by Application
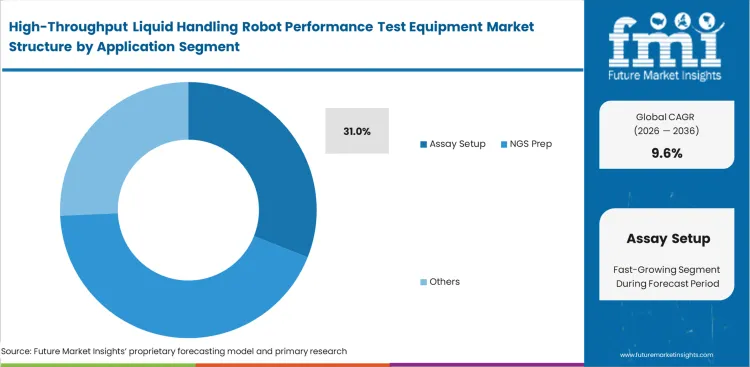
Viscosity variations routinely destroy standard calibration profiles without active monitoring. Formulation scientists deal with complex biological reagents that flow differently depending on temperature, age, and concentration. As per FMI's projection, standard factory calibration curves fail completely when handling non-standard fluids, explaining why the assay setup segment is forecast to command 31.0% share in 2026. Using liquid class optimization tools does not simply verify volume; it allows technicians to build custom dispensing profiles for specific challenging reagents. Laboratories relying on generic settings for sensitive genomics applications or NGS library prep liquid handler validation experience massive variability in final sequence data. Delaying implementation of assay setup liquid handling verification results in unexplainable batch-to-batch inconsistencies that plague research timelines.
- High Performance: Custom liquid class calibration exceeds standard factory specifications when tuned correctly. Automation scientists achieve exceptional precision even with highly viscous glycerol mixtures.
- Edge Degradation: Performance drops sharply when dealing with highly volatile solvents that evaporate during transfers. Technicians struggle to get consistent mass readings before liquid vanishes from balances.
- Standardized Acceptance: Regulatory bodies demand proof that equipment performs adequately with actual assay fluids rather than just water. Quality directors must maintain separate validation logs for every unique compound handled by systems during compound screening dispensing verification.
High-Throughput Liquid Handling Robot Performance Test Equipment Market Drivers, Restraints, and Opportunities
Laboratories running automated sequencing workflows place a premium on first-pass accuracy because one blocked channel during library preparation can wipe out costly reagents and compromise patient samples that cannot be replaced. Periodic manual inspection leaves too much room for undetected mechanical drift, especially when automated systems are running extended workflows with limited operator intervention. Errors in volume transfer need to be identified at the point of occurrence, not after sequencing data has already been generated and the batch is lost. Continuous automated testing addresses that exposure by turning equipment performance into a traceable control point that also strengthens audit readiness under regulated diagnostic settings. The investment case is straightforward. Preventing even a single failed assay batch can justify the cost of the testing system in many molecular diagnostics laboratories.
Software integration remains the larger obstacle once laboratories move from purchase decisions to actual deployment. Verification hardware alone does not solve the problem when new systems must interface with legacy robotic decks, schedulers, and closed automation software. Many clinical laboratories do not have in-house development teams capable of resolving these interoperability issues on their own. Standard software patches often miss key process details, which can weaken data continuity and create gaps in the audit trail. Integration work frequently falls back on automation engineers, who may spend weeks writing custom code to establish stable communication between sensors and core workflow software. That implementation burden continues to slow upgrade cycles, even where the need for tighter process control is already well understood.
- Third-Party Calibration Contracts: Independent service companies can secure lucrative maintenance deals in mixed-brand laboratories by developing universal testing hardware that bypasses restricted manufacturer software.
- Low-Volume Specialized Sensors: Standard verification tools struggle to measure microscopic fluid drops accurately, creating a clear opening to engineer highly sensitive sensors specifically for advanced high-end cellomics research.
- Cloud-Based Compliance Dashboards: Global testing networks find it difficult to consolidate validation data across multiple geographic locations, making secure online performance tracking portals highly valuable for simplifying the audit process.
Regional Analysis
Global adoption of performance test equipment depends heavily on local regulatory enforcement and the expansion rate of contract testing facilities. Regions enforcing strict volumetric audit trails show the fastest transition from manual checks to integrated software validation.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 11.4% |
| China | 11.0% |
| South Korea | 9.2% |
| Germany | 8.8% |
| United Kingdom | 8.6% |
| United States | 8.4% |
| Japan | 8.1% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Asia-Pacific High-Throughput Liquid Handling Robot Performance Test Equipment Market Analysis
Western pharmaceutical companies increasingly shift trial volumes to Asian contract research organizations, demanding identical audit trails regardless of geography. State-sponsored precision medicine programs across the region also require massive biobanking infrastructure that cannot function without strict volumetric control.
- India: Local contract research organizations must prove their data integrity matches Western standards to win lucrative outsourcing deals. India is anticipated to witness demand grow at a CAGR of 11.4% over the forecast period, as quality control directors use automated verification systems as a competitive advantage during sponsor audits. Securing these certifications directly opens access to global clinical trial pipelines.
- China: Large sequencing centers in China depend on high-speed photometric kits to verify extensive liquid handling fleets without interrupting routine throughput. Daily control checks in these facilities place a higher premium on speed and workflow continuity than on absolute gravimetric precision. That operating model is expected to support 11.0% CAGR in sales through 2036, while pushing suppliers to design bulk verification solutions suited to mega-lab environments.
- South Korea: Diagnostic manufacturing in South Korea places verification directly within production workflows to maintain consistent reagent volumes across large commercial test runs. Quality uniformity carries added weight because exported medical products face close scrutiny across regional markets. Sales in South Korea are projected to rise at a 9.2% CAGR through 2036, supported by this sustained focus on process control and manufacturing consistency.
- Japan: Laboratory automation is advancing steadily in Japan as aging technical workforces reduce the availability of specialized staff for manual verification tasks. Automated pipetting checks are increasingly treated as part of routine compliance infrastructure rather than an optional upgrade. The market in Japan is forecast to grow at 8.1% CAGR through 2036, with adoption supported by the need to maintain reliable operations with leaner technical teams.
FMI analyses, the facilities across the Asia-Pacific region initially resisted the capital expense of automated verification, but sponsor mandates and labor shortages have turned these systems into operational necessities. Developing clinical networks here prioritize affordable laboratory benchtop automation validation tools before scaling to fully integrated platforms.
Europe High-Throughput Liquid Handling Robot Performance Test Equipment Market Analysis
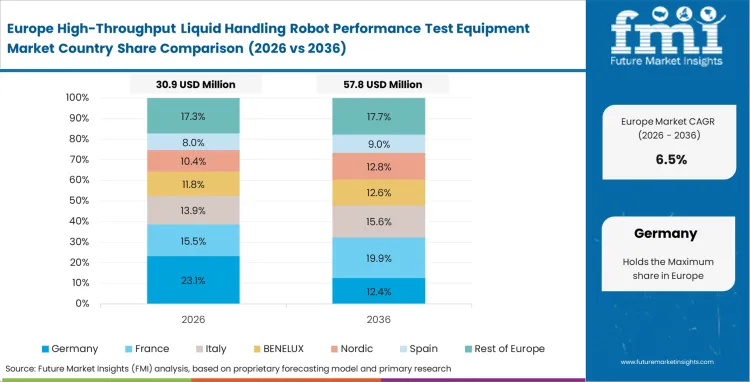
Strict diagnostic regulations mandate comprehensive traceability for every patient sample processed within the union. European laboratories face intense scrutiny regarding instrument calibration schedules and data provenance, with compliance officers frequently rejecting data from systems lacking timestamped verification logs.
- Germany: The sector for verification tools in Germany is anticipated to grow at a CAGR of 8.8% during the study period, driven by stringent local accreditation standards that force clinical laboratories to maintain continuous physical proof of instrument performance. Quality assurance teams prefer gravimetric systems to ensure absolute compliance with national metrology laws. Maintaining these strict logs shields facilities from aggressive retrospective audits.
- United Kingdom: Centralized health service purchasing structures push facilities to standardize validation hardware across entire hospital networks. Industry adoption in the United Kingdom is expected to advance at a CAGR of 8.6% through 2036 as software compatibility requirements support centralized compliance monitoring. Centralized oversight is accelerating the retirement of older, disconnected standalone testing stations.
As per FMI’s assessments, European facilities navigating these tight regulatory environments lean heavily on automated systems to reduce their daily compliance burden. The regional focus remains on securing bulletproof precision metrology robots and software audit trails rather than just raw processing speed.
North America High-Throughput Liquid Handling Robot Performance Test Equipment Market Analysis
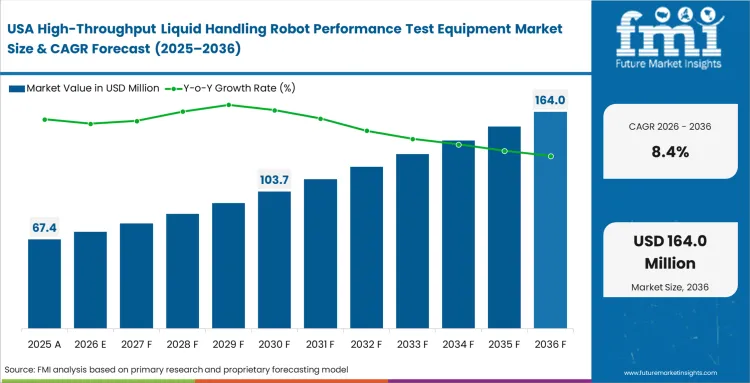
High regulatory scrutiny dictates exactly how validation hardware enters major testing centers. Purchasing committees base their decisions strictly on audit-readiness rather than minor hardware features or brand legacy.
- United States: Buyers in the United States place heavy weight on documentation quality when selecting liquid handling verification systems, especially where FDA compliance depends on clean, inspection-ready records with minimal manual intervention. API connectivity has become a central selection criterion because laboratories need verification data to flow directly into broader quality and workflow software without creating audit gaps. Demand in the United States is expected to rise at a CAGR of 8.4% through 2036. Suppliers that cannot provide reliable open-data integration often face longer qualification cycles and weaker positioning in large institutional accounts.
FMI notes that buying patterns across smaller regional diagnostic networks are beginning to reflect those seen in larger enterprise systems. Laboratories across North America now place greater emphasis on verifiable data governance, often linking equipment selection to broader large molecule bioanalytical testing services compliance objectives.
Competitive Aligners for Market Players
Software compatibility shapes equipment selection. Laboratories already run extensive fleets of robotic decks from companies such as Hamilton Company and Tecan Group. Test equipment vendors need to ensure their systems transmit data directly into the software used to manage these installed platforms. Equipment that cannot communicate with established integrated chemistry systems rarely advances beyond the initial technical screening. Operations teams avoid verification tools that require staff to work across separate interfaces or maintain isolated data files.
Early market entrants maintain a tight grip on customer loyalty because they already own the necessary software connections. Brands such as Artel have spent years testing the code that links their hardware to major liquid handlers. New competitors face a steep learning curve to replicate these digital pathways from scratch. FMI observes that writing secure code to transfer data without triggering strict hospital IT alarms presents a serious hurdle for startup manufacturers. This established software base acts as a protective wall for suppliers providing medical automation validation tools.
Major pharmaceutical buyers actively push back against vendor lock-in by demanding open access to their validation data. Automation engineers at these large facilities frequently write their own code to pull raw test results directly from the hardware. They bypass the manufacturer's software entirely to maintain control over their internal records. Equipment makers who try to restrict data access face immediate resistance from enterprise clients, especially when those clients are validating high-value enzyme activity assays. To secure long-term contracts, hardware manufacturers have to support open data sharing instead of forcing laboratories to stay inside a closed digital system.
Key Players in High-Throughput Liquid Handling Robot Performance Test Equipment Market
- Nova Biomedical (including former Advanced Instruments / Artel)
- Hamilton Company
- Tecan Group
- Beckman Coulter Life Sciences
- Analytik Jena
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 121.2 million to USD 303.1 million, at a CAGR of 9.6% |
| Market Definition | High-Throughput Liquid Handling Robot Performance Test Equipment comprises hardware systems, photometric kits, and gravimetric stations that actively verify volumetric accuracy for automated dispensing arms to detect blockages and calibration drift. |
| Segmentation | Product Type, Measurement Mode, Channel Format, End Use, Application, Region |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | United States, China, India, Germany, United Kingdom, Japan, South Korea |
| Key Companies Profiled | Nova Biomedical, Hamilton Company, Tecan Group, Beckman Coulter Life Sciences, Analytik Jena |
| Forecast Period | 2026 to 2036 |
| Approach | Procurement volumes of photometric dye kits tied to annual service contracts |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Segments
Product Type:
- Gravimetric systems
- Photometric systems
- Hybrid stations
- Software kits
Measurement Mode:
- Integrated kits
- Bench stations
- Service tools
Channel Format:
- 96-channel
- 384-channel
- 1536-channel
- Single-channel
End Use:
- Pharma biopharma
- CRO labs
- Clinical labs
- Academic labs
Application:
- Assay setup
- NGS prep
- Compound screening
- Sample normalization
Regions:
- Asia Pacific
- India
- China
- Japan
- South Korea
- Indonesia
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- Italy
- France
- United Kingdom
- Spain
- Benelux
- Nordics
- Central & Eastern Europe
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Turkey
- Rest of Middle East & Africa
Bibliography
- National Institute of Standards and Technology. (2024, November). Advancing cell-free manufacturing: Challenges in scale-up and automation workshop report (NIST SP 1500-26). USA Department of Commerce.
- Lo, C. S. Y., Taneja, N., & Ray Chaudhuri, A. (2024). Enhancing quantitative imaging to study DNA damage response: A guide to automated liquid handling and imaging. DNA Repair, 144, 103769.
- Quijano Velasco, P., Low, K. Y. A., Leong, C. J., Ng, W. T., Qiu, S., Jhunjhunwala, S., Li, B., Qian, A., Hippalgaonkar, K., & Cheng, J. J. W. (2024). Optimization of liquid handling parameters for viscous liquid transfers with pipetting robots, a “sticky situation”. Digital Discovery, 3(5), 1011–1020.
- Schuster, J., Kamuju, V., Zhou, J., & Mathaes, R. (2024). Piston-driven automated liquid handlers. SLAS Technology, 29(3), 100128.
- Moukarzel, G., Wang, Y., Xin, W., Hofmann, C., Joshi, A., Loughney, J. W., & Bowman, A. (2024). Automation of biochemical assays using an open-sourced, inexpensive robotic liquid handler. SLAS Technology, 29(6), 100205.
- Wu, G., Wang, R., & Coley, C. W. (2025). Optimization of robotic liquid handling as a capacitated vehicle routing problem. Digital Discovery, 4(9), 2593-2601.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Exact regulatory requirements driving photometric kit purchases across top-tier clinical labs.
- Explain the market for liquid handling robot test equipment dynamics impacting legacy formats.
- Hidden software integration costs preventing laboratories from deploying standalone bench stations.
- True audit failure rates causing massive batch invalidations in next-generation sequencing runs.
- Qualification benchmarks used by pharmaceutical giants to assess regional contract research organizations.
- Viscosity limitations that render standard factory calibration curves useless during complex assay setups.
- Commercial stakes of maintaining open API architectures for established liquid handling hardware vendors.
- Capital expenditure shifts favoring integrated data governance over pure mechanical throughput.
Frequently Asked Questions
What is the high-throughput liquid handling robot performance test equipment market?
This sector encompasses hardware systems, photometric kits, and gravimetric stations that actively verify volumetric accuracy for automated dispensing arms to prevent assay failures and satisfy strict regulatory compliance audits.
Why do automated liquid handlers need verification and calibration?
Automated liquid handlers require continuous verification because undetected mechanical drift or blocked channels during sample library preparation destroy thousands of dollars in proprietary reagents and invalidate days of patient data.
What is the difference between gravimetric and photometric liquid handling verification?
Gravimetric testing provides slow but absolute NIST-traceable mass verification required for factory acceptance, while photometric testing uses rapid dye-based methods to confirm daily operational accuracy without causing severe instrument downtime.
Which standards govern automated liquid handling performance testing?
Automated liquid handling performance testing is heavily governed by strict frameworks like ISO 23783, ISO 8655, ASTM E1154, and CLSI guidelines that dictate exact baseline technical specifications for clinical diagnostic hardware.
Which companies lead the liquid handling verification equipment market?
The landscape is primarily led by established incumbents holding deep libraries of pre-written integration scripts, including prominent players like Nova Biomedical, Hamilton Company, Tecan Group, and Beckman Coulter Life Sciences.
What end-use sectors buy liquid handling robot validation systems?
Pharmaceutical and biopharmaceutical entities act as the primary buyers, aggressively deploying these systems to ensure absolute process control and to rigorously qualify the reliability of new contract manufacturing partners.
Which countries are growing fastest for liquid handling QA tools?
India expands at a rapid pace of 11.4% as local contract research organizations upgrade data governance to win Western outsourcing deals, followed closely by China at 11.0% due to state-sponsored precision medicine initiatives.
How does liquid handling verification affect assay reproducibility?
Continuous verification directly ensures assay reproducibility by allowing technicians to build custom dispensing profiles for specific challenging reagents, completely eliminating the batch-to-batch inconsistencies caused by variable fluid viscosities.
How often should automated liquid handlers be requalified?
While gravimetric requalification occurs during scheduled compliance events or initial factory acceptance testing, modern laboratories run rapid photometric checks daily to confirm instruments have not drifted since morning startups.
What is included and excluded in this market?
This sector includes dual-dye photometric testing plates and deck-integrated microbalances with proprietary software, but strictly excludes general-purpose analytical balances, single-channel handheld pipette tools, and the biological reagents themselves.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Gravimetric Systems
- Photometric Systems
- Others
- Gravimetric Systems
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Measurement Mode
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Measurement Mode, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Measurement Mode, 2026 to 2036
- Integrated Kits
- Bench Stations
- Others
- Integrated Kits
- Y to o to Y Growth Trend Analysis By Measurement Mode, 2021 to 2025
- Absolute $ Opportunity Analysis By Measurement Mode, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Channel Format
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Channel Format, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Channel Format, 2026 to 2036
- 96-Channel
- 384-Channel
- Others
- 96-Channel
- Y to o to Y Growth Trend Analysis By Channel Format, 2021 to 2025
- Absolute $ Opportunity Analysis By Channel Format, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Pharma Biopharma
- CRO Labs
- Others
- Pharma Biopharma
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Assay Setup
- NGS Prep
- Others
- Assay Setup
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Measurement Mode
- By Channel Format
- By End Use
- By Application
- Competition Analysis
- Competition Deep Dive
- Nova Biomedical
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Hamilton Company
- Tecan Group
- Beckman Coulter Life Sciences
- Analytik Jena
- Nova Biomedical
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Measurement Mode, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Channel Format, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Measurement Mode, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Channel Format, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Measurement Mode, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Channel Format, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Measurement Mode, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Channel Format, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Measurement Mode, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Channel Format, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Measurement Mode, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Channel Format, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Measurement Mode, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Channel Format, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Measurement Mode, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Channel Format, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Measurement Mode, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Measurement Mode, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Measurement Mode
- Figure 9: Global Market Value Share and BPS Analysis by Channel Format, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Channel Format, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Channel Format
- Figure 12: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by End Use
- Figure 15: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Application
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Product Type
- Figure 32: North America Market Value Share and BPS Analysis by Measurement Mode, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Measurement Mode, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Measurement Mode
- Figure 35: North America Market Value Share and BPS Analysis by Channel Format, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Channel Format, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Channel Format
- Figure 38: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by End Use
- Figure 41: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Application
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Product Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Measurement Mode, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Measurement Mode, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Measurement Mode
- Figure 51: Latin America Market Value Share and BPS Analysis by Channel Format, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Channel Format, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Channel Format
- Figure 54: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by End Use
- Figure 57: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Application
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Product Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Measurement Mode, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Measurement Mode, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Measurement Mode
- Figure 67: Western Europe Market Value Share and BPS Analysis by Channel Format, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Channel Format, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Channel Format
- Figure 70: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by End Use
- Figure 73: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Application
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Measurement Mode, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Measurement Mode, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Measurement Mode
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Channel Format, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Channel Format, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Channel Format
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Application
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Product Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Measurement Mode, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Measurement Mode, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Measurement Mode
- Figure 99: East Asia Market Value Share and BPS Analysis by Channel Format, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Channel Format, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Channel Format
- Figure 102: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by End Use
- Figure 105: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Application
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Measurement Mode, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Measurement Mode, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Measurement Mode
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Channel Format, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Channel Format, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Channel Format
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Measurement Mode, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Measurement Mode, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Measurement Mode
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Channel Format, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Channel Format, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Channel Format
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

