The robotic microsurgery systems market was valued at USD 48.0 million in 2025. The sector is set to hit USD 58.4 million in 2026 at a CAGR of 21.6% during the forecast period. Continued capital infusion is expected to drive the market’s value to USD 412.8 million by 2036, as surgical centres reach the performance limits of human tremor‑control and increasingly transition to untethered, field‑directed micromanipulation for ultra‑fine anatomical tasks.
Surgical directors analyzing the growth outlook for swarm robotics microsurgery systems must recognize an important change: buyers are no longer deciding whether to adopt motion-scaling systems, but rather how to navigate anatomies where tethered instruments cannot physically fit. This transition forces a switch from rigid kinematic linkages to programmable magnetic fields that allow untethered entities to perform collective tasks like thrombus treatment microrobots or delicate tissue reconstruction. The commercial stakes for delaying this capability involve surrendering high-margin neurological and ophthalmic reconstruction caseloads to specialized centers that have already qualified these advanced digital microsurgery workflows. According to FMI's estimates, the initial capital friction is high, but the resulting procedural throughput offsets the acquisition cost for early adopters.
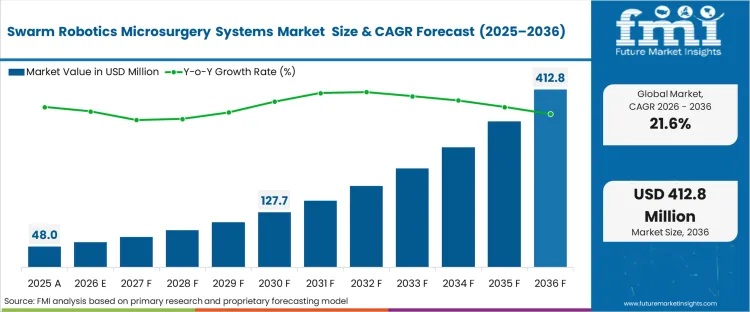
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 58.4 million |
| Industry Value (2036) | USD 412.8 million |
| CAGR (2026-2036) | 21.6% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Scaling this capability requires passing a critical structural gate: the integration of real-time imaging with collective microrobot control in surgery. Regulatory bodies and clinical governance boards trigger this transition when they approve software frameworks that filter human latency out of the navigation loop. Once this threshold is crossed, the cost of adding subsequent surgical robot procedures drops dramatically, allowing parallel utilization across multiple surgical theaters using advanced supermicrosurgery robot systems.
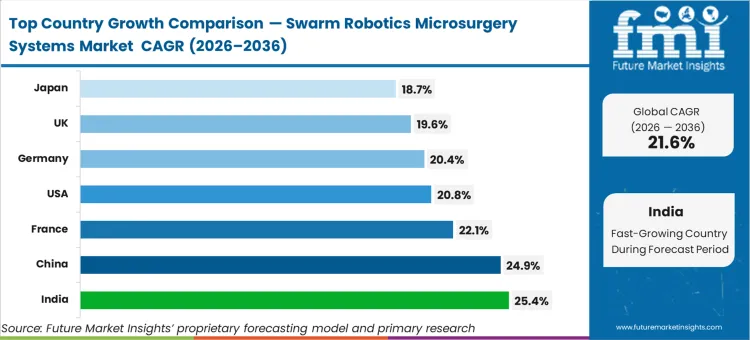
India is expected to advance at 25.4%, driven by rapid scaling of tertiary infrastructure unburdened by legacy robotic investments. China tracks at 24.9% as domestic translation institutes aggressively pilot frontier surgical platforms. France is poised to expand at 22.1%, anchored by concentrated European microrobot innovation hubs. The United States will record a 20.8% trajectory, constrained slightly by extensive qualification cycles within mature hospital networks. Germany follows at 20.4%, balancing strong baseline clinical engineering with cautious reimbursement frameworks. The United Kingdom is likely to grow at 19.6%, followed by Japan posting 18.7%. The divergence across this range reflects a reality regarding which countries will lead the swarm robotics microsurgery market: markets with smaller legacy installed bases are leapfrogging directly to swarm-level deployments, while mature healthcare economies manage slower, phased replacements of existing tethered capital.
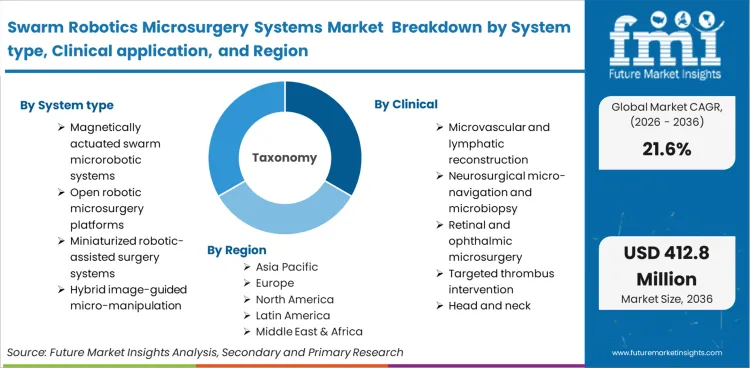
Swarm robotics microsurgery systems encompass untethered, micro- or nano-scale robotic entities that operate cooperatively inside the human body under external guidance, typically magnetic, acoustic, or chemical,to perform localised interventions. This boundary explicitly defines digital microsurgery robotics by the absence of continuous physical linkages between the operator and the end-effector, distinguishing them entirely from conventional macro-scale robotic platforms.
The scope includes magnetically actuated micro-swarms, biohybrid microrobots surgery platforms, image-guided magnetic control consoles, and the specialized navigational software required to coordinate multiple untethered agents simultaneously. It incorporates both the external generating hardware and the injectable or deployable consumable agents utilized during procedures. Platforms orchestrating these interventions often function alongside medical robots designed for gross anatomical access.
Traditional single-port laparoscopy systems and macro-scale orthopedic robots are excluded because they rely on fixed kinematic chains rather than untethered swarm behaviours. Passive contrast agents and standalone imaging modalities without a dedicated actuation layer are also omitted, as they provide observation rather than active, programmable mechanical intervention at the microvascular level.
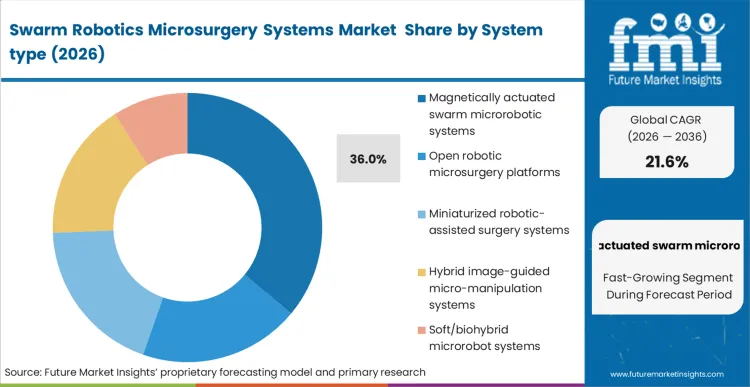
The reality behind magnetically actuated swarm microrobotic systems holding 36.0% of the magnetic microrobot surgery market is that chemical and acoustic actuation methods struggle with deep-tissue attenuation and precise steering in variable fluid flows. Procurement teams at translational institutes prioritize magnetic architectures because they integrate cleanly with existing MRI and fluoroscopic infrastructure.
In FMI's view, these microrobotic surgery systems transform the surgical approach, allowing clinicians to project force deep within the body without opening a physical tract. When operators deploy these swarms, they switch from manual spatial navigation to algorithmic trajectory planning, drastically reducing the cognitive load required to manage complex micro-environments. Institutions that attempt to utilise hybrid or softer modalities often face extended qualification timelines as they struggle to prove reliable retrieval or degradation of the untethered agents.
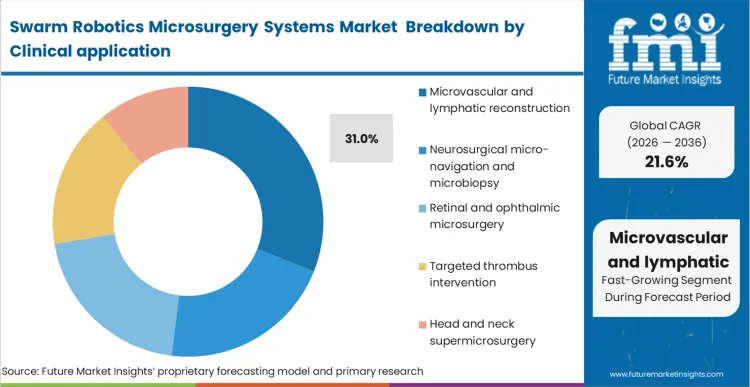
When traditional suturing techniques fail to achieve reliable patency in sub-millimeter vessels, surgical teams are forced to qualify advanced alternatives like microvascular reconstruction robots. Microvascular and lymphatic reconstruction commands 31.0% share because the cost of failure, tissue necrosis or graft loss, justifies the premium associated with robotic intervention, particularly in demanding fields like lymphatic microsurgery robotics.
As per FMI's projection, the precision required to manipulate 0.3mm vessels exceeds human physiological limits, making external tremor filtration non-negotiable for neurosurgical microrobots. When utilizing these systems, reconstructive surgeons operate through robotics assisted telesurgery interfaces, relying heavily on visual overlays rather than tactile feedback. Departments that delay integrating these capabilities find their success rates plateauing, ultimately losing referrals for complex free-flap reconstructions to more advanced centers.
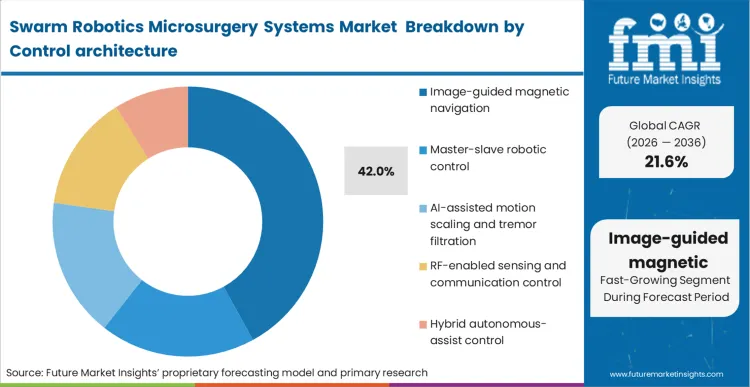
The decision facing clinical engineering directors is no longer whether to adopt external guidance, but how to ensure zero-latency tracking of untethered entities using image-guided microrobotics for surgery. Image-guided magnetic navigation holds 42.0% share because operators cannot manipulate what they cannot see, and swarms require continuous, closed-loop spatial verification.
According to FMI's estimates, magnetic navigation in microsurgery dictates the entire workflow, as the tracking software must communicate with the magnetic generators at millisecond intervals to prevent swarm dispersion. By embedding this control layer into their robot assisted surgical microscope systems, hospitals achieve the necessary synchronization for complex tasks. Facilities attempting to rely on open-loop or purely predictive models face unacceptable risks of collateral tissue damage during delicate interventions.
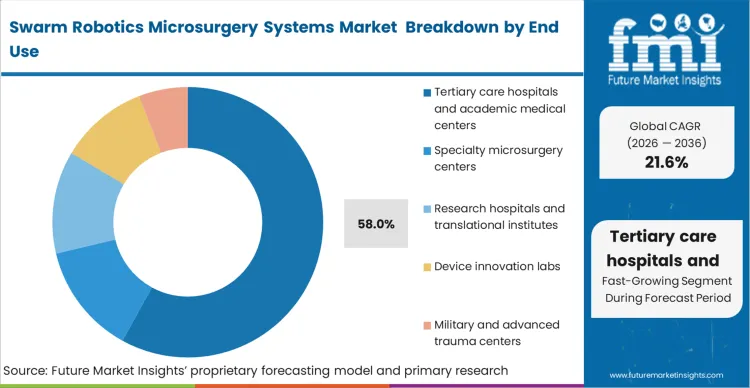
Tertiary care hospitals and academic medical centers do not just purchase equipment; they construct entire ecosystems capable of supporting experimental modalities, which is why they capture 58.0% of the market. FMI projects that these institutions possess the multi-disciplinary teams,surgeons, physicists, and bioengineers, required to safely deploy untethered microrobots, including specialized soft microrobots in microsurgery. They are uniquely positioned to absorb the massive footprint of the magnetic generators and the specialized surgical operating microscope suites. When these centers transition to swarm protocols, they restructure their entire scheduling and sterilization workflows to accommodate the new hardware. Smaller specialty centers that attempt to integrate these systems without the prerequisite imaging and engineering support face severe operational bottlenecks and extended downtime.
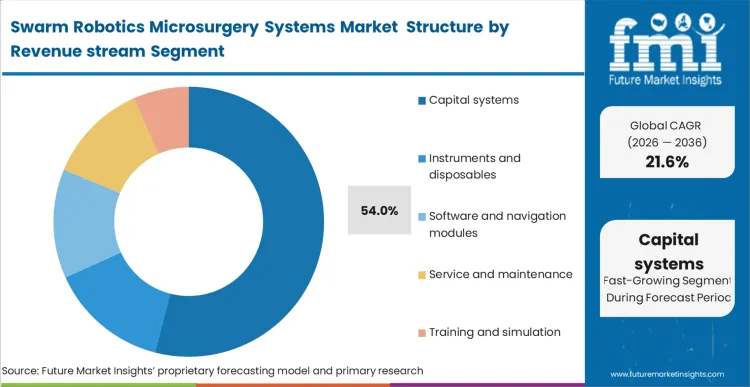
The commercial trajectory of this market is heavily front-loaded, with capital systems representing 54.0% of total revenue.
Based on FMI's assessment, the sheer complexity of the external magnetic generation coils, combined with sophisticated 3d surgical microscope systems, forces a massive initial layout before a single procedure occurs. Hospitals treat this not as a tool purchase, but as a facility-level infrastructure upgrade. Once the capital is installed, the dynamic changes; procurement teams lock into proprietary consumable streams for the microrobots themselves. Vendors who fail to secure the initial capital footprint are entirely locked out of the subsequent, highly lucrative recurring revenue generated by the specialized agents and proprietary software updates.
The necessity to resolve complex microvascular pathologies without inducing collateral trauma compels neurosurgical and ophthalmic departments to abandon traditional instrumentation in favour of a specialized head and neck supermicrosurgery robot. This pressure requires surgical directors to evaluate untethered platforms that can navigate deep, tortuous anatomy while maintaining sub-millimeter precision. Because the clinical penalty for endothelial damage in procedures like targeted thrombus intervention is severe, hospital networks must invest in systems that completely eliminate human tremor and physical tether friction. The inability to achieve these outcomes with legacy tools makes the transition to swarm architectures an operational imperative rather than a discretionary upgrade.
When evaluating what are the biggest barriers to swarm robotic microsurgery adoption, integrating these systems into existing clinical workflows faces severe friction due to the massive physical and infrastructural footprint required by magnetic field generators. Hospital facility managers cannot simply wheel a swarm control unit into a standard operating room; the surrounding environment requires extensive magnetic shielding and integration with advanced surgical navigation system arrays. This constraint limits early adoption to institutions capable of undertaking significant architectural renovations. While modular, lower-field generators are emerging, they currently lack the penetration depth necessary for complex cranial or deep-torso interventions.
The regional breakdown shows that the Swarm Robotics Microsurgery Systems market spans more than 40 countries and is divided into three primary regions: Asia Pacific, Europe, and North America.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 25.4% |
| China | 24.9% |
| France | 22.1% |
| United States | 20.8% |
| Germany | 20.4% |
| United Kingdom | 19.6% |
| Japan | 18.7% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Infrastructure readiness uniquely shapes the adoption pattern in this region, as expanding healthcare networks leapfrog legacy master-slave robotics directly into advanced untethered paradigms. Unlike mature Western markets burdened by massive installed bases of conventional robotic systems, major hospitals here are integrating swarm-capable hybrid imaging suites directly into their greenfield facility designs.
In FMI's view, this lack of entrenched capital allows clinical directors to bypass intermediate technologies. The procurement cycle prioritises high-throughput platforms capable of managing immense patient volumes, favouring systems that integrate flawlessly with orthopedic navigation systems and neurological imaging tools.
FMI's report includes secondary markets across Southeast Asia and Oceania. These emerging zones largely function as fast-followers, waiting for regulatory harmonization and cost-reduction in capital hardware before committing to extensive facility upgrades.
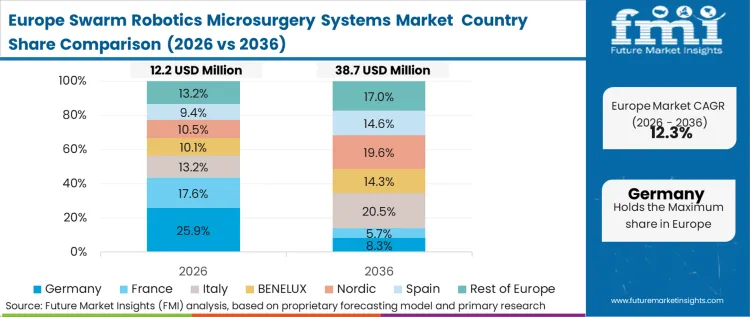
The regulatory environment defining the Europe robotic microsurgery systems market, driven by stringent directives on clinical efficacy and patient safety, forces buyers to heavily prioritize systems with impeccable retrieval and degradation profiles. Rather than evaluating sheer technological capability, procurement committees focus strictly on the biocompatibility and clearance mechanisms of the untethered agents. FMI analysts suggests that this policy-led environment restricts early commercialization to platforms that can definitively prove zero residual toxicity. Consequently, adoption is concentrated in specialized innovation hubs that maintain close relationships with regulatory bodies, allowing for careful, phased rollouts of robotic assisted endovascular systems.
FMI's report includes analysis of the Nordic and Benelux regions. Hospitals in these areas prioritize software interoperability, ensuring new swarm control modules can interface securely with their existing digital health records and imaging archives.
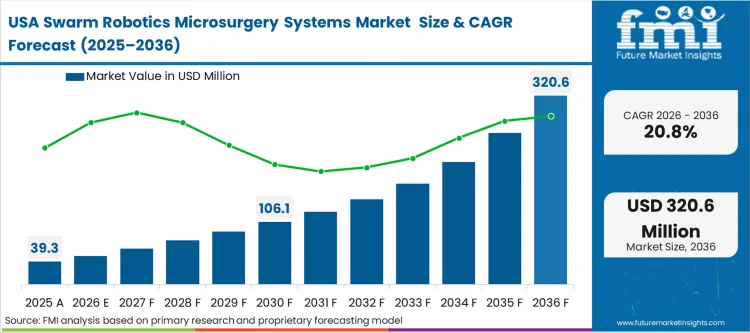
Capital availability and competitive intensity define the North American trajectory, as elite academic medical centers race to be the first to market with cutting-edge micro-interventions. This economics-led environment shaping the United States swarm robotics microsurgery market means that while the technology is still experimental, the marketing value of possessing a "swarm-capable" surgical suite drives immediate investment from top-tier institutions. These early adopters leverage their massive endowments to absorb the high initial costs, utilizing the technology to attract high-net-worth patients and leading surgical innovators. The integration with existing micro guide catheters infrastructure provides a familiar operational baseline for surgical teams transitioning to untethered modalities.
FMI's report includes detailed assessments of the Canadian market landscape. Provincial health authorities there exhibit a markedly different pattern, pooling capital resources into centralized, highly specialized surgical hubs rather than attempting distributed adoption.
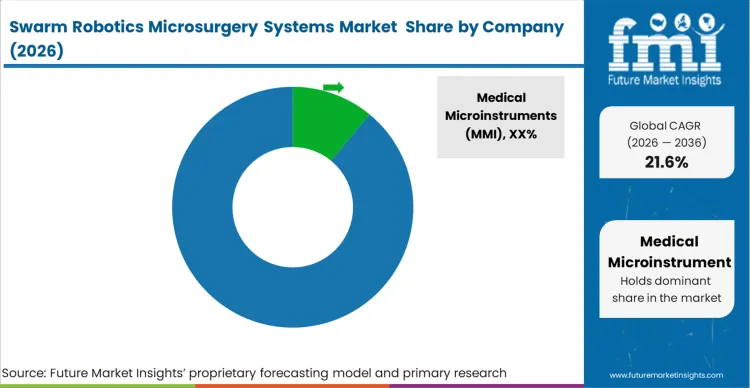
Evaluating the supplier landscape for robotic microsurgery systems, the highly concentrated nature of the swarm robotics microsurgery market stems from the massive intellectual property and capital barriers required to develop real-time closed-loop magnetic navigation. Medical Microinstruments (MMI), Intuitive Surgical, and Distalmotion operate in an environment where the primary variable determining vendor qualification among robotic microsurgery system manufacturers is the ability to guarantee untethered agent retrieval or safe biodegradation. Procurement boards simply will not review alternatives to conventional microsurgery robots that cannot demonstrate flawless tracking within deep anatomical structures.
When assessing which companies make robotic microsurgery systems, incumbents like Moon Surgical and Asensus Surgical hold a distinct advantage because they have already navigated the complex regulatory pathways for integrating advanced motion-scaling and imaging into surgical workflows. This advantage persists because hospital IT and clinical engineering departments are highly resistant to onboarding entirely new software architectures. Challengers like Virtual Incision and Robeauté must build native interoperability with legacy microcatheter imaging arrays to replicate this capability. In FMI's view, building an isolated, proprietary imaging ecosystem is a direct path to vendor disqualification.
As the market approaches 2036, large healthcare networks are actively resisting the proprietary lock-in characteristic of early capital deployments, focusing deeply on long-term microsurgery robot pricing. Procurement directors are demanding modular control architectures that allow them to source swarm consumables independently of the magnetic field generators, leading them to engage with diverse robotic microsurgery training platform vendors. This tension between buyer desire for interoperability and vendor incentives for closed ecosystems will dictate the next phase of consolidation, forcing early hardware innovators to pivot toward high-margin micro balloon catheter software and algorithmic licensing models, moving beyond simple capability comparisons like MMI Symani vs da Vinci SP.
| Metric | Value |
|---|---|
| Quantitative Units | USD 58.4 million to USD 412.8 million, at a CAGR of 21.6% |
| Market Definition | Swarm robotics microsurgery encompasses untethered, micro- or nano-scale robotic entities guided by external fields to perform localized interventions. It functionally excludes traditional tethered master-slave systems, focusing entirely on programmable, fluid-environment navigation without physical linkages. |
| System type Segmentation | Magnetically actuated swarm microrobotic systems, Open robotic microsurgery platforms, Miniaturized robotic-assisted surgery systems, Hybrid image-guided micro-manipulation systems, Soft/biohybrid microrobot systems |
| Clinical application Segmentation | Microvascular and lymphatic reconstruction, Neurosurgical micro-navigation and microbiopsy, Retinal and ophthalmic microsurgery, Targeted thrombus intervention, Head and neck supermicrosurgery |
| Control architecture Segmentation | Image-guided magnetic navigation, Master-slave robotic control, AI-assisted motion scaling and tremor filtration, RF-enabled sensing and communication control, Hybrid autonomous-assist control |
| End user Segmentation | Tertiary care hospitals and academic medical centers, Specialty microsurgery centers, Research hospitals and translational institutes, Device innovation labs, Military and advanced trauma centers |
| Revenue stream Segmentation | Capital systems, Instruments and disposables, Software and navigation modules, Service and maintenance, Training and simulation |
| Regions Covered | Asia Pacific, Europe, North America |
| Countries Covered | India, China, France, United States, Germany, United Kingdom, Japan, and 40 plus countries |
| Key Companies Profiled | Medical Microinstruments (MMI), Intuitive Surgical, Distalmotion, Moon Surgical, Asensus Surgical (part of KARL STORZ), Virtual Incision, Robeauté |
| Forecast Period | 2026 to 2036 |
| Approach | Primary interviews targeted Chief Medical Officers and surgical robotics directors. The baseline was anchored to hospital procurement budgets allocated for advanced imaging and surgical suite retrofits. Forecasts were cross-validated against institutional grant funding flows and bioengineering venture deployment patterns. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
What is the market size of robotic microsurgery?
The industry reaches USD 58.4 million in 2026. This figure reflects the intense initial capital outlays required by early-adopter academic centers installing dedicated magnetic generation suites, rather than high procedural volumes.
What will it be valued at by 2036?
The sector advances to USD 412.8 million by 2036. This expansion signals the shift from isolated research installations to standardized deployment across broader tertiary hospital networks handling complex neurovascular caseloads.
What CAGR is projected?
A CAGR of 21.6% is projected over the forecast period. This rate is constrained by the lengthy architectural upgrade cycles required to physically shield and house peripheral micro catheters tracking modalities within existing surgical footprints.
What are swarm robotics microsurgery systems?
Functionally, they encompass untethered, micro- or nano-scale robotic entities that operate cooperatively inside the human body under external guidance,typically magnetic, acoustic, or chemical, to perform localized interventions. This boundary explicitly defines the market by the absence of continuous physical linkages between the operator and the end-effector.
How do swarm microrobots work in surgery?
Operators switch from executing manual physical pathways to plotting strategic coordinates, allowing the software to manage micro-corrections. The integration of real-time imaging with algorithmic field shaping removes human latency, directing magnetic or acoustic fields to drive untethered entities to precise sub-millimeter targets.
Compare swarm microrobots with conventional robotic surgery?
Conventional robotic surgery relies on master-slave kinematics and physical tethers to execute macroscopic motion scaling. Swarm microrobots completely eliminate physical linkages, utilizing programmed external fields to manipulate hundreds of micro-agents simultaneously within fluid environments or deep-tissue regions where mechanical arms cannot fit.
Are microrobots used in real surgeries today?
While fully autonomous swarms remain largely in advanced translational trials, foundational components of magnetically guided single-entity interventions are actively utilized in early-adopter clinical settings. Academic medical centers are the primary entities piloting these systems under strict regulatory oversight for specialized neurosurgical and ophthalmic cases.
What are the biggest barriers to swarm robotic microsurgery adoption?
The massive infrastructural footprint required to support hybrid magnetic generation suites prevents rapid adoption. Facilities must execute extensive architectural shielding and integrate new endoscopy visualization system arrays before deploying these platforms.
Which clinical specialties will adopt these systems first?
Microvascular and lymphatic reconstruction commands the highest priority as the clinical cost of anastomotic failure outweighs the acquisition premium of robotic platforms. Motion scaling is mandatory when maneuvering 0.3mm vessels beyond human physiological limits.
Why is magnetic navigation important in microrobot surgery?
Untethered entities require continuous, closed-loop spatial verification to prevent systemic migration. Operators cannot manipulate deep-tissue swarms using purely predictive minimally invasive neurosurgery devices models without the deep penetration capabilities of magnetic fields.
Which countries will lead the swarm robotics microsurgery market?
India expands at 25.4%, significantly outpacing mature markets, alongside China at 24.9%. The difference lies in greenfield tertiary expansion; Asian hospital networks are integrating swarm-capable suites into new builds, bypassing the friction of retrofitting legacy operations prevalent in the US and Europe.
Which companies make robotic microsurgery systems?
The market is led by pioneers focused on untethered guidance and motion scaling, including Medical Microinstruments (MMI), Intuitive Surgical, Distalmotion, Moon Surgical, Asensus Surgical, Virtual Incision, and Robeauté. These vendors control the proprietary consumable streams for the swarms, effectively locking the hospital into long-term service relationships.
How do European biocompatibility regulations shape the market?
Strict EU directives force procurement boards to heavily scrutinize the degradation and retrieval profiles of the untethered agents. This limits adoption to highly specialized hubs capable of executing rigorous, longitudinal safety validations before broad commercialization.
Why is the vendor landscape so concentrated?
The requirement to seamlessly interlock proprietary magnetic field hardware with sophisticated, low-latency tracking software establishes a massive barrier to entry. Buyers demand fully integrated endoscopy video systems ecosystems, leaving little room for fragmented component suppliers to compete.
Why does China experience such rapid adoption?
Domestic translational research institutes in China operate under aggressive government-mandated commercialization timelines. This accelerates the jump from benchtop prototyping to advanced clinical trials, establishing a highly self-reliant surgical capital ecosystem.
How do soft biohybrid systems differ operationally from magnetic platforms?
Biohybrid systems offer exact tissue compliance, making them essential for delicate retinal procedures where rigid instruments induce tearing. They trade the deep-tissue penetration of magnetic systems for superior surface-level precision in ocular and soft-tissue environments.
Why are academic medical centers the primary end users?
Only tier-1 academic centers possess the multidisciplinary teams, surgeons, physicists, and bioengineers required to troubleshoot early-stage untethered platforms. They leverage this capability to secure elite trial qualifications and dictate future reimbursement models.
How do targeted thrombus interventions utilize swarm technology?
Swarms can be directed to mechanically agitate and chemically degrade blockages in distal neurovascular beds that standard catheters cannot physically access. This capability alters the treatment window for critical stroke interventions.
Why does Japan's market grow slower than India's despite high healthcare expenditure?
Japan's mature, data-driven clinical governance boards require exhaustive systemic toxicity and retrieval data before authorizing untethered interventions. This meticulous qualification process delays initial adoption but results in highly optimized workflows once implemented.
How should investors benchmark this niche against digital microsurgery?
Investors should view swarm architectures as the final frontier of the digital microsurgery continuum. While digital microsurgery focuses on ergonomic visualization and tethered motion scaling, swarm systems monetize the complete removal of the mechanical instrument, capturing high-margin consumable streams that digital optics alone cannot secure.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.