Pharmaceutical Sterility Testing Market
Pharmaceutical Sterility Testing Market Size and Share Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 to 2023 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2026 to 2035
Pharmaceutical Sterility Testing Market Size and Share Forecast Outlook 2025 to 2035
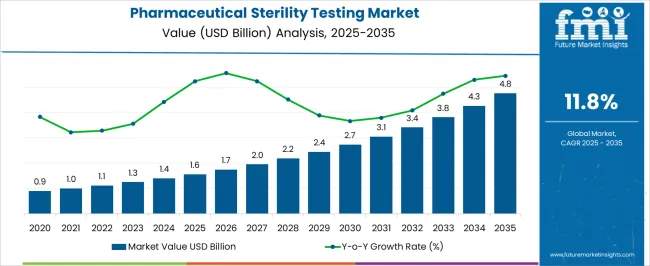
The Pharmaceutical Sterility Testing Market is estimated to be valued at USD 1.6 billion in 2025 and is projected to reach USD 4.8 billion by 2035, registering a compound annual growth rate (CAGR) of 11.8% over the forecast period.
Quick Stats for Pharmaceutical Sterility Testing Market
- Pharmaceutical Sterility Testing Market Industry Value (2025): USD 1.6 billion
- Pharmaceutical Sterility Testing Market Forecast Value (2035): USD 4.8 billion
- Pharmaceutical Sterility Testing Market Forecast CAGR: 11.8%
- Leading Segment in Pharmaceutical Sterility Testing Market in 2025: Kits And Reagents (54.2%)
- Key Growth Region in Pharmaceutical Sterility Testing Market: North America, Asia-Pacific, Europe
- Top Key Players in Pharmaceutical Sterility Testing Market: Pacific Bio labs, STERIS, Boston Analytical, Nelson Laboratories, LLC, Sartorius AG, SOLVIAS AG, SGS SA, Laboratory Corporation of America Holding, Pace Analytical, Charles River Laboratories, Thermo Fisher Scientific, Inc.
| Metric | Value |
|---|---|
| Pharmaceutical Sterility Testing Market Estimated Value in (2025 E) | USD 1.6 billion |
| Pharmaceutical Sterility Testing Market Forecast Value in (2035 F) | USD 4.8 billion |
| Forecast CAGR (2025 to 2035) | 11.8% |
Rationale for Segmental Growth in the Pharmaceutical Sterility Testing Market
The pharmaceutical sterility testing market is expanding steadily as regulatory bodies place greater emphasis on ensuring the safety and efficacy of drugs and biologics. Increasing production of sterile pharmaceuticals, injectable formulations, and biologic therapies has heightened the demand for rigorous sterility validation procedures.
The rising complexity of drug development and the growing prevalence of chronic conditions are driving manufacturers to adopt advanced testing solutions that guarantee product integrity. The integration of rapid microbiological methods and automation in sterility testing is further enhancing accuracy, reducing turnaround time, and improving compliance with international standards.
With biopharmaceutical innovation and stringent global regulatory frameworks shaping industry practices, the market outlook remains favorable, supported by the critical need to safeguard patient health and maintain product quality across diverse therapeutic categories.
Segmental Analysis
Insights into the Kits and Reagents Product Segment
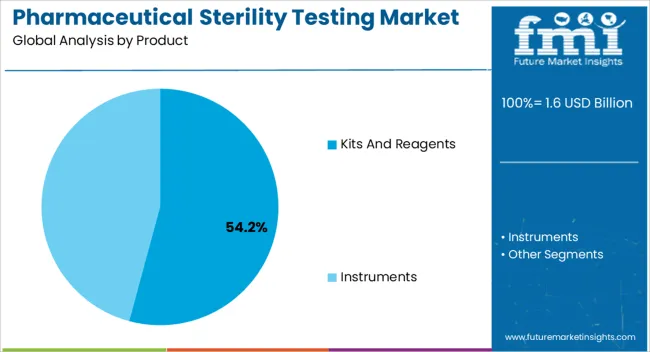
The kits and reagents segment is projected to hold 54.20% of total market revenue by 2025 within the product category, making it the leading contributor. Growth in this segment is supported by the wide availability of ready-to-use kits that streamline sterility testing workflows, reduce preparation errors, and enhance efficiency.
The use of high-quality reagents ensures consistency, reliability, and compliance with pharmacopeial standards, making them indispensable for routine testing in pharmaceutical and biotechnology industries.
Continuous advancements in reagent formulations and kit design have further improved sensitivity and reproducibility, driving adoption across both small and large-scale manufacturing facilities.
Insights into the Sterile Drugs Application Segment
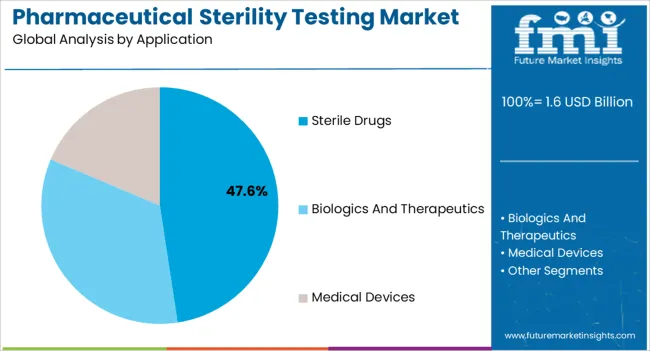
The sterile drugs segment is expected to account for 47.60% of total market revenue by 2025 within the application category, establishing it as the dominant area. This prominence is attributed to the growing demand for injectables, ophthalmic solutions, and other sterile dosage forms where patient safety is paramount.
Strict regulatory requirements governing the manufacture of sterile pharmaceuticals have intensified the need for robust sterility testing protocols. Increasing investments in biologics and biosimilars are further fueling the demand for advanced testing approaches that can ensure safety and quality.
The reliance on sterility validation as a core compliance requirement continues to position sterile drugs as the largest application segment.
Insights into the Sterility Testing Test Type Segment
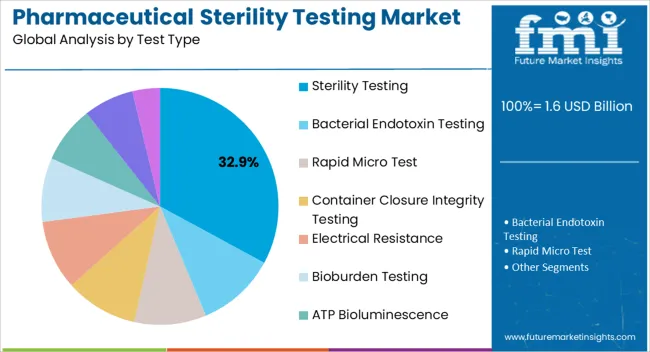
The sterility testing segment is projected to represent 32.90% of market revenue by 2025 within the test type category, making it a key growth driver. The necessity of confirming the absence of viable microorganisms in final products has cemented the importance of sterility testing across pharmaceutical production lines.
Regulatory mandates have reinforced its adoption as a standard quality control practice. Furthermore, the shift toward rapid microbiological methods and automated sterility testing systems is improving accuracy and reducing the risk of false positives or contamination during testing.
These advancements are strengthening sterility testing’s role as a critical component of pharmaceutical quality assurance.
2020 to 2025 Sterility Testing Adoption Vs Forecast Outlook 2025 to 2035
The background of the market is quite promising, and numbers are likely to increase during the research period. A number of developing nations have sped up the launch of new medicines in order to meet the world's rising need for healthcare. By doing this, the scope for CAGR increases from 9.8% (2020 to 2025) to 11.8% by 2035. As a result of the rising prevalence of chronic and infectious diseases, there is an increasing demand for pharmaceuticals on a global scale. For instance,
- The WHO (World Health Organization) announced in February 2024 that cardiovascular diseases remain one of the leading causes of mortality worldwide, killing 17.9 million people a year.
Future illness load is anticipated to increase demand for pharmaceuticals, supporting the need for pharmaceutical sterility testing. Due to this, the global market had the potential to thrive. In 2020, the market was valued at USD 1.6 million, and by 2025, it reached USD 1,124.7 million.
The regulatory changes have significantly improved the likelihood of widespread adoption. Drugs like Elagolix and Lanadelumab, whose debut is considerably assisting the market's growth, are biopharmaceutical products that require special sterility testing. Given all of these influences, the market is probably anticipating a 1.1x growth between 2025 and 2025.
How Sterility Testing Brings Profitability to Pharma Pipelines
- The need for various types of sterility testing is driving the growth of the entire industry. These sterility testing types include sterility testing medical devices, b/f testing sterility, hypothesis testing, sgs sterility testing, VHP(Vaporized Hydrogen Peroxide) Ingress Testing, etc.,
- High levels of consolidation, strict regulatory environments, and drawn-out licensing procedures all pose serious obstacles for new market entrants. However, this can be resolved as many businesses enter the kit testing market, which offers a consistent flow of client revenue.
Regional Analysis of Pharmaceutical Sterility Testing Market

In 2025, it is anticipated that North America accounted for 39% of the global pharmaceutical sterility testing market. The existence of numerous international and domestic competitors in the area is responsible for the region's rise.
During the forecast period, a huge amount of money is likely to be spent on research and development to support the growth of North America market. The biopharmaceutical business has a substantial total economic impact on the economy of America.
What Benefits the Key Players in the United States Market?
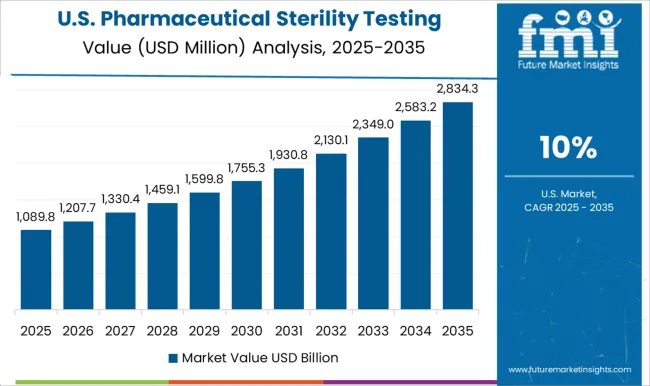
The United States held a dominant market share of 41.5% in 2025. This is due to the factors such as:
- Presence of numerous international pharmaceutical and biopharmaceutical businesses.
- Significant financing from both the public and commercial sectors in the nation.
For instance, PreHevbrio was introduced by VBI Vaccines Inc. in March 2025 for the treatment of persons aged 18 years old. It is for those who are at risk of contracting any of the recognized subtypes of the hepatitis B virus (HBV). In November 2024, the FDA authorized this vaccine as the exclusive adult 3-antigen HBV vaccine. Consequently, Canada spent USD 23,400 million internally on pharmaceutical research and development in 2024, and it reached USD 23,800 million in 2025, a 1.7% increase.
Pharmaceutical Sterility Testing in China Market Focus on Compiling with the Standards
By 2035, China is anticipated to hold a significant rank in the pharmaceutical sterility testing sector, with a CAGR of 12.1%. This is a result of emerging countries increasingly harmonizing their regulatory norms with ICH standards. Additionally, the region's emerging economies are taking a variety of steps to expand their domestic pharmaceutical market and are enticing multinational corporations to establish operations there.
Contract research organizations (CROs) are anticipated to expand significantly over the next few years. Sterile testing services are significantly being outsourced to CROs because of the benefits they provide including flexibility, cost-effectiveness, and access to specialist knowledge. The primary end-users of sterility testing services in the nation are anticipated to be the biopharmaceutical industry and CROs.
Research and Development Creates a Sizable Market in India
India is prepared to manufacture medical devices and conduct extensive sterility testing due to substantial pharmaceutical investment. By 2035, India is projected to have a 12.8% CAGR.
According to the Union Budget for the years 2024 to 2025, the Indian Ministry of Science and Technology allocated USD 1.4 million, a 25% increase from the budget for the years 2024 to 2024, for the DBT (Department of Biotechnology). This is primarily for biotechnology research and development, as well as industrial and entrepreneurship development in the nation. This budget sets new goals for India pharmaceutical sterility testing market. In accordance with the analysis, the Indian biotechnology market was estimated to be worth USD 70,200 million in 2024; by 2025, it is anticipated to be worth USD 150,000 million.
Automated Techniques Lead the Market in Germany
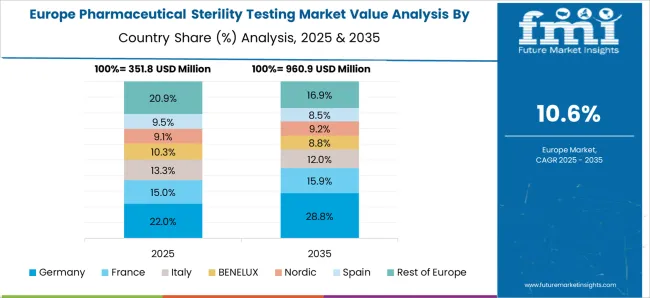
In Germany, automated methods rule the market. The market was initially growing, with a share of 6.2% in 2025, and by 2035, it is projected to have a wide scope. Automated systems that contribute effectively to the global industry include the BacT/ALERT system. These techniques are made to be highly sensitive and specific in identifying the presence of microorganisms in pharmaceutical items.
Germany has a stringent regulatory framework that is overseen by the European Pharmacopoeia (Ph. Eur.) and the German Medicines Act (AMG). All pharmaceutical products that are intended for infusion, implantation, or injection into the human body must pass sterility testing. This testing is essential to make sure the product is devoid of living microorganisms like bacteria, viruses, and fungi that possibly infect people or create other adverse effects.
Category-Wise Analysis of Sterility Testing Techniques
| Category | Product Type |
|---|---|
| Top-segment | Kits & Reagents |
| Market Share (2025) | 39.7% |
| Category | Services |
|---|---|
| Top-segment | Outsourced |
| Market Share (2025) | 54.1% |
| Category | Test type |
|---|---|
| Top-segment | Bacterial endotoxin testing |
| Market Share (2025) | 38.4% |
Which Services are Popular in the Pharmaceutical Sterility Testing Industry?
- By accounting for 54.1% of the market in 2025, the outsourcing segment had the top share.
- Small and medium-sized manufacturers of pharmaceuticals and medical devices do not always have the resources to undertake reliable sterility testing. Thus, they are likely to rather outsource these services to meet FDA standards.
- Outsourcing helps pharmaceutical and medical device companies save time and money, as they don't have to find and train new employees or buy new equipment.
- Pharmaceutical sterility testing services are gradually being outsourced by large pharmaceutical companies as they attempt to concentrate on their core capabilities, such as marketing. These elements have had a key role in the segment's growth.
Kits and Reagents: Top Segment based on Product Type
Through 2035, the kits and reagents market is predicted to experience significant growth. The availability of a variety of items that aid in reducing the danger of false negative and false positive results is credited with this increase. These also consist of sterility testing pumps, which are easily integrated into any testing environment, streamline workflow, and help to maximize safety.
Over the anticipated period, segmental growth is expected to be boosted by the rising demand for comparable products.
Which Testing Type Gives Accurate Results?
In 2025, the segment for bacterial endotoxin testing held the leading market share with 38.4%. These tests are used to evaluate medical equipment like implants. The market for this category is expanding as a result of rising parenteral medication and implant demand. Along with an increase in the number of drug and medical device launches, there is a growing emphasis on product quality.
Competitive Landscape of the Sterility Testing Market
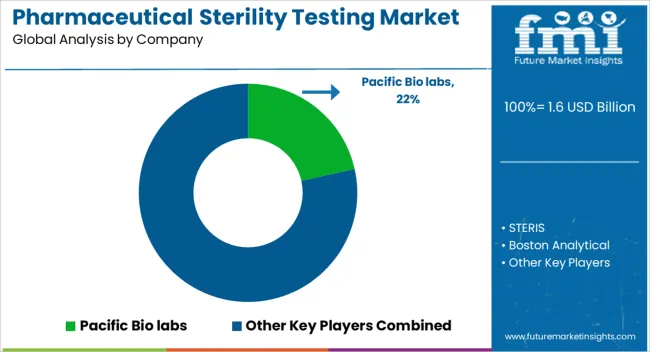
How do Pharmaceutical Sterility Testing Manufacturers Expand in the Market?
The demand for pharmaceutical sterility testing is projected to increase along with medical improvements, and producers can profit from these technological developments.
The growth of the biotechnology sector offers businesses a lot of room for future advancements.
Concerns like the safety of biological pharmaceuticals are surging. New technological advancements in the area of sterility testing have improved the procedure's efficiency, allowing manufacturers to introduce new products in the market.
Regulatory Scrutiny: Bane or Boon for Key Players?
The market is highly fragmented due to the presence of multiple local and MNC contract service providers and research facilities. The main strategies employed by the bulk of these organizations include regional growth and digitalization of service delivery for sterility testing. Companies in the sector are subject to intense regulatory scrutiny and must abide by rules relating to the security of pharmaceutical products. As a result, specialized consulting companies have emerged. These companies offer services related to pharmaceutical sterility testing that are related to regulatory compliance and quality assurance.
Recent Developments:
- In June 2025, Lee, Massachusetts-based fill-finish CMO Berkshire Sterile Manufacturing (BSM) declared that its sterility testing isolator was prepared to conduct on-site sterility testing for its GMP batches. Most of the clients of the business, if not all of them, should see a short release due to the new service.
- The establishment of a newly expanded center of excellence for microbiological laboratory testing and improved sterilizing capacity in Nelson Labs' and Sterigenics Deutschland GmbHs Wiesbaden, Germany, facilities were both announced for September 2024.
- The EndoScan-V software platform for endotoxin testing and the Celsis automated detection system for assessing the fast sterility of pharmaceutical pharmaceuticals were both introduced by Charles River Laboratories in September 2020.
Key Companies Operating in the Pharmaceutical Sterility Testing Market
- Merck KGaA
- bioMerieux SA
- Thermo Fisher Scientific Inc.
- Charles River Laboratories International Inc.
- Others
Key Segments Covered in the Sterility Testing Market Report
By Product:
- Kits and Reagents
- Instruments
By Application:
- Sterile Drugs
- Biologics and Therapeutics
- Medical Devices
By Test Type:
- Sterility Testing
- Membrane Filtration
- Direct Inoculation
- Bacterial Endotoxin Testing
- Rapid Micro Test
- Container Closure Integrity Testing
- Electrical Resistance
- Bioburden Testing
- ATP Bioluminescence
- Fluorescent Labeling
- Others
By Services:
- In-House
- Outsourced
By End Use:
- Compounding Pharmacies
- Medical Devices Companies
- Pharmaceutical Companies
By Region:
- North America
- Latin America
- Europe
- East Asia
- South Asia
- Oceania
- The Middle East & Africa (MEA)
Frequently Asked Questions
How big is the pharmaceutical sterility testing market in 2025?
The global pharmaceutical sterility testing market is estimated to be valued at USD 1.6 billion in 2025.
What will be the size of pharmaceutical sterility testing market in 2035?
The market size for the pharmaceutical sterility testing market is projected to reach USD 4.8 billion by 2035.
How much will be the pharmaceutical sterility testing market growth between 2025 and 2035?
The pharmaceutical sterility testing market is expected to grow at a 11.8% CAGR between 2025 and 2035.
What are the key product types in the pharmaceutical sterility testing market?
The key product types in pharmaceutical sterility testing market are kits and reagents and instruments.
Which application segment to contribute significant share in the pharmaceutical sterility testing market in 2025?
In terms of application, sterile drugs segment to command 47.6% share in the pharmaceutical sterility testing market in 2025.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size Value (USD Million) Analysis, 2020 to 2024
- Current and Future Market Size Value (USD Million) Projections, 2025 to 2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2020 to 2024 and Forecast 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product , 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product , 2025 to 2035
- Kits And Reagents
- Instruments
- Y-o-Y Growth Trend Analysis By Product , 2020 to 2024
- Absolute $ Opportunity Analysis By Product , 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2025 to 2035
- Sterile Drugs
- Biologics And Therapeutics
- Medical Devices
- Y-o-Y Growth Trend Analysis By Application, 2020 to 2024
- Absolute $ Opportunity Analysis By Application, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Test Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Test Type, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Test Type, 2025 to 2035
- Sterility Testing
- Membrane Filtration
- Direct Inoculation
- Bacterial Endotoxin Testing
- Rapid Micro Test
- Container Closure Integrity Testing
- Electrical Resistance
- Bioburden Testing
- ATP Bioluminescence
- Fluorescent Labeling
- Others
- Sterility Testing
- Y-o-Y Growth Trend Analysis By Test Type, 2020 to 2024
- Absolute $ Opportunity Analysis By Test Type, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Services
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Services, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Services, 2025 to 2035
- In-House
- Outsourced
- Y-o-Y Growth Trend Analysis By Services, 2020 to 2024
- Absolute $ Opportunity Analysis By Services, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2025 to 2035
- Compounding Pharmacies
- Medical Devices Companies
- Pharmaceutical Companies
- Y-o-Y Growth Trend Analysis By End Use, 2020 to 2024
- Absolute $ Opportunity Analysis By End Use, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2020 to 2024
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2025 to 2035
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- USA
- Canada
- Mexico
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- Key Takeaways
- Latin America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- Key Takeaways
- Western Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- Key Takeaways
- Eastern Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- Key Takeaways
- East Asia Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- China
- Japan
- South Korea
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- Key Takeaways
- South Asia and Pacific Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- Key Takeaways
- Middle East & Africa Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- Canada
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- Mexico
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- Brazil
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- Chile
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- Germany
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- UK
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- Italy
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- Spain
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- France
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- India
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- China
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- Japan
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- South Korea
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- Russia
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- Poland
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- Hungary
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- South Africa
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Application
- By Test Type
- By Services
- By End Use
- Competition Analysis
- Competition Deep Dive
- Pacific Bio labs
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- STERIS
- Boston Analytical
- Nelson Laboratories, LLC
- Sartorius AG
- SOLVIAS AG
- SGS SA
- Laboratory Corporation of America Holding
- Pace Analytical
- Charles River Laboratories
- Thermo Fisher Scientific, Inc.
- Pacific Bio labs
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2020-2035
- Table 2: Global Market Value (USD Million) Forecast by Product , 2020-2035
- Table 3: Global Market Value (USD Million) Forecast by Application, 2020-2035
- Table 4: Global Market Value (USD Million) Forecast by Test Type, 2020-2035
- Table 5: Global Market Value (USD Million) Forecast by Services, 2020-2035
- Table 6: Global Market Value (USD Million) Forecast by End Use, 2020-2035
- Table 7: North America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 8: North America Market Value (USD Million) Forecast by Product , 2020-2035
- Table 9: North America Market Value (USD Million) Forecast by Application, 2020-2035
- Table 10: North America Market Value (USD Million) Forecast by Test Type, 2020-2035
- Table 11: North America Market Value (USD Million) Forecast by Services, 2020-2035
- Table 12: North America Market Value (USD Million) Forecast by End Use, 2020-2035
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 14: Latin America Market Value (USD Million) Forecast by Product , 2020-2035
- Table 15: Latin America Market Value (USD Million) Forecast by Application, 2020-2035
- Table 16: Latin America Market Value (USD Million) Forecast by Test Type, 2020-2035
- Table 17: Latin America Market Value (USD Million) Forecast by Services, 2020-2035
- Table 18: Latin America Market Value (USD Million) Forecast by End Use, 2020-2035
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 20: Western Europe Market Value (USD Million) Forecast by Product , 2020-2035
- Table 21: Western Europe Market Value (USD Million) Forecast by Application, 2020-2035
- Table 22: Western Europe Market Value (USD Million) Forecast by Test Type, 2020-2035
- Table 23: Western Europe Market Value (USD Million) Forecast by Services, 2020-2035
- Table 24: Western Europe Market Value (USD Million) Forecast by End Use, 2020-2035
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Product , 2020-2035
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Application, 2020-2035
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Test Type, 2020-2035
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Services, 2020-2035
- Table 30: Eastern Europe Market Value (USD Million) Forecast by End Use, 2020-2035
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2020-2035
- Table 32: East Asia Market Value (USD Million) Forecast by Product , 2020-2035
- Table 33: East Asia Market Value (USD Million) Forecast by Application, 2020-2035
- Table 34: East Asia Market Value (USD Million) Forecast by Test Type, 2020-2035
- Table 35: East Asia Market Value (USD Million) Forecast by Services, 2020-2035
- Table 36: East Asia Market Value (USD Million) Forecast by End Use, 2020-2035
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2020-2035
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Product , 2020-2035
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2020-2035
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Test Type, 2020-2035
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Services, 2020-2035
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2020-2035
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2020-2035
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Product , 2020-2035
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Application, 2020-2035
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Test Type, 2020-2035
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Services, 2020-2035
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2020-2035
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2020-2035
- Figure 3: Global Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 4: Global Market Y-o-Y Growth Comparison by Region, 2025-2035
- Figure 5: Global Market Attractiveness Analysis by Region
- Figure 6: North America Market Incremental Dollar Opportunity, 2025-2035
- Figure 7: Latin America Market Incremental Dollar Opportunity, 2025-2035
- Figure 8: Western Europe Market Incremental Dollar Opportunity, 2025-2035
- Figure 9: Eastern Europe Market Incremental Dollar Opportunity, 2025-2035
- Figure 10: East Asia Market Incremental Dollar Opportunity, 2025-2035
- Figure 11: South Asia and Pacific Market Incremental Dollar Opportunity, 2025-2035
- Figure 12: Middle East & Africa Market Incremental Dollar Opportunity, 2025-2035
- Figure 13: North America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 14: Latin America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 15: Western Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 16: Eastern Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 17: East Asia Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 18: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 19: Middle East & Africa Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 20: Global Market - Tier Structure Analysis
- Figure 21: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

