The biopsy closure device market begins 2026 at USD 574.6 million and is projected to grow to USD 1,323.3 million by 2036 at an 8.7% CAGR. Device adoption depends on hemostasis reliability, ease of deployment, and tissue compatibility rather than brand recognition. Once a protocol is validated, replacement cycles follow the same device specifications to maintain staff training and workflow consistency. Clinicians evaluate needle compatibility, closure speed, and patient safety features. Procurement monitors lot traceability, expiration management, and service support. Inventory planning aligns with procedure schedules, and training programs reduce procedural errors and device misuse.
Market growth is driven by procedural volume, clinical adoption, and multi-site deployment rather than promotional activity. Manufacturing emphasizes needle precision, closure mechanism consistency, and sterilization integrity. Release requires dimensional checks, functional testing, and packaging verification. Distributors stage devices near high use hospitals to meet time-sensitive demand. Technical support provides troubleshooting, training, and usage monitoring. Profitability depends on yield stability, low rework, and predictable reorder patterns rather than frequent design changes or aggressive discounting. Expansion follows biopsy volumes, protocol standardization, and hospital program scaling.

Procedure efficiency and clinical workflow integration drive demand in the biopsy closure device market more than total diagnostic volume. In 2026, at about USD 574.6 million, demand is concentrated in hospitals, outpatient imaging centers, and oncology clinics where speed, safety, and hemostasis control during biopsies influence device selection. Hospitals favor systems that integrate with existing instrumentation and simplify post procedure handling. Suppliers that provide reliable kits with consistent performance, inventory support, and clinician training are more likely to secure long term adoption. Growth is supported by the rising number of minimally invasive procedures and increased focus on patient comfort and reduced recovery times.
Supply chain responsiveness and procedural standardization shape the later phase of the biopsy closure device market. As value approaches roughly USD 1,323.3 million by 2036, buyers focus on availability of components, device compatibility with multiple biopsy types, and post sale technical support. Manufacturers that streamline kit assembly, ensure predictable lot to lot performance, and provide training for clinical staff gain a competitive advantage. The increase from mid-five hundred into the high one point five billion reflects cumulative adoption across multiple hospital networks and high volume diagnostic centers rather than a single procedural change. Companies offering reliable delivery, consistent product performance, and integrated support tend to secure preferred vendor status.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 574.6 million |
| Forecast Value (2036) | USD 1,323.3 million |
| Forecast CAGR 2026 to 2036 | 8.7% |
Biopsy closure devices are used to seal tissue and vascular entry sites following percutaneous biopsies, reducing bleeding risk and enabling faster patient recovery. Earlier procedures relied on manual compression and prolonged monitoring, which increased staff time and patient discomfort. Modern closure systems use mechanical plugs, absorbable materials, or suture-mediated technology to provide rapid hemostasis and predictable sealing. Hospitals, interventional radiology units, and outpatient clinics evaluate these devices by deployment reliability, material biocompatibility, ease of use, and compatibility with different needle sizes. Adoption is concentrated in high-volume centers performing liver, kidney, or soft tissue biopsies, where consistent closure reduces complications, shortens recovery times, and optimizes procedural workflow. Device performance influences clinical efficiency, patient throughput, and nursing resource allocation.
Manufacturers differentiate products through material consistency, deployment accuracy, and regulatory compliance rather than cost alone. Early market adoption focused on pilot programs, limiting scale and feedback for design refinement. Current demand favors standardized devices that are compatible with a range of biopsy needles and imaging modalities. Suppliers compete on device reliability, ease of integration into procedural protocols, and post-market training support. Hospitals assess total value through reduced complication rates, shorter post-procedure observation, and decreased re-intervention needs, rather than per-unit price. Distributors add value through supply chain reliability, staff education, and device traceability. Over time, growth tracks the volume of image-guided biopsies, minimally invasive procedures, and demand for safer, faster recovery workflows in both inpatient and outpatient settings.
Hospital protocols and clinical safety requirements are the primary drivers of the Biopsy Closure Device Market in 2026, with emphasis on minimizing post-procedural complications and ensuring reliable hemostasis. High-volume cardiovascular and oncology procedures require devices that integrate with workflow, reduce recovery time, and comply with regulatory standards. Segment adoption is shaped by closure mechanism, ease of deployment, and compatibility with various biopsy needles. Suppliers that offer consistent consumable supply, technical training, and device support can capture long-term contracts. Market growth is concentrated in cardiovascular and oncology biopsies, creating predictable adoption cycles for manufacturers capable of combining reliability, service, and regulatory compliance.

Collagen-based plugs dominate roughly 46% of the market because they provide rapid hemostasis for cardiovascular biopsies. Manufacturers supporting these devices must ensure staff training, lot-to-lot quality consistency, and reliable consumable supply. Sealant-based systems are commonly used in oncology biopsies, requiring guidance on preparation, handling, and storage to maintain tissue integrity. Suture-mediated devices address complex vascular procedures and demand ongoing procedural support and technical guidance from suppliers. Product type selection dictates the level of engagement suppliers must provide, including preventive maintenance, operator training, and inventory management, which directly influences adoption across clinical programs.
Variation in workflow requirements and procedural complexity is another critical consideration for suppliers. Collagen-based plugs need precise deployment and consistent quality to prevent complications during cardiovascular biopsies. Sealant-based systems involve preparation steps and controlled application, increasing training and procedural oversight. Suture-mediated devices require advanced technical skills and continued on-site support. Switching device types within a facility is uncommon due to training, inventory, and validation requirements. Suppliers that provide comprehensive technical support, consumable management, and training packages strengthen adoption rates, maximize retention, and enhance procedural safety across multiple use cases.
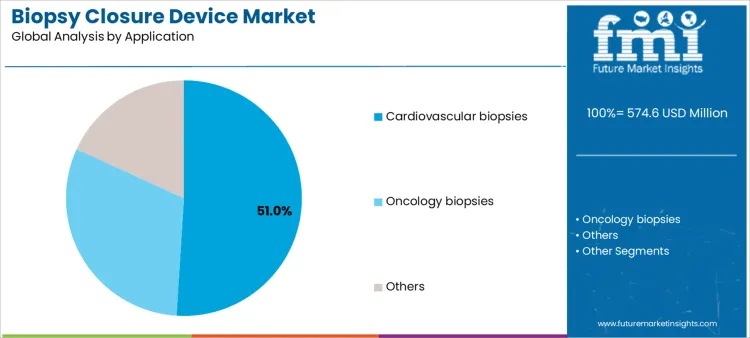
Cardiovascular biopsy procedures represent approximately 51% of demand because frequent procedures require reliable vascular closure to minimize bleeding and improve recovery time. Oncology biopsies, though less frequent, necessitate precise tissue handling and closure, presenting high-value opportunities for specialized devices. Niche applications, including interventional radiology procedures, generate lower volumes but require complex technical support. Device adoption patterns guide suppliers in prioritizing inventory, procedural training, and technical support allocation. Aligning products with both high-volume cardiovascular and specialized oncology use ensures optimal market penetration, service delivery, and predictable revenue streams.
Variation in clinical applications shapes supplier engagement and operational planning. High-volume cardiovascular programs benefit from standardized devices, integrated training, and preventive maintenance contracts to ensure consistent procedural outcomes. Oncology procedures demand flexibility, rapid consumable replenishment, and on-demand technical guidance. Specialized or rare procedures require customized support, device guidance, and consistent consumables. Segment shares are determined by procedure frequency, clinical complexity, and workflow integration rather than short-term purchasing trends. Suppliers offering reliable training, responsive service, and continuous consumable availability can capture larger market share and reinforce long-term hospital relationships.
The Biopsy Closure Device Market is shaped by hospitals and clinics prioritizing reliable hemostasis and minimal patient complications following tissue biopsy procedures. Devices are evaluated based on closure consistency, ease of deployment, compatibility with multiple tissue types, and procedural speed. Selection decisions involve interventional radiologists, nurses, and hospital procurement teams. Suppliers that provide validated devices with predictable performance and clear usage instructions are preferred. This positions biopsy closure devices as essential procedural tools rather than optional accessories, emphasizing operational reliability, patient safety, and repeatable outcomes.
Hospitals increasingly follow standardized post-biopsy protocols to reduce complications and improve workflow efficiency. This drives demand in the Biopsy Closure Device Market toward devices that reliably close tissue sites, integrate with imaging or guidance systems, and require minimal operator intervention. Buyers prefer devices with consistent deployment, clear handling instructions, and minimal training requirements. Suppliers are evaluated on product consistency, documentation quality, and service support rather than on isolated design features. Devices that align with established procedural protocols are more likely to be adopted across multiple sites and operators.
Recurring demand comes from hospitals, diagnostic centers, and outpatient clinics performing high volumes of tissue biopsies. Each procedure requires a reliable closure device, creating predictable repeat orders. The Biopsy Closure Device Market also benefits when multi-site hospital systems standardize on a single supplier or device type. Suppliers that provide training, quality assurance, and responsive replacement support can secure long-term business. Embedding devices into procedural protocols ensures ongoing demand tied to biopsy volumes and clinical workflow requirements.
Closure devices carry additional cost per procedure compared with manual hemostasis methods, which can limit adoption in low-volume centers or cost-sensitive facilities. The Biopsy Closure Device Market also faces adoption constraints from staff familiarity, training needs, and hospital procurement cycles. Regulatory compliance for device sterility and performance validation further slows adoption. Suppliers entering the market must provide robust clinical support, training, and consistent product availability. Adoption is concentrated in high-volume facilities and specialty centers where operational efficiency and patient safety are prioritized.

| Country | CAGR |
|---|---|
| USA | 11.5% |
| Brazil | 12.0% |
| China | 12.5% |
| India | 12.7% |
| Germany | 9.7% |
Demand for biopsy devices is rising as hospitals, oncology centers, and interventional radiology units adopt minimally invasive procedures with improved hemostasis and patient safety. India leads with a 12.7% CAGR, supported by expanding diagnostic infrastructure, growing cancer screening programs, and adoption of advanced closure technologies. China follows at 12.5%, driven by high procedure volumes, hospital modernization, and preference for efficient post biopsy care. Brazil records 12.0%, reflecting increasing use in private and public healthcare facilities. The USA grows at 11.5%, shaped by replacement of conventional closure methods and adoption of automated devices. Germany, at 9.7%, reflects steady uptake in a mature healthcare market with standardized interventional protocols.
United States demand for biopsy closure devices is growing along an 11.5% CAGR trajectory due to increasing outpatient procedures, minimally invasive diagnostics, and the expansion of interventional radiology programs. Hospital procurement committees, radiology directors, and surgical supply teams control most purchases. Device selection emphasizes safety, hemostasis efficiency, and ease of deployment in both inpatient and outpatient settings. Once validated, closure devices are standardized across multiple hospital networks. Training, procedural protocols, and adoption metrics influence usage. Import devices dominate complex tissue and vascular applications, while domestic suppliers cover routine procedures. Capital planning aligns with facility expansion and procedural volume growth. Replacement cycles follow procedure intensity. Commercial opportunities exist for suppliers offering integrated training, technical support, and consistent supply contracts across large healthcare networks.
Brazil is experiencing a 12% CAGR in biopsy closure device demand as private hospital chains increase minimally invasive procedures in oncology and diagnostic services. Network purchasing offices and interventional radiology teams control most procurement decisions. Adoption focuses on rapid hemostasis, patient safety, and procedure efficiency. Import devices dominate premium applications, while domestic suppliers cover routine biopsies. Once approved, deployment expands across multiple affiliated hospitals and outpatient centers. Training programs, workflow integration, and protocol adherence influence adoption rates. Capital spending follows network expansion and new procedure launches. Replacement cycles track procedural load rather than device age. Commercial success depends on suppliers providing comprehensive technical support, standardized instrument kits, and reliable logistics across multiple network hospitals rather than isolated facility sales.
Biopsy closure device market in China is growing at a 12.5% CAGR due to expansion of tertiary hospitals, oncology centers, and outpatient diagnostic programs. Large hospital networks control most procurement. Adoption emphasizes hemostasis effectiveness, tissue compatibility, and procedural efficiency in high-volume biopsy programs. Domestic suppliers serve standard applications, while international brands retain positions in premium and complex procedures. Tender processes and multi-year capital planning govern procurement schedules. Once approved, devices are deployed across multiple hospitals and departments. Training, certification, and adherence to clinical protocols influence adoption. Replacement cycles track procedural volume rather than device aging. Budget allocation aligns with hospital expansion and new oncology programs. Commercial success depends on inclusion in provincial and national procurement catalogs and integration into standardized workflows rather than isolated clinic purchases.
Expansion of interventional radiology and specialty hospital programs supports a 12.7% CAGR for biopsy closure devices in India. Large private hospitals and diagnostic centers lead procurement. Device selection prioritizes hemostasis efficiency, procedural speed, and integration with imaging workflows. Import devices dominate complex tissue or vascular cases, while domestic suppliers cover routine procedures. Procurement authority sits with interventional committees and capital boards. Once approved, devices are deployed across multiple affiliated sites. Training, protocol adherence, and workflow standardization influence utilization. Replacement cycles track procedural load and device wear. Capital allocation aligns with facility expansions and new service launches. Commercial success depends on early involvement in new program planning, reliable supply, and integrated training rather than individual hospital or single department sales.
Biopsy closure device demand in Germany is growing at a 9.7% CAGR due to structured guideline implementation, standardized interventional procedures, and reimbursement-driven adoption. University hospitals and regional medical centers control most procurement. Adoption focuses on hemostasis performance, procedure reproducibility, and safety. Evaluation emphasizes device reliability, operator ergonomics, and workflow integration. Procurement follows health technology assessments and hospital capital approval schedules. Import devices dominate premium applications, while domestic suppliers serve standard procedures. Once approved, deployment expands across multiple departments and hospitals. Training, documentation, and compliance influence utilization. Replacement cycles follow procedure volume. Budget allocation aligns with service line expansion. Commercial success depends on framework inclusion, reference site adoption, and provision of technical support rather than sales to isolated clinics or individual departments.

Biopsy closure device selection follows procedural workflow, site sterility requirements, and hemostasis protocols rather than simple catalog review. Clinicians define tissue type, needle gauge, closure speed, and track sealing requirements before approving suppliers. Cardinal Health, Becton Dickinson, Merit Medical, Terumo, Teleflex compete during evaluation and protocol integration stages. Once a device is incorporated into procedural SOPs, staff training, inventory management, and quality control measures lock the choice. Cardinal Health leverages hospital distribution networks and bundled procedural kits. Becton Dickinson positions devices through broad interventional portfolios and standardization programs. Merit Medical emphasizes procedural flexibility and hemostasis reliability. Terumo focuses on clinical evidence and cross procedure compatibility. Teleflex targets rapid deployment and user friendly designs for high volume interventional suites.
Competitive outcomes depend on procedural reliability, service support, and documentation completeness rather than pricing alone. Clinicians assess device deployment accuracy, track sealing performance, ease of handling, and tissue compatibility during trials. Procurement teams value predictable supply, batch traceability, and training support across multiple facilities. Cardinal Health benefits from integrated supply chains and procedural standardization. Becton Dickinson secures positions where device familiarity aligns with existing interventional platforms. Merit Medical gains traction through adaptability across tissue types. Terumo holds share in programs requiring established clinical evidence and multi procedure compatibility. Teleflex competes in high throughput environments with rapid device readiness. Market positions vary by hospital size, procedure volume, and internal protocol adoption.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD million |
| Product Type | Collagen-based plugs; Sealant-based systems; Suture-mediated devices |
| Application | Cardiovascular biopsies; Oncology biopsies; Others |
| End User | Hospitals; Diagnostic centers; ASCs |
| Region | Asia Pacific; Europe; North America; Latin America; Middle East & Africa |
| Countries Covered | United States, Canada, Mexico, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Brazil, Chile, Saudi Arabia, Turkey, South Africa, Rest of regions |
| Key Companies Profiled | Cardinal Health, Inc.; Becton, Dickinson and Company; Merit Medical Systems, Inc.; Terumo Corporation; Teleflex Incorporated |
| Additional Attributes | Dollar by sales by product and application, cardiovascular versus oncology case mix, consumable usage per procedure, hemostasis method split, device deployment time impact, training and service intensity, inventory turnover, regulatory and sterility compliance requirements |
How big is the biopsy closure device market in 2026?
The global biopsy closure device market is estimated to be valued at USD 574.6 million in 2026.
What will be the size of biopsy closure device market in 2036?
The market size for the biopsy closure device market is projected to reach USD 1,323.3 million by 2036.
How much will be the biopsy closure device market growth between 2026 and 2036?
The biopsy closure device market is expected to grow at a 8.7% CAGR between 2026 and 2036.
What are the key product types in the biopsy closure device market?
The key product types in biopsy closure device market are collagen-based plugs, sealant-based systems and suture-mediated devices.
Which application segment to contribute significant share in the biopsy closure device market in 2026?
In terms of application, cardiovascular biopsies segment to command 51.0% share in the biopsy closure device market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.