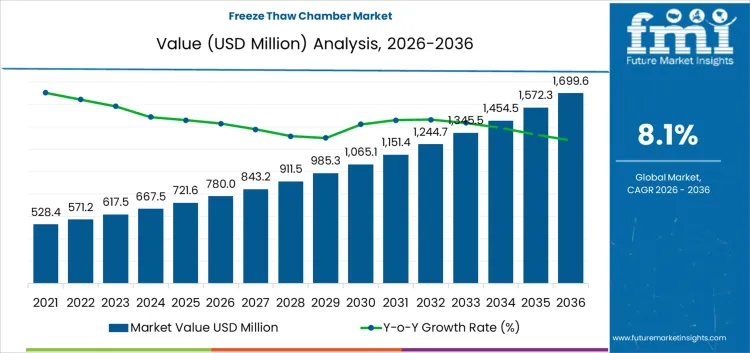
The freeze thaw chamber market is likely to be valued at USD 780.0 million in 2026 and is projected to reach USD 1,699.6 million by 2036, expanding at a CAGR of 8.1%. Value creation is directly linked to mandatory stability, durability, and excursion testing across pharmaceuticals, biologics, food products, and construction materials rather than discretionary laboratory upgrades.
Each freeze-thaw protocol addresses defined risk scenarios involving temperature excursions during storage, transport, or environmental exposure. Equipment demand scales with regulatory testing frequency, product sensitivity to phase transitions, and validation requirements embedded in quality systems. Capital allocation prioritizes chambers capable of repeatable cycling profiles, tight temperature tolerances, and traceable data capture aligned with audit expectations.
Adoption dynamics are shaped by protocol duration, sample residence time, and laboratory throughput constraints rather than simple test volume growth. Long cycle lengths limit sample turnover, increasing reliance on chambers with reliable ramp performance and uniform exposure under sustained operation. Energy intensity, compressor duty cycles, and defrost management influence total cost of ownership, especially in high-frequency testing environments. Laboratories favor configurations that balance footprint, capacity, and utility demand while supporting qualification and recalibration without extended downtime. Replacement demand emerges as legacy chambers struggle with uniformity drift, control obsolescence, or documentation gaps under evolving regulatory scrutiny. Growth persistence reflects expansion of stability-sensitive product portfolios and infrastructure durability testing rather than acceleration of experimental or exploratory research activity.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 780.0 million |
| Market Forecast Value (2036) | USD 1,699.6 million |
| Forecast CAGR 2026 to 2036 | 8.1% |
Demand for freeze thaw chambers is increasing as pharmaceutical and biotechnology developers require controlled environmental stress testing to assess stability of biologics, vaccines, and complex formulations under repeated temperature cycling. Stability scientists specify chambers with precise temperature control, uniform air circulation, and data logging to simulate transport and storage conditions that can impact product efficacy and shelf life. Procurement teams evaluate chamber capacity, ramp rate performance, calibration traceability, and integration with laboratory information systems to support high throughput testing and regulatory documentation. Regulatory authorities emphasize documented stability data across defined stress conditions to support product registration dossiers and ongoing quality systems, and chambers that deliver reproducible results help laboratories align with these expectations.
Growth in cell and gene therapy pipelines, vaccine development, and complex drug delivery systems reinforces uptake of freeze thaw chambers that support rigorous stress protocols. Quality assurance professionals require equipment with robust safety controls and fail safe features to protect samples and maintain test integrity during extended trials. Engineering groups coordinate chamber specifications with laboratory layouts to optimize utility connections, footprint, and maintenance access. Service and support networks provide preventive maintenance and calibration services that support predictable uptime and reduce risk of test delays. These technical and operational priorities are contributing to sustained demand for freeze thaw chambers in regulated research and development environments.
Demand for freeze thaw chambers is shaped by material durability verification, environmental stress simulation, and regulatory testing protocols. Laboratories and industrial users require controlled cycling to evaluate performance under repeated temperature transitions. Adoption aligns with quality assurance programs supporting pharmaceuticals, construction materials, food safety, and packaging integrity. System selection emphasizes temperature accuracy, cycle repeatability, chamber uniformity, and documentation capability. Segment classification reflects differentiation by chamber configuration, operational end use, and environmental control capability influencing testing scope, throughput, and compliance alignment.
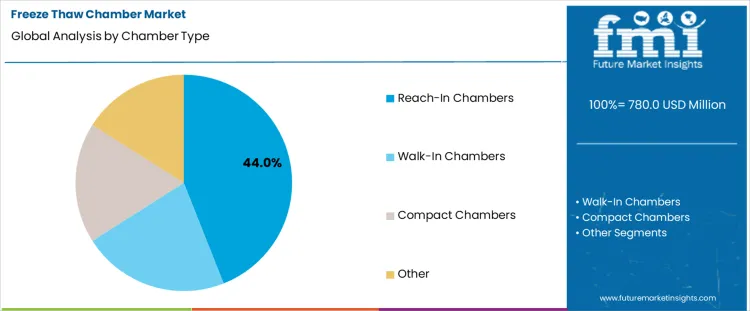
Reach-in chambers hold 44.0%, representing the largest share among chamber types due to compact footprint and operational flexibility. These chambers support standardized testing volumes while fitting laboratory and production-adjacent environments. Rapid access and ease of loading improve workflow efficiency during frequent cycling protocols. Energy consumption remains manageable compared with larger enclosures. Walk-in chambers support oversized samples and batch testing with higher space requirements. Compact chambers address benchtop testing with limited capacity. Chamber type segmentation reflects preference for versatile configurations balancing capacity, accessibility, and installation constraints across diverse testing environments.
Key Points
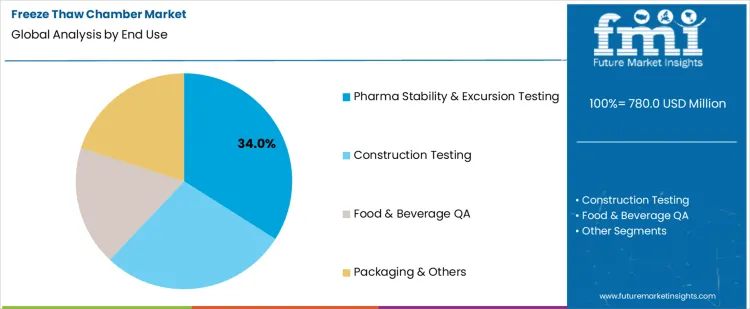
Pharma stability and excursion testing holds 34.0%, representing the largest share among end-use categories due to stringent validation requirements. Freeze thaw cycling verifies product stability during storage, transport, and handling deviations. Regulatory expectations drive routine testing with documented repeatability. Controlled environments ensure data integrity for quality submissions. Construction testing applies chambers to assess material resilience under thermal stress. Food and beverage quality assurance evaluates texture and integrity across cycles. Packaging and other uses address distribution durability. End-use segmentation reflects concentration where compliance, risk mitigation, and documentation rigor remain highest.
Key Points
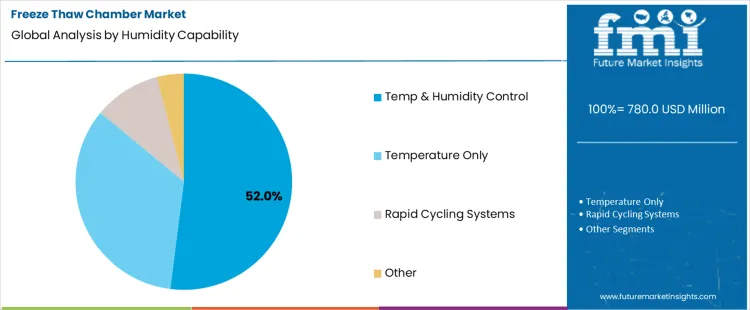
Temperature and humidity control systems hold 52.0%, representing the largest share among capability categories due to broader test coverage. Combined control enables simulation of realistic environmental conditions affecting moisture-sensitive materials. Integrated humidity management supports condensation assessment and material interaction analysis. Precision control improves repeatability across complex protocols. Temperature-only systems address basic cycling with narrower scope. Rapid cycling systems prioritize speed for specific applications. Capability segmentation reflects preference for comprehensive environmental control where test realism and data robustness are required.
Key Points
Demand for freeze thaw chambers reflects controlled testing requirements to evaluate material stability under repeated temperature cycling. Adoption concentrates in pharmaceutical stability testing, biologics handling, food quality assurance, packaging validation, and construction materials testing. Global scope aligns with protocol-driven validation and quality assurance needs. Usage centers on programmable chambers capable of precise temperature ramping, uniform exposure, and repeatable cycling profiles.
Pharmaceutical and biologics developers require freeze thaw testing to assess degradation risk during storage and distribution. Demand increases as protein therapeutics, vaccines, and injectables exhibit sensitivity to phase change stress. Food and beverage processors rely on cycling tests to validate texture, emulsion stability, and shelf integrity. Packaging manufacturers adopt chambers to evaluate seal integrity and material fatigue under temperature fluctuation. Construction and materials laboratories use freeze thaw cycles to assess durability of concrete, composites, and coatings. Programmable control supports protocol alignment and batch repeatability. Data logging enables audit readiness and comparability across test runs. Adoption reflects necessity to quantify stability risk before commercialization and shipment.
Freeze thaw chambers involve high capital cost due to refrigeration systems, insulation, and control accuracy requirements. Demand sensitivity rises where laboratory budgets limit acquisition of multi-chamber setups. Throughput constraints emerge when long cycle durations restrict sample capacity. Energy consumption remains significant during rapid temperature transitions. Uniformity challenges affect result consistency in densely loaded chambers. Maintenance exposure increases due to compressor cycling and defrost requirements. Validation and calibration obligations extend deployment timelines. Space and power infrastructure constraints limit installation in smaller laboratories, constraining scalable expansion across decentralized testing sites.
Demand for freeze thaw chambers is expanding globally due to rising stability testing requirements, cold chain validation, and material durability assessment across regulated industries. Pharmaceutical, biotechnology, food, and construction materials testing drive adoption where controlled temperature cycling is mandatory. Laboratories require repeatable freeze thaw profiles to validate product integrity under transport and storage stress. Growth rates in India at 9.9%, Saudi Arabia at 9.2%, UAE at 8.8%, Indonesia at 8.1%, and Vietnam at 7.8% indicate sustained expansion driven by laboratory capacity growth, regulatory enforcement, and infrastructure development rather than discretionary testing upgrades.
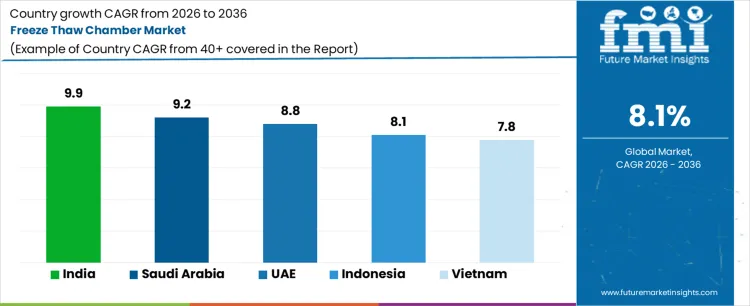
| Country | CAGR (%) |
|---|---|
| India | 9.9% |
| Saudi Arabia | 9.2% |
| UAE | 8.8% |
| Indonesia | 8.1% |
| Vietnam | 7.8% |
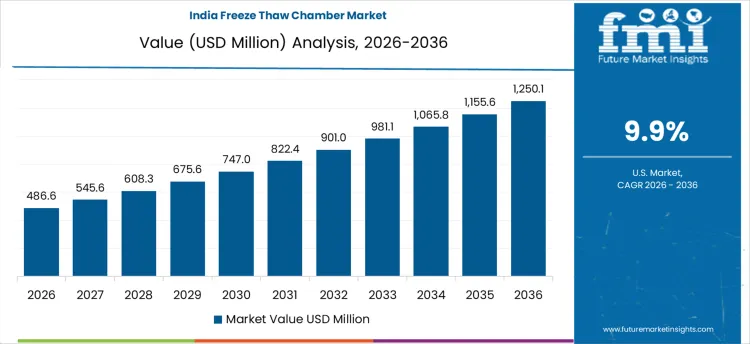
Freeze thaw chamber demand in India is growing at a CAGR of 9.9%, supported by pharmaceutical manufacturing scale-up and stability testing mandates. Injectable drugs, vaccines, and biologics require validated freeze thaw studies to meet regulatory expectations. Cold chain logistics expansion increases need for simulation of temperature excursions. Infrastructure materials testing for roads and concrete adds incremental demand. Contract research laboratories expand environmental testing services. Demand growth reflects compliance-driven testing volume expansion and infrastructure stress validation rather than experimental research usage.
Saudi Arabia is expanding at a CAGR of 9.2%, driven by infrastructure investment and pharmaceutical capacity development. Construction materials testing requires freeze thaw validation for imported and locally produced products. Healthcare expansion increases demand for pharmaceutical stability testing. Research institutions and quality laboratories invest in controlled environmental testing equipment. Harsh climate conditions heighten focus on material performance under thermal stress. Demand growth reflects infrastructure quality assurance and regulated testing requirements rather than academic research intensity.
Freeze thaw chamber demand in United Arab Emirates is growing at a CAGR of 8.8%, influenced by logistics, pharmaceutical, and construction quality testing. Cold chain logistics hubs require validation of packaging and product resilience. Pharmaceutical distributors and manufacturers adopt freeze thaw testing to meet international standards. Construction materials testing laboratories expand capabilities to support large-scale projects. Demand growth reflects quality assurance emphasis and regional logistics importance rather than domestic manufacturing scale alone.
Indonesia is expanding at a CAGR of 8.1%, shaped by food, pharmaceutical, and infrastructure testing needs. Processed food and beverage producers validate packaging durability under temperature cycling. Pharmaceutical manufacturers increase stability testing to meet regulatory standards. Infrastructure development drives material testing for durability under environmental stress. Public and private laboratories invest in environmental simulation equipment. Demand growth reflects testing volume increases and industrial expansion rather than transition to advanced automated testing platforms.
Freeze thaw chamber demand in Vietnam is growing at a CAGR of 7.8%, supported by pharmaceutical manufacturing expansion and export-oriented quality testing. Drug manufacturers require freeze thaw studies to support international regulatory submissions. Food exporters validate packaging integrity for cold storage transport. Industrial testing laboratories expand service offerings to support manufacturing clients. Demand growth reflects export compliance and laboratory capacity development rather than replacement of advanced installed testing systems.
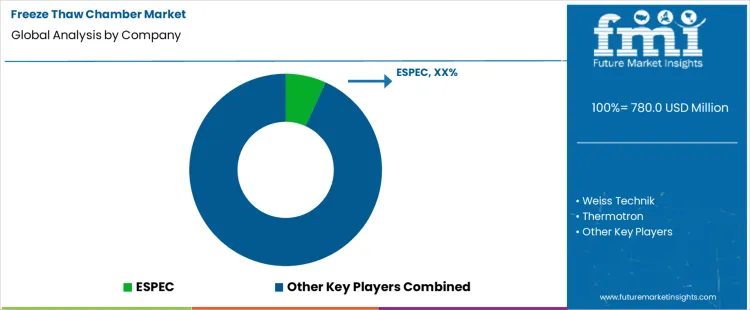
Demand for freeze thaw chambers is driven by climate simulation needs in automotive, aerospace, electronics, pharmaceuticals, and materials testing to assess durability under rapid temperature cycling. These environmental test chambers enable controlled freeze-thaw cycles to evaluate product performance under extreme thermal stress and simulate seasonal or operational conditions. Buyers evaluate temperature range, uniformity, cycle rate precision, control system responsiveness, safety and alarm features, and compliance with industry standards such as ASTM and IEC. Procurement teams prioritize suppliers with proven reliability, calibration and validation support, global service networks, and capacity to integrate chambers within automated test labs. Trend in the global market reflects tightening product quality requirements, accelerated product development cycles, and adoption of digital control platforms that support data logging and remote monitoring.
ESPEC leads the market through comprehensive freeze thaw chamber portfolios and strong global after-sales support tailored to demanding industrial and laboratory environments. Weiss Technik supports demand with engineered environmental chambers offering precise temperature control, robust construction, and integration with thermal shock and climatic test systems. Thermotron contributes high-performance freeze thaw solutions with advanced control systems favored by laboratories testing complex assemblies and components. Darwin Chambers participates with freeze thaw and climatic chambers designed for rigorous cycling and reliability in engineering and quality assurance workflows. BINDER supplies environmental test chambers including freeze thaw models with intuitive interfaces and uniform temperature distribution. Competitive differentiation depends on temperature control fidelity, chamber reliability under frequent cycling, ease of integration with data systems, service coverage, and ability to meet diverse industry standard protocols.
| Items | Values |
|---|---|
| Quantitative Units | USD million |
| Chamber Type | Reach-In Chambers; Walk-In Chambers; Compact Chambers; Other |
| End Use | Pharma Stability & Excursion Testing; Construction Testing; Food & Beverage QA; Packaging & Others |
| Humidity Capability | Temp & Humidity Control; Temperature Only; Rapid Cycling Systems; Other |
| Sales Channel | Direct OEM Sales; Lab & Test Equipment Distributors; Engineering Firms; Other |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, Saudi Arabia, UAE, Indonesia, Vietnam, and 40+ countries |
| Key Companies Profiled | ESPEC; Weiss Technik; Thermotron; Darwin Chambers; BINDER; Memmert; CTS; HAMCO; Newtronic |
| Additional Attributes | Dollar sales by chamber type and end use; adoption trends for rapid cycling and combined temperature-humidity systems supporting stability and excursion protocols; temperature uniformity, ramp rate, recovery time, and control accuracy performance metrics; suitability for pharma stability, construction materials, and food QA testing; refrigeration architecture, energy efficiency, and reliability considerations; validation documentation, data logging, and audit trail requirements; procurement dynamics across direct OEM sales, distributors, and engineering firms; compliance with laboratory qualification standards and test method requirements influencing freeze thaw chamber selection. |
How big is the freeze thaw chamber market in 2026?
The global freeze thaw chamber market is estimated to be valued at USD 780.0 million in 2026.
What will be the size of freeze thaw chamber market in 2036?
The market size for the freeze thaw chamber market is projected to reach USD 1,699.6 million by 2036.
How much will be the freeze thaw chamber market growth between 2026 and 2036?
The freeze thaw chamber market is expected to grow at a 8.1% CAGR between 2026 and 2036.
What are the key product types in the freeze thaw chamber market?
The key product types in freeze thaw chamber market are reach-in chambers, walk-in chambers, compact chambers and other.
Which end use segment to contribute significant share in the freeze thaw chamber market in 2026?
In terms of end use, pharma stability & excursion testing segment to command 34.0% share in the freeze thaw chamber market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.