The single-use biopsy devices market is set to record around USD 2.0 billion and is projected to reach USD 4.7 billion by 2036 at a 9% CAGR. Device choice revolves around patient safety, sterility assurance, and sampling precision rather than brand visibility. Once a procedure standardizes a specific device type, workflows are locked to maintain consistent results and reduce training overhead. Clinicians consider needle gauge, handle ergonomics, and tissue retention performance. Supply planning accounts for shelf life, packaging integrity, and refill timing. Quality teams track lot traceability, regulatory compliance, and procedural success rates.
Commercial growth is influenced by procedural adoption, case complexity, and hospital expansion rather than promotional campaigns. Manufacturing focuses on needle sharpness, material uniformity, and dimensional tolerances to ensure consistent biopsy quality. Release requires sterility verification, functional testing, and documentation checks. Distributors position devices near hospitals with high biopsy volume. Technical support provides guidance on handling, troubleshooting, and staff training. Profitability depends on consistent yield, low failure rates, and predictable reorder cycles rather than aggressive pricing or frequent design changes. Inventory strategy aligns with scheduled procedure volume to maintain uninterrupted availability.

Clinical workflow integration and procedural safety guidelines are the primary drivers of growth in the single-use biopsy devices market rather than overall procedure count. In 2026, at around USD 2 billion, hospitals and outpatient diagnostic centers prioritize devices that minimize infection risk, reduce reprocessing requirements, and streamline sample collection. Expansion is shaped by the increasing shift toward outpatient and minimally invasive procedures, higher patient throughput, and adoption of image-guided techniques. Device versatility, ergonomic handling, and compatibility with different biopsy systems determine purchasing decisions. Growth is incremental and tied to standardization of disposable instruments across multiple care settings rather than single-procedure spikes.
Strategic procurement planning and operational efficiency influence the later stage of the market. As the market approaches roughly USD 4.7 billion by 2036, decision makers focus on supplier responsiveness, product availability for high-volume cycles, and consistency in clinical performance across batches. Hospitals seek single-use devices that integrate seamlessly with existing equipment while reducing staff training time and handling errors. Manufacturers that offer predictive production capacity, rapid replenishment models, and adaptable product portfolios benefit from stronger adoption across regional hospital networks. The increase from USD 2 billion to over USD 4.7 billion results from expansion into new outpatient facilities and multi-site institutional programs rather than sudden market spikes.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 2.0 billion |
| Forecast Value (2036) | USD 4.7 billion |
| Forecast CAGR 2026 to 2036 | 9.0% |
Single-use biopsy devices are designed for one-time tissue collection, eliminating the need for sterilization and reducing the risk of cross-contamination in clinical and diagnostic settings. Unlike reusable systems, they combine preloaded needles, integrated collection chambers, and ergonomic handling to simplify procedures and improve sample integrity. Hospitals, outpatient centers, and pathology laboratories evaluate these devices based on puncture precision, consistent sample volume, ease of handling, and compatibility with imaging guidance. Adoption is concentrated in high-volume oncology and interventional radiology units, where standardizing devices reduces procedure variability and streamlines workflow. Device reliability also affects specimen adequacy, laboratory processing efficiency, and patient throughput.
Manufacturers differentiate products through needle design, material quality, and collection efficiency rather than price alone. Earlier biopsy practices relied on reusable tools, which required extensive sterilization cycles and carried variable performance. Today, production emphasizes tight quality control, uniform tolerances, and safe disposal mechanisms. Hospitals and laboratories assess total operational value through minimized sample errors, reduced procedure time, and lower infection risk rather than cost per device. Suppliers provide value-added services such as staff training, lot traceability, and rapid replenishment to maintain uninterrupted operations. Market growth follows increased diagnostic volumes, expansion of outpatient interventional programs, and the drive for safer, reproducible tissue sampling, creating opportunities for manufacturers to standardize clinical protocols and improve procedural efficiency.
Clinical safety, procedural efficiency, and infection control are the primary drivers of the Single-use Biopsy Devices Market in 2026 rather than short-term purchasing trends. Hospitals and diagnostic centers select devices that reduce cross-contamination risk, maintain sample integrity, and integrate with imaging guidance systems. Segment adoption varies according to needle design, deployment mechanism, and procedural complexity. Suppliers providing consistent consumable supply, technical training, and device support can secure long-term adoption. Market growth is concentrated in oncology and pulmonology procedures, where high procedural volumes and standardized workflows create predictable demand patterns for manufacturers capable of delivering reliable devices and comprehensive service programs.
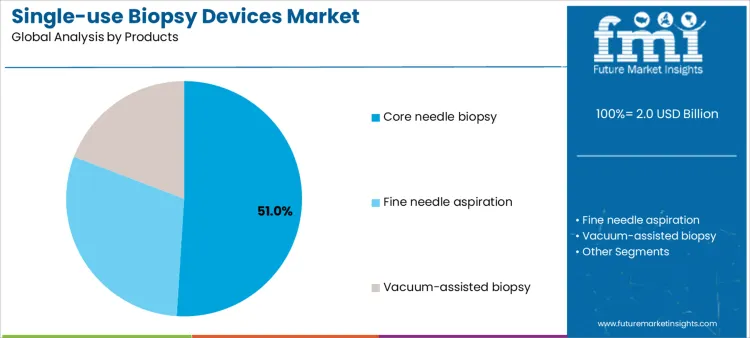
Core needle biopsy devices account for approximately 51% of demand due to their precision and reliability in oncology tissue sampling. Suppliers supporting these devices must ensure batch quality, operator training, and consistent consumable delivery. Fine needle aspiration devices are widely applied in pulmonology procedures, where ease of use and minimal invasiveness are critical, requiring supplier engagement in procedural guidance and staff education. Vacuum-assisted biopsy devices serve specialized indications, including high-volume breast imaging programs, increasing supplier involvement in calibration, technical support, and workflow integration. Device type directly dictates supplier responsibilities in training, consumable management, and technical service for successful adoption across diverse clinical settings.
Clinical workflow and operational impact differ depending on device type. Core needle biopsies require precise handling, integration with imaging systems, and adherence to strict procedural protocols, demanding ongoing supplier support. Fine needle aspiration emphasizes rapid deployment and minimal patient risk, with training and inventory management as key supplier contributions. Vacuum-assisted devices necessitate advanced technical guidance, equipment setup, and post-procedural support. Hospitals standardizing on specific device types rely on suppliers for consistent product performance, training, and consumable supply. Providers offering modular support, preventive guidance, and technical services can maintain adoption and improve clinical efficiency across all procedure types.
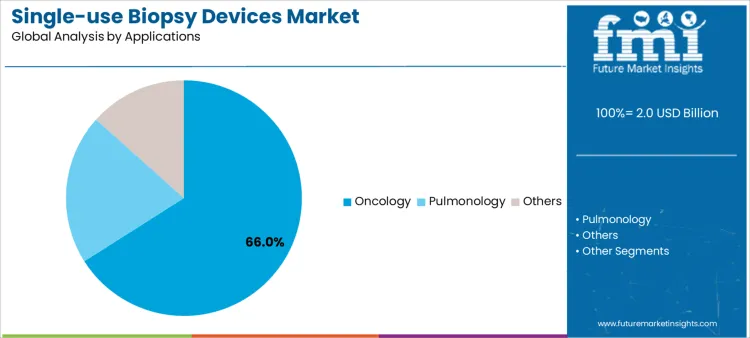
Oncology procedures represent approximately 66% of market demand due to high-volume tissue sampling and the critical need for accurate diagnostics. Pulmonology applications, while lower in volume, require specialized needles and guidance systems, presenting opportunities for suppliers offering technical support and consumable reliability. Other applications, such as interventional radiology or rare tissue sampling, create smaller but technically demanding markets. Application-specific demand informs inventory allocation, training programs, and service coverage. Aligning product offerings with high-volume oncology and specialized pulmonology procedures enables suppliers to maximize adoption, procedural efficiency, and long-term revenue streams.
Variation in clinical application also drives supplier engagement and resource planning. Oncology-focused centers prioritize consistent device supply, staff training, and procedural standardization to reduce variability and optimize outcomes. Pulmonology programs demand flexible device options, guidance on imaging integration, and ongoing technical support. Niche or low-volume applications require specialized training, high-quality device performance, and rapid consumable availability. Segment distribution depends on procedure frequency, clinical complexity, and workflow integration rather than short-term procurement fluctuations. Suppliers providing training, reliable supply, and responsive support can capture higher market share and strengthen long-term partnerships.
The Single-Use Biopsy Devices Market is shaped by hospitals and diagnostic centers prioritizing sterility, reduced infection risk, and consistent procedural outcomes. Devices are evaluated based on ease of use, tissue sample quality, compatibility with imaging or guidance systems, and disposal convenience. Selection decisions involve pathologists, interventional radiologists, and procurement teams. Suppliers that provide reliable, pre-sterilized devices with clear handling instructions are preferred. This positions single-use biopsy devices as essential clinical tools rather than optional consumables, emphasizing safety, workflow efficiency, and predictable performance.
Healthcare providers increasingly adopt standardized biopsy protocols to improve efficiency and reduce patient complications. This drives demand in the Single-Use Biopsy Devices Market toward devices that deliver consistent tissue samples, integrate with standard procedural workflows, and minimize handling errors. Buyers prefer products that are ready-to-use, compatible with existing devices, and require minimal training. Suppliers are evaluated on quality consistency, regulatory compliance, and technical support rather than on minor design variations. Devices aligned with standardized protocols are more likely to be adopted across multiple hospital units.
Recurring demand comes from hospitals, outpatient diagnostic centers, and specialty clinics performing high volumes of biopsies. Each procedure consumes a single-use device, creating predictable repeat orders. The Single-Use Biopsy Devices Market also benefits when multi-site hospital networks standardize on a single supplier or device model. Suppliers that provide training, consistent product supply, and responsive technical support secure long-term partnerships. Embedding devices into procedural protocols ensures ongoing demand tied to patient volumes and repeat procedures.
Single-use biopsy devices have higher per-procedure costs compared with reusable systems, which can limit adoption in low-volume or cost-sensitive facilities. The market also faces adoption constraints from regulatory compliance, staff training requirements, and workflow adjustments. Clinicians are cautious about changing device types mid-procedure due to sample quality and patient safety concerns. Suppliers entering the market must provide clinical support, documentation, and reliable product supply. Adoption is concentrated in high-volume diagnostic centers and specialized hospitals where procedural efficiency and safety are critical.
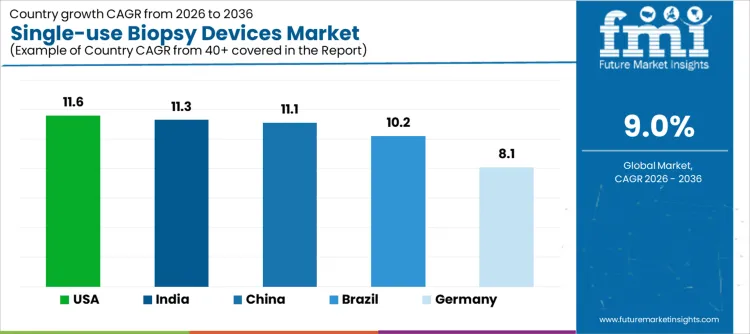
| Country | CAGR |
|---|---|
| USA | 11.6% |
| Brazil | 10.2% |
| China | 11.1% |
| India | 11.3% |
| Germany | 8.1% |
Demand for single use biopsy devices is rising as hospitals, diagnostic centers, and oncology clinics prioritize infection control, procedural efficiency, and patient safety during tissue sampling. India leads with an 11.3% CAGR, supported by expanding cancer care infrastructure, higher diagnostic procedure volumes, and adoption of disposable, pre sterilized biopsy systems. China follows at 11.1%, driven by rising hospital procedures, regulatory focus on safety, and modernization of diagnostic workflows. Brazil records 10.2%, reflecting broader access in public and private healthcare facilities. The USA grows at 11.6%, shaped by replacement of reusable devices and adoption of streamlined single use solutions. Germany, at 8.1%, reflects stable demand in a mature healthcare system with established clinical protocols.
In United States, single-use biopsy device demand is growing at an 11.6% CAGR as hospitals and outpatient clinics expand interventional oncology and tissue sampling procedures. Procurement decisions are controlled by interventional radiology directors, surgical committees, and supply chain teams. Adoption emphasizes device sterility, cutting efficiency, and compatibility with imaging guidance systems. Once approved, devices are standardized across multiple hospital networks and outpatient centers. Training programs and protocol alignment influence utilization. Import systems dominate complex or high-acuity procedures, while domestic suppliers provide standard single-use kits. Replacement cycles follow procedure volume rather than device aging. Capital allocation aligns with facility expansion and procedure growth. Commercial opportunities exist for suppliers providing comprehensive technical support, training programs, and multi-year supply agreements.
Brazil is experiencing a 10.2% CAGR for single-use biopsy devices as private hospital chains and specialty oncology centers expand minimally invasive procedures. Procurement authority resides with network purchasing offices, radiology committees, and clinical service directors. Adoption focuses on sterile, high-quality devices for needle biopsies, core biopsies, and vacuum-assisted procedures. Import products dominate premium applications, while local suppliers cover standard procedures. Once validated, rollout spans multiple affiliated hospitals and outpatient clinics. Training, workflow integration, and clinical protocols influence adoption. Capital allocation follows network expansion and procedure volume growth. Replacement cycles track usage intensity. Commercial success depends on inclusion in network-approved supply catalogs, reference site adoption, and reliable logistics rather than ad hoc hospital or clinic purchases.
China is experiencing an 11.1% CAGR in single-use biopsy devices due to expansion of tertiary hospitals, oncology units, and outpatient diagnostic programs. Large hospital networks and procedural committees control most procurement decisions. Adoption emphasizes accuracy, sterility, and workflow efficiency in ultrasound-guided, CT-guided, and mammography biopsy procedures. Domestic suppliers cover routine single-use devices while international brands retain positions in high-precision applications. Tender systems and multi-year capital planning govern procurement timing. Once approved, devices are deployed across multiple departments and affiliated hospitals. Training programs, clinical protocol adherence, and workflow integration influence adoption. Replacement cycles follow procedural load rather than device lifespan. Capital allocation aligns with hospital expansion and new oncology programs. Commercial success depends on inclusion in provincial and national hospital catalogs rather than small independent clinic sales.
India is witnessing an 11.3% CAGR in single-use biopsy devices due to growth of hospital oncology programs, specialized radiology centers, and minimally invasive procedure adoption. Procurement authority rests with hospital committees, interventional radiology directors, and pharmacy boards. Device selection emphasizes sterility, precision, imaging compatibility, and ease of use. Import devices dominate premium applications, while domestic suppliers cover routine procedures. Once approved, devices are deployed across multiple affiliated hospital networks. Training, protocol adherence, and procedural standardization influence utilization. Replacement cycles track procedure volumes. Capital allocation aligns with hospital expansion and new service line openings. Commercial success depends on inclusion in network approved supply lists, reference site adoption, and multi-year support contracts rather than isolated clinic or department purchases.
Germany is experiencing an 8.1% CAGR in single-use biopsy devices due to regulatory oversight, structured clinical guidelines, and hospital standardization of interventional oncology protocols. University hospitals and regional medical centers control most procurement decisions. Adoption focuses on sterile, precision biopsy devices compatible with imaging and workflow systems. Evaluation emphasizes safety, reliability, and operator ergonomics. Procurement follows health technology assessment and hospital capital planning processes. Import suppliers dominate premium applications, while domestic suppliers cover standard devices. Once approved, devices are deployed across multiple departments and hospitals. Training, protocol adherence, and workflow integration influence utilization. Replacement cycles follow procedural load. Capital allocation aligns with hospital program expansion. Commercial success depends on framework inclusion, reference site adoption, and integrated supplier support rather than isolated hospital or clinic purchases.

Single-use biopsy device selection occurs within procedural workflow planning, sterility protocols, and pathology requirements rather than catalog comparison. Clinicians define needle gauge, tissue type, suction control, and insertion mechanism before approving suppliers. BD, Hologic, Cook Medical, Argon Medical, Merit Medical compete during evaluation and integration stages. Once a device is included in standard operating procedures, staff training, inventory management, and disposal protocols anchor its adoption. BD leverages hospital supply networks and procedural standardization. Hologic positions products through diagnostic platform integration and workflow compatibility. Cook Medical emphasizes versatility across organ systems and procedure types. Argon Medical targets rapid deployment and consistent performance. Merit Medical supports customization for specialty procedures and high volume interventional programs.
Competitive outcomes reflect procedural reliability, clinical confidence, and regulatory compliance rather than price alone. Clinicians assess deployment accuracy, tissue yield, tract closure performance, and handling ergonomics during trials. Central sterile teams review packaging, labeling, and disposal safety. Procurement prioritizes suppliers with predictable supply and batch traceability. BD benefits from installed base familiarity and broad procedural coverage. Hologic secures placements through integration with imaging and pathology workflows. Cook Medical gains share in programs requiring cross organ applicability. Argon Medical competes where rapid setup and ease of use are critical. Merit Medical holds positions in high volume centers with multiple procedure types. Market positions vary by hospital protocol, procedure volume, and departmental adoption policies.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD billion |
| Product Type | Core needle biopsy; Fine needle aspiration; Vacuum-assisted biopsy |
| Application | Oncology; Pulmonology; Others |
| End User / Sales Channel | Hospitals; Diagnostic labs; ASCs |
| Region | Asia Pacific; Europe; North America; Latin America; Middle East & Africa |
| Countries Covered | United States, Canada, Mexico, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Brazil, Chile, Saudi Arabia, Turkey, South Africa, Rest of regions |
| Key Companies Profiled | BD; Hologic; Cook Medical; Argon Medical; Merit Medical |
| Additional Attributes | Dollar by sales by product and application, oncology versus pulmonology case mix, core versus FNA versus vacuum mix, sterility driven replacement cycles, shelf life and packaging constraints, procedure volume linkage, training and handling requirements, inventory turnover patterns, regulatory and disposal compliance needs |
How big is the single-use biopsy devices market in 2026?
The global single-use biopsy devices market is estimated to be valued at USD 2.0 billion in 2026.
What will be the size of single-use biopsy devices market in 2036?
The market size for the single-use biopsy devices market is projected to reach USD 4.7 billion by 2036.
How much will be the single-use biopsy devices market growth between 2026 and 2036?
The single-use biopsy devices market is expected to grow at a 9.0% CAGR between 2026 and 2036.
What are the key product types in the single-use biopsy devices market?
The key product types in single-use biopsy devices market are core needle biopsy, fine needle aspiration and vacuum-assisted biopsy.
Which applications segment to contribute significant share in the single-use biopsy devices market in 2026?
In terms of applications, oncology segment to command 66.0% share in the single-use biopsy devices market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.