Freeze Thaw System Market
The Freeze Thaw System Market is segmented by Product Type (freeze-thaw modular platforms, controlled rate chambers, single-use bags, and shippers), Application (R&D, pre-clinical & clinical, and commercial), End User (academic & research institutes, pharmaceutical & biotechnology companies, contract research organizations, and contract manufacturing organizations), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Freeze Thaw System Market Size, Market Forecast and Outlook By FMI
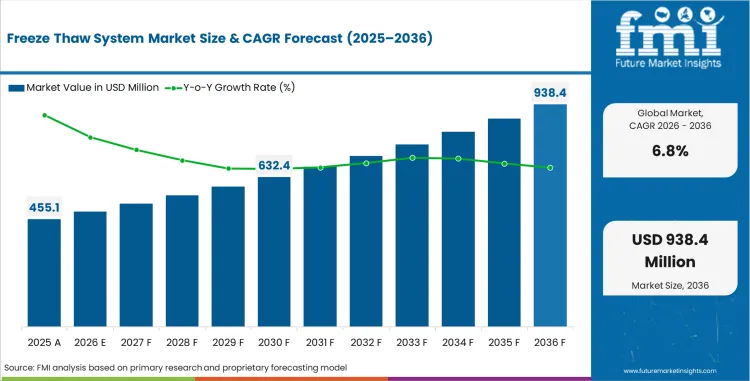
In 2026, the freeze thaw system market was valued at USD 455.1 million. Based on Future Market Insights analysis, demand for freeze thaw system is estimated to grow to USD 877.6 million by 2036. FMI projects a CAGR of 6.8% during the forecast period.
Absolute dollar growth of USD 422.5 million over the decade signals steady expansion rather than a demand reset. As per FMI, demand is expected to remain supported by rising biologics production volumes, broader use of single-use bioprocessing workflows, and increasing need for controlled material handling during upstream and downstream operations. At the same time, capital budgeting discipline, validation requirements, and process standardization are expected to keep value growth measured across mature biomanufacturing markets.
Summary of Freeze Thaw System Market
-
Freeze Thaw System Market Definition
- The industry covers controlled temperature handling systems used to freeze, store, thaw, and transfer biological materials across research and biomanufacturing workflows.
- Demand Drivers in the Market
- Demand is being sustained by increasing biologics batch volumes requiring controlled freeze-thaw handling across production workflows.
- Greater use of single-use bioprocessing is driving broader adoption of contamination-conscious freeze-thaw systems and bags.
- Expansion in commercial biologics manufacturing is raising demand for scalable and validated freeze-thaw handling solutions, as reported by FMI.
- Key Segments Analyzed in the FMI Report
- By Product Type: single-use bags lead at 37.1% share in 2026.
- By Application: commercial leads at 62.3% share in 2026.
- Geography: India and the USA show strong growth at 8.7% and 7.6% CAGR, supported by rising biologics capacity and expanding process infrastructure.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Consultant at Future Market Insights, opines, ‘Buyer preference is being shaped by controlled thaw performance, single-use process compatibility, and operating consistency, as bioprocess stakeholders place greater value on batch protection, handling efficiency, and contamination-aware workflow design.’
- Strategic Implications and Executive Takeaways
- Strengthen buyer contracting with validation-ready system support, scale-compatible workflow design, and process integration aligned to biologics handling needs.
- Build growth plans around commercial bioprocessing environments where higher material volumes require controlled and repeatable freeze-thaw operations.
- Protect approved supplier positions through temperature reliability and workflow familiarity, since validated process handling systems are not switched easily once embedded into manufacturing routines.
- Methodology
- As per FMI, sizing and segment splits were reconciled using company primary disclosures and demand mapping by bioprocess stage, product format, and application setting.
- Product and application shares were validated by linking freeze-thaw demand patterns to biologics handling throughput across research, clinical, and commercial environments.
- Pricing sensitivity was stress tested under scale-up and process substitution scenarios to confirm revenue response under realistic biomanufacturing conditions.
“With this partnership, Sartorius continues to strengthen its leading position in the freeze-thaw market, the combination of our robust Celsius single-use containers with market-leading freeze-thaw equipment from HOF will provide our customers with a simplified supply chain for improved management of drug substance.” said Eva Lindskog, Head of Fluid Management Technologies at Sartorius. [1]
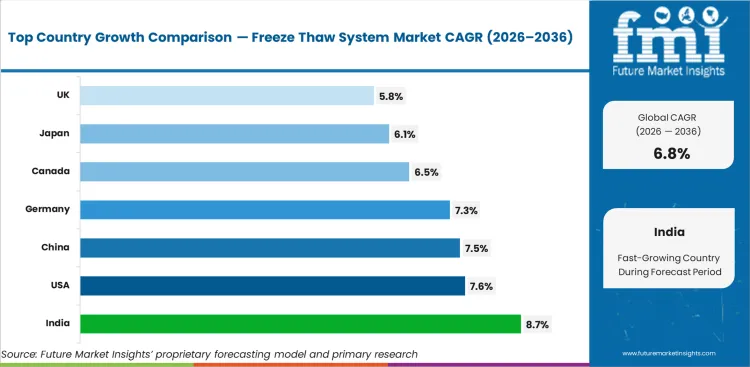
Based on FMI’s report, India (8.7% CAGR), the USA (7.6%), China (7.5%), Germany (7.3%), and France (7.0%) are expected to lead growth due to expanding biologics infrastructure, stronger commercial manufacturing activity, and growing integration of controlled temperature handling into regulated process workflows. Mature markets such as Spain (6.8%), Canada (6.5%), Japan (6.1%), the UK (5.8%), and Italy (5.3%) are expected to contribute mainly through replacement demand, validation-led upgrades, and continued process optimization across established biologics environments.
Freeze Thaw System Market Definition
The market includes systems and related product formats used to freeze, store, thaw, and transfer temperature-sensitive biological materials during bioprocessing operations. These products are sold to academic and research institutes, pharmaceutical and biotechnology companies, contract research organizations, and contract manufacturing organizations to support controlled handling of biologics, intermediates, and process fluids where temperature-managed transfer and batch protection are operationally required.
The market includes freeze-thaw modular platforms, controlled rate chambers, single-use bags, and shippers used across research, clinical, and commercial bioprocessing environments. The market is driven by biologics manufacturing volumes, single-use process adoption, workflow standardization, and demand for validated temperature control. The revenue stream is derived from sales of freeze thaw systems and related handling formats across bioprocessing settings.
Freeze Thaw System Market Inclusions
The report provides global and regional market sizing, as well as a 10-year forecast from 2026 to 2036. The report also provides market sizing by product type, application, and end user, with country-level CAGR comparisons across key markets. The report also provides analysis on competitive positioning of key suppliers, process workflow dynamics, and the influence of temperature control precision, contamination management, and scale-up capability on supplier choice, as per FMI.
Freeze Thaw System Market Exclusions
The scope does not include general cold storage equipment, standalone laboratory refrigerators, cryogenic transport systems outside freeze-thaw workflow use, or unrelated upstream and downstream bioprocessing hardware. Standard packaging materials, conventional freezers not positioned within controlled freeze-thaw operations, and unrelated logistics equipment are also excluded. The scope also does not include non-bioprocess temperature handling products not used in structured freeze-thaw workflows, but only freeze thaw systems used for controlled biological material handling.
Freeze Thaw System Market Research Methodology
- Primary Research: Interviews were carried out with bioprocess engineers, manufacturing managers, process development teams, and supplier-side commercial participants to confirm workflow demand patterns and purchase behavior.
- Desk Research: Public data from regulatory bodies, company materials, bioprocessing publications, manufacturing references, and life sciences infrastructure sources were analyzed to compare product use trends and market positioning.
- Market Sizing and Forecasting: A hybrid approach was created based on process-stage demand mapping, product mix, and biologics handling requirements, and then harmonized through volume and price triangulation.
- Data Validation and Update Cycle: Results were filtered for variance, tested for internal consistency, and assessed through structured peer review before distribution.
Segmental Analysis
Freeze Thaw System Market Analysis by Product Type
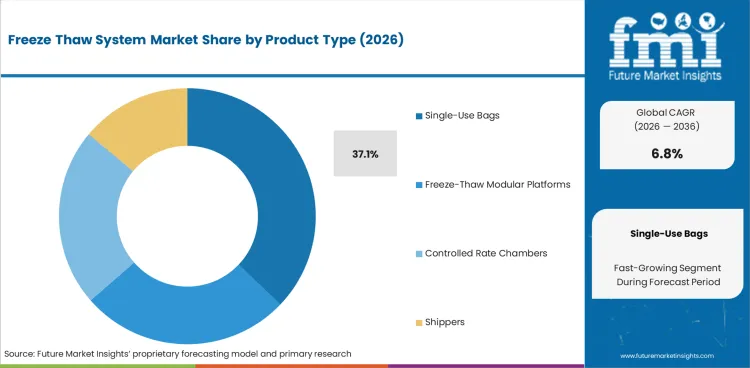
Based on FMI’s report, single-use bags are estimated to hold 37.1% share in 2026. This lead position is supported by their strong fit within contamination-controlled bioprocessing workflows, growing use in flexible biologics manufacturing environments, and broader adoption of single-use systems across upstream and downstream operations. Their larger share is being reinforced by lower cleaning burden, easier integration into disposable process architecture, and increasing preference for scalable handling formats across regulated production settings.
- Strong Biologics Process Compatibility: Freeze-thaw handling is widely used to preserve biologic drug substance, and recent literature shows this step must be tightly controlled to protect product quality. Bag-based single-use systems fit this need well because they are designed for scalable frozen storage and controlled thawing within modern biologics workflows. [2]
- Scalable Batch-Handling Preference: Single-use bags hold the highest share because they support flexible batch sizing and easier scale-up across biologics manufacturing, allowing frozen intermediates and bulk drug substance to be handled more efficiently within modern modular production setups.
- Storage and transfer simplicity: Single-use bags hold the highest share because they simplify frozen material storage, movement, and connection across bioprocess steps, making them a practical format for handling biologic intermediates without adding complexity to closed manufacturing operations.
Freeze Thaw System Market Analysis by Application
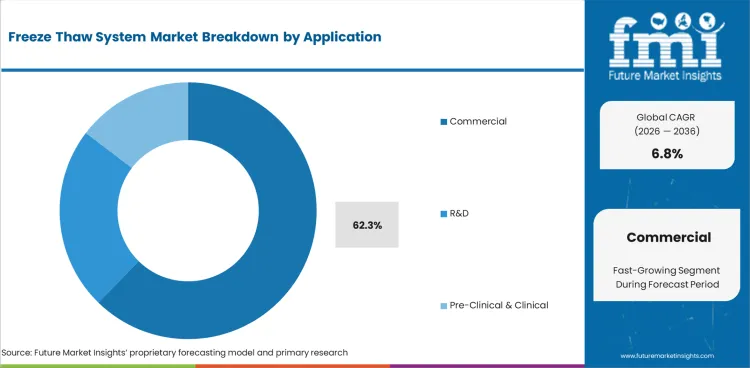
Commercial accounts for 62.3% share in 2026, based on FMI’s report, reflecting its role as the primary demand pool within freeze-thaw system use. This leading position is supported by higher biologics production volumes, stronger dependence on repeatable material handling, and broader requirement for validated freeze-thaw control in full-scale manufacturing environments. Demand is therefore being concentrated in commercial use where batch protection, throughput, and process consistency remain operational priorities.
- Higher Manufacturing-Scale Dependency: Commercial biologics production relies more heavily on freeze-thaw operations because bulk drug substance is often frozen to enhance stability, extend shelf life, and increase manufacturing flexibility at scale, making controlled freeze-thaw handling more central in full-scale production than in early-stage use. [3]
- Batch Protection Drives Deployment: Commercial environments account for the highest share because post-manufacturing handling stresses can affect biologic quality, safety, and efficacy, so manufacturers depend on more controlled freeze-thaw workflows when managing repeated high-volume batches intended for market supply. [4]
- Routine Production-Cycle Reliance: Commercial holds the highest share because freeze-thaw handling becomes a routine operational requirement in full-scale biologics manufacturing, where repeated batch movement, storage, and release workflows create more consistent system demand than development-stage applications.
Freeze Thaw System Market Drivers, Restraints, and Opportunities
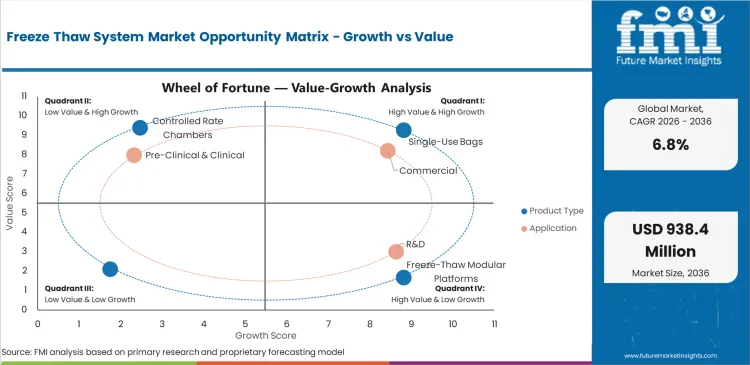
Future Market Insights analysis indicates that historical patterns point to a process-support biomanufacturing category where demand is anchored in temperature control, biologic stability, and contamination-aware material handling across development and production workflows. Estimated valuation in 2026 is being supported by broader biologics manufacturing activity, increasing use of single-use technologies, and continuing need for controlled storage and thawing of process materials, as per FMI.
While category demand is being restrained by validation complexity, capital approval cycles, and the need for integration within highly standardized process environments, value is being supported by scale-up of commercial biologics, outsourcing growth, and increasing process sophistication across life sciences manufacturing. Based on FMI’s report, the forecast trajectory reflects a market where single-use bags remain the leading product type, while suppliers are differentiated by temperature performance, scale flexibility, and process integration strength.
- Biologics-Backed: Demand is being supported as manufacturers continue to expand controlled handling capacity for sensitive biological materials.
- Validation-Led: Growth is being moderated where qualification, integration, and process compliance requirements increase adoption complexity.
- Commercial Scale-Up: Demand is being supported as more biologics production is being expanded into higher-volume and more structured manufacturing environments.
Regional Analysis
Based on the regional analysis, freeze thaw system market is segmented into North America, Latin America, East Asia, South Asia & Pacific, Western Europe, Eastern Europe, and Middle East & Africa across 40+ countries. Regional performance is assessed using country-level demand signals tied to biologics production, bioprocess infrastructure, outsourcing activity, and manufacturing modernization, as per FMI. The full report also offers market attractiveness analysis based on regional trends.
.webp)
| Country | CAGR |
|---|---|
| United States | 7.6% |
| Canada | 6.5% |
| Japan | 6.1% |
| China | 7.5% |
| India | 8.7% |
| United Kingdom | 5.8% |
| Germany | 7.3% |
| France | 7.0% |
| Spain | 6.8% |
| Italy | 5.3% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Freeze Thaw System Market Analysis
North America is shaped by advanced biologics manufacturing capability and strong process standardization, where product choice is influenced by temperature control precision, contamination management, and compatibility with commercial-scale bioprocessing workflows. GE Healthcare holds strong placement through broad bioprocess integration and established customer relationships across production environments. BioLife Solutions, Inc. and Thermo Fisher Scientific, Inc. compete through process-focused portfolios and strong relevance across scalable life sciences handling systems. Based on FMI’s report, demand remains anchored in commercial biologics manufacturing and process optimization activity.
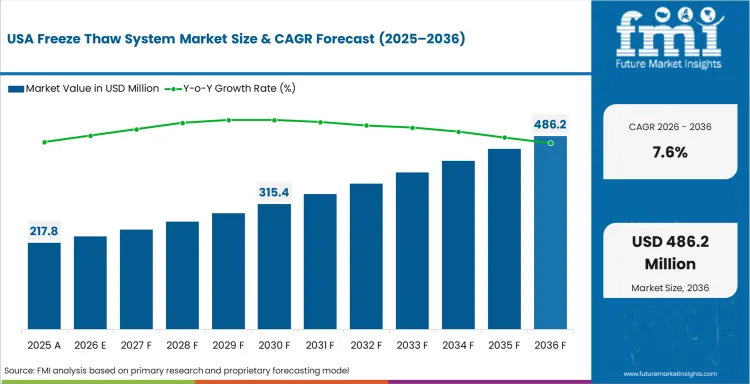
- United States: The demand for freeze thaw system in the United States is expected to grow at a CAGR of 7.6% during the forecast period of 2036. This is driven by broad biologics production activity, continued investment in process control, and strong integration of single-use and temperature-managed workflows, although this is tempered by validation intensity and structured capital planning.
- Canada: Demand for freeze thaw system in Canada is projected to rise at 6.5% CAGR through 2036. Growth is supported by expanding life sciences infrastructure, continued bioprocess investment, and stronger use of controlled material handling across research and manufacturing environments.
FMI’s report includes a detailed analysis of the growth in the North American region, along with a country-wise assessment that includes the United States and Canada. Readers can also find regional trends, regulations, and market growth based on different segments and countries in the North America region.
East Asia Freeze Thaw System Market Analysis
East Asia continues to be a high-growth demand market, driven by expanding biomanufacturing infrastructure, rising biologics development activity, and broader investment in temperature-controlled process systems. Supplier positioning in the region is influenced by workflow scalability, single-use compatibility, and ability to support both development and commercial operations across regulated production settings. Based on FMI’s report, demand remains supported by expanding manufacturing capacity and stronger process modernization across biologics value chains.
- China: The freeze thaw system market in China is estimated to expand at a CAGR of 7.5% during the forecast period of 2036. This is being driven by rising biologics capacity, broader commercial manufacturing build-out, and increasing need for validated material handling, with market dynamics influenced by infrastructure expansion and process standardization.
- Japan: The demand for freeze thaw system in Japan is expected to grow at a CAGR of 6.1% during the forecast period of 2036. This is driven by structured bioprocess use, stable commercial manufacturing demand, and continuing preference for reliable process control, although this remains shaped by mature infrastructure and disciplined capital allocation.
The full report analyzes the freeze thaw system market across East Asia from 2021 to 2036, covering pricing, trends, and growth drivers in China, Japan, South Korea, and related regional markets. The assessment highlights trends that dictate regional demand and procurement behaviour.
South Asia & Pacific Freeze Thaw System Market Analysis
South Asia & Pacific is being shaped by rising biologics manufacturing activity, expanding life sciences capacity, and stronger integration of controlled process handling across growing production networks. Supplier positioning in the region is influenced by product affordability, system validation support, and ability to align with evolving bioprocess requirements. Based on FMI’s report, demand remains supported by commercial build-out and growing use of structured freeze-thaw workflows in development and manufacturing settings.
- India: Demand for freeze thaw system in India is projected to rise at 8.7% CAGR through 2036. Growth is supported by expanding biologics production capacity, stronger contract manufacturing activity, and increasing use of controlled temperature handling across commercial and development-stage bioprocessing operations.
The full report analyzes the freeze thaw system market across South Asia & Pacific from 2021 to 2036, covering pricing, trends, and growth drivers in India, ASEAN Countries, Australia & New Zealand, and Rest of South Asia. The assessment highlights trends that dictate regional demand and procurement behaviour.
Western Europe Freeze Thaw System Market Analysis
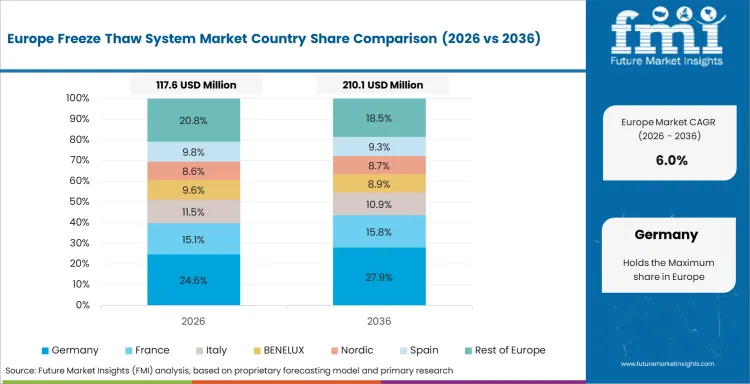
Western Europe is quality-led and process-disciplined, where demand is shaped by biologics production maturity, structured validation practice, and strong reliance on controlled bioprocess workflows. Sartorius, Single Use Support GmbH, and Meissner Filtration Products, Inc. maintain relevance through process-integrated portfolios, manufacturing credibility, and alignment with regulated life sciences environments. Use continues to be influenced by scale-up requirements, contamination control expectations, and adoption of validated process handling standards. As per FMI, recurring commercial and development-stage demand continues to shape system deployment across Western Europe.
- Germany: Demand for freeze thaw system in Germany is projected to rise at 7.3% CAGR through 2036. Growth is supported by advanced bioprocess infrastructure, strong biologics manufacturing depth, and continuing investment in validated temperature-controlled workflow systems.
- United Kingdom: The demand for freeze thaw system in the United Kingdom is expected to grow at a CAGR of 5.8% during the forecast period of 2036. This is driven by stable biologics development demand, structured process modernization, and broader use of controlled freeze-thaw operations, although this remains filtered through capital discipline and validation requirements.
- France: The demand for freeze thaw system in France is expected to grow at a CAGR of 7.0% during the forecast period of 2036. This is driven by expanding life sciences production demand, growing biologics process complexity, and increasing reliance on validated batch handling across development and manufacturing workflows.
- Spain: The demand for freeze thaw system in Spain is expected to grow at a CAGR of 6.8% during the forecast period of 2036. This is driven by rising manufacturing modernization, broader process handling needs, and continued use of controlled systems across biologics workflows.
- Italy: The demand for freeze thaw system in Italy is expected to grow at a CAGR of 5.3% during the forecast period of 2036. This is driven by stable bioprocess investment and increasing need for temperature-managed material handling, although this remains moderated by measured capacity expansion and validation-led adoption discipline.
FMI’s analysis of freeze thaw system market in Western Europe consists of country-wise assessment that includes Germany, the UK, France, Italy, Spain, BENELUX, Nordic Countries, and Rest of Western Europe. Readers can know various regulations and latest trends in the regional market.
Competitive Aligners for Market Players
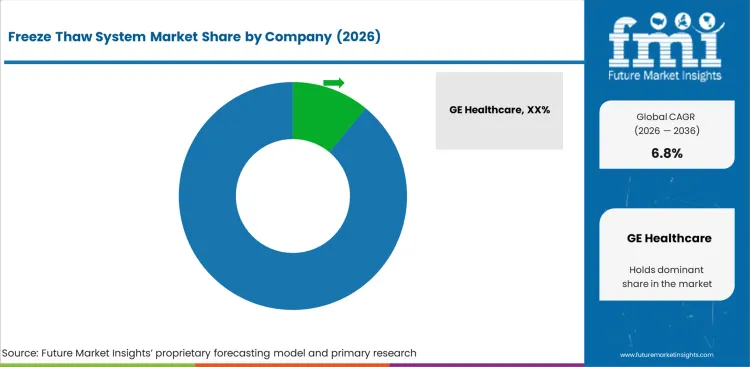
The market structure remains moderately concentrated, with effective competition centered on suppliers capable of supporting temperature control precision, process integration, and dependable handling performance across biologics production workflows. The data provided shows the presence of a leading participant in the market, with the rest of demand distributed across a focused set of bioprocess equipment and handling specialists. The key competitive factor is workflow reliability aligned to batch protection, as supplier qualification is filtered through validated performance, scalability, and compatibility with structured freeze-thaw operations. The product portfolio also reflects this emphasis, as single-use bags account for the leading share in the supplied segmentation, as indicated by FMI.
Companies with established life sciences manufacturing relationships, strong validation support, and broad process handling capability carry structural advantages because buyer qualification is governed by batch integrity, contamination control, and confidence in repeatable operation across sensitive biologics materials. Scale leaders benefit from deeper commercial bioprocess access, wider product integration capability, and the ability to serve multiple process steps under the same equipment framework. Portfolio breadth across platforms, chambers, bags, and shippers helps protect account positions when manufacturers standardize around validated suppliers. Suppliers with narrower operational reach tend to compete through selective geographies, niche bioprocess applications, or localized support capability, which can weaken their position during structured supplier evaluation.
Customer concentration reinforces buyer leverage. Pharmaceutical & biotechnology companies account for the largest end-user pool in the supplied segmentation, with academic & research institutes, CROs, and CMOs forming the next tier of demand. Large production networks commonly keep approved process suppliers active, prioritize systems that align with contamination-aware workflows, and favor vendors with dependable support across recurring batch activity. Pricing power is therefore moderated for standard freeze-thaw categories, with measured advantage retained only where temperature performance, validation support, or process fit creates stronger buyer preference, Future Market Insights analysis.
Recent Developments
- In March 2026, Ferring Pharmaceuticals today announced that the USA Food and Drug Administration (FDA) has approved a label update for ADSTILADRIN enabling an accelerated water-bath thawing method, completed in about 25 minutes, to enhance efficient clinical preparation for healthcare teams. [5]
- In January 2026, Meissner expands the application of its TepoFlex biocontainer platform into freeze and thaw operations, delivering the same industry-trusted quality and performance to this demanding workflow. [6]
- In May 2025, Hoshizaki Corporation has launched an industry-first thawing machine combining vacuum cooling and microwave heating. Designed for foodservice and retail sectors, the solution enables faster, high-quality thawing while reducing labor intensity, food waste, and operational inefficiencies-addressing key supply chain and workforce challenges across supermarkets, restaurants, and convenience store networks.
Key Players in Freeze Thaw System Market
- GE Healthcare
- BioLife Solutions, Inc.
- Farrar Scientific
- HOF Sonderanlagenbau GmbH
- Meissner Filtration Products, Inc.
- Single Use Support GmbH
- Thermo Fisher Scientific, Inc.
- W. L. Gore & Associates, Inc.
- ZETA Holding GmbH
- Sartorius
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 455.1 million (2026) to USD 877.6 million (2036), at a CAGR of 6.8% |
| Market Definition | The freeze thaw system market comprises controlled systems and related handling formats used to freeze, thaw, store, and transfer temperature-sensitive biological materials across research and biomanufacturing workflows, where demand is shaped by batch integrity protection, process control, and validated temperature management across regulated life sciences settings. |
| Product Type Segmentation | Freeze-Thaw Modular Platforms, Controlled Rate Chambers, Single-Use Bags, Shippers |
| Application Segmentation | R&D, Pre-Clinical & Clinical, Commercial |
| End User Segmentation | Academic & Research Institutes, Pharmaceutical & Biotechnology Companies, Contract Research Organizations, Contract Manufacturing Organizations |
| Regions Covered | North America, Latin America, East Asia, South Asia & Pacific, Western Europe, Eastern Europe, and Middle East & Africa |
| Countries Covered | United States, Canada, United Kingdom, Germany, France, China, Japan, South Korea, Brazil, Australia, India, and 40+ countries |
| Key Companies Profiled | GE Healthcare, BioLife Solutions, Inc., Farrar Scientific, HOF Sonderanlagenbau GmbH, Meissner Filtration Products, Inc., Single Use Support GmbH, Thermo Fisher Scientific, Inc., W. L. Gore & Associates, Inc., ZETA Holding GmbH, Sartorius |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top down and bottom up market modeling validated through primary interviews with bioprocess and procurement stakeholders, supported by demand triangulation across workflow volumes, application settings, and product adoption patterns, as per FMI. |
Freeze Thaw System Market Analysis by Segments
Product Type
- Freeze-Thaw Modular Platforms
- Controlled Rate Chambers
- Single-Use Bags
- Shippers
Application
- R&D
- Pre-Clinical & Clinical
- Commercial
End User
- Academic & Research Institutes
- Pharmaceutical & Biotechnology Companies
- Contract Research Organizations
- Contract Manufacturing Organizations
Region
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- East Asia
- China
- Japan
- South Korea
- South Asia & Pacific
- India
- ASEAN Countries
- Australia & New Zealand
- Rest of South Asia
- Western Europe
- Germany
- UK
- France
- Italy
- Spain
- BENELUX
- Nordic Countries
- Rest of Western Europe
- Eastern Europe
- Russia
- Hungary
- Poland
- Rest of Eastern Europe
- Middle East & Africa
- Saudi Arabia
- Türkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- [1] Sartorius (2021), Sartorius and HOF Sonderanlagenbau Partnership Creates Streamlined Offering of Freeze-Thaw Solutions for Biological Products
- [2] ScienceDirect (2024), Directional freezing and thawing of biologics in drug substance bottles
- [3] PubMed Central (2024), Optimization of Methodologies to Study Freeze/Thaw Processes in Drug Substance Bottles
- [4] ScienceDirect (2024), Impact of Post Manufacturing Handling of Protein-Based Biologic Drugs on Product Quality and User Centricity.
- [5] Ferring Pharmaceuticals (2026), USA FDA Approves Label Update to Accelerate Thaw Time for ADSTILADRIN.
- [6] Meissner (2026), Meissner’s TepoFlex Biocontainer Is Now Available for Freeze & Thaw Applications.
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
This Report Addresses
- Market intelligence to enable structured strategic decision making across biologics workflow expansion and freeze-thaw system planning
- Market size estimation and 10-year revenue forecasts from 2026 to 2036 for freeze thaw system demand
- Growth opportunity mapping across product type classes and application-stage handling pools
- Segment and regional revenue forecasts aligned to the stated taxonomy and covered geographies
- Competition strategy assessment including supplier positioning, portfolio fit, and bioprocess channel coverage benchmarking
- Product and compliance tracking focused on temperature control standards, process integrity, and validation-led quality expectations
- Regulatory impact analysis covering process-led system selection and provider-level protocol adoption in regulated care settings
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats for executive and operational use
Frequently Asked Questions
How large is the demand for Freeze Thaw System in the global market in 2026?
Demand for Freeze Thaw System in the global market is estimated to be valued at USD 455.1 million in 2026, as per FMI.
What will be the market size of Freeze Thaw System in the global market by 2036?
Market size for Freeze Thaw System is projected to reach USD 877.6 million by 2036.
What is the expected demand growth for Freeze Thaw System in the global market between 2026 and 2036?
Demand for Freeze Thaw System in the global market is expected to grow at a CAGR of 6.8% between 2026 and 2036.
Which product type is poised to lead global sales by 2026?
Single-use bags are expected to be the dominant product type, capturing 37.1% share in 2026.
How significant is the role of commercial in driving application demand in 2026?
Commercial is projected to hold 62.3% share of application demand in 2026.
Which country is expected to record the fastest growth during the forecast period?
India is projected to grow at a CAGR of 8.7% during 2026 to 2036 among the listed countries.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Single-Use Bags
- Freeze-Thaw Modular Platforms
- Controlled Rate Chambers
- Shippers
- Single-Use Bags
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Commercial
- R&D
- Pre-Clinical & Clinical
- Commercial
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Application
- Competition Analysis
- Competition Deep Dive
- GE Healthcare
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- BioLife Solutions, Inc.
- Farrar Scientific
- HOF Sonderanlagenbau GmbH
- Meissner Filtration Products, Inc.
- Single Use Support GmbH
- Thermo Fisher Scientific, Inc.
- W. L. Gore & Associates, Inc.
- ZETA Holding GmbH
- Sartorius
- GE Healthcare
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Product Type
- Figure 23: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Application
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Product Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Application
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Product Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Application
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Application
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Product Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Application
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

