Cell and Gene Therapy Bioreactor PAT and Analytics Test Platforms Market
The Cell And Gene Therapy Bioreactor PAT And Analytics Test Platforms Market Is Segmented By Platform Type (Integrated Bioreactors, At-Line Analyzers, Single-Use Sensors, Control Software), Analytics Modality (Spectroscopy, Metabolite Analyzers, Cell Counters, Imaging Systems, Molecular Assays), Workflow Stage (Process Development, Clinical Manufacturing, Commercial Manufacturing), Therapy Type (Cell Therapy, Gene Therapy), End User (Biopharma Developers, CDMOs, Academic Centers, Research Institutes), And Region. Forecast For 2026 To 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Cell and Gene Therapy Bioreactor PAT and Analytics Test Platforms Market Size, Market Forecast and Outlook By FMI
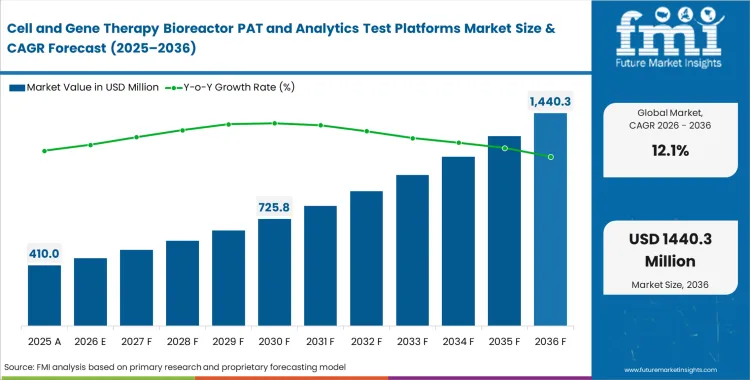
The cell and gene therapy bioreactor PAT and analytics test platforms market crossed a valuation of USD 370 million in 2025. Revenue is expected to reach USD 410 million in 2026 at a CAGR of 12.1% during the forecast period. Sustained growth drives total valuation to USD 1,290 million by 2036 as clinical trial pipelines accelerate the transition from manual sampling toward continuous automated monitoring.
Quality control directors at contract development firms face intense pressure to eliminate batch-failure risks caused by offline sampling delays. Relying on periodic manual extraction exposes fragile cellular materials to contamination and requires dedicated technicians. Incorporating inline bioprocessing analytics equipment changes this dynamic entirely, embedding robust bioreactor analytics for cell therapy manufacturing right at the production line. Failing to automate parameter tracking threatens regulatory approval timelines and dramatically increases per-dose production costs.
Summary of Cell and Gene Therapy Bioreactor PAT and Analytics Test Platforms Market
- Market Snapshot
- The cell and gene therapy bioreactor PAT and analytics test platforms market is valued at USD 370 million in 2025 and is projected to reach USD 1,290 million by 2036.
- The industry is set to expand at a CAGR of 12.1% from 2026 to 2036, creating an incremental opportunity of USD 880 million over the period.
- Platform selection in this market is centered on process visibility, data integrity, and the ability to monitor critical culture conditions without disrupting closed bioreactor environments.
- Growth is being supported by a larger pool of approved cell and gene therapies and continued investment in specialized manufacturing capacity.
- Demand and Growth Drivers
- Drug developers are under pressure to reduce batch variability and assemble compliance packages that can support the shift from clinical production to commercial manufacturing.
- Manufacturing sites are investing in automated platforms that can run multiple bioreactors in parallel, since faster process mapping shortens development timelines and eases transfer into larger-scale operations.
- Batch failure carries a high cost in autologous therapy production, which is pushing manufacturers toward real-time analytical tools that can detect metabolic drift before product value is lost.
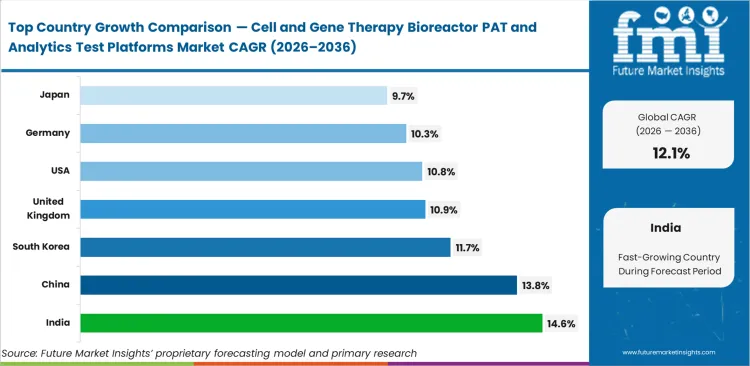
- India is forecast to record the fastest growth, with demand for these platforms rising at a 14.6% CAGR through 2036. China follows at 13.8%, South Korea at 11.7%, the United Kingdom at 10.9%, the United States at 10.8%, Germany at 10.3%, and Japan at 9.7%. This spread reflects differences in manufacturing build-out, clinical scale-up activity, and local investment in advanced therapy infrastructure.
- Legacy plant infrastructure remains a restraint. Connecting new analytical software with installed hardware adds validation work, extends deployment cycles, and raises capital commitment in regulated facilities.
- Product and Segment View
- Spending in this market covers small-scale automated bioreactor systems, single-use sensors, at-line media testing tools, and the software layer used to manage upstream process control.
- These platforms are used across early cell-expansion studies, viral vector optimization, and late-stage tech transfer into commercial production.
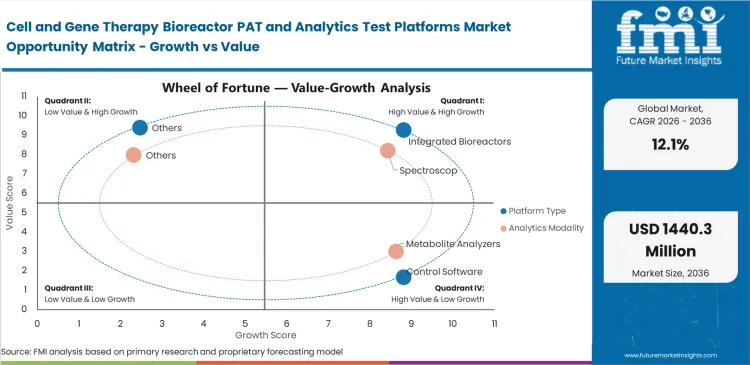
- Integrated bioreactors are expected to account for 34% of the market in 2026, as buyers continue to reduce the number of disconnected instruments on the manufacturing floor and tighten digital oversight.
- Spectroscopy is anticipated to represent 28% share of the modality segment in 2026, supported by demand for non-destructive monitoring methods such as Raman-based analysis.
- Process development is projected to secure 41% share in 2026 because culture conditions, feed strategy, and control parameters are typically fixed well before commercial launch.
- Cell therapy is likely to contribute 58% of total market share in 2026, reflecting the operational intensity of autologous workflows and the need for tighter control during expansion stages.
- Biopharma developers are expected to account for 46% of the end-user base in 2026, since many sponsors prefer to establish process control internally before transferring production to contract manufacturers.
- This assessment covers upstream testing platforms connected directly to the bioreactor and excludes downstream purification systems and general laboratory instruments not configured for live process control.
- Geography and Competitive Outlook
- Manufacturing capacity additions are making India, China, and South Korea the fastest-growing markets, while the United States continues to provide a large and established base of high-value demand.
- Vendor success depends heavily on software compatibility with installed factory systems and on whether the platform can support scale-up from pilot work to commercial production without reworking the control strategy.
- The competitive structure remains moderately fragmented. Sartorius, Cytiva, Thermo Fisher Scientific, and Repligen are prominent suppliers, alongside Getinge, and PBS Biotech.
- Buyers show a clear preference for suppliers that can provide bioreactors, sensors, and analytics software as one integrated package, since single-vendor procurement reduces interface issues and simplifies workflow management.
Once initial clinical validation proves inline measurement reduces batch deviation, broad commercial adoption becomes inevitable. Continuous data streams replace retrospective batch records. Production managers implementing cell therapy PAT systems no longer guess about cell viability mid-run.
India is forecasted to rapidly scale at an estimated 14.6% CAGR as domestic CDMOs build specialized infrastructure, while China is likely to track at an anticipated 13.8% on aggressive regenerative medicine investments. South Korea is projected to advance at 11.7% based on rapid scale-up initiatives. Buyers in United Kingdom are poised to realize an estimated 10.9% growth trajectory. United States is likely to maintains steady expansion at 10.8% due to high clinical trial volumes. Germany is set to grow at 10.3% and Japan is predicted to show 9.7% compound annual progress. Divergence across these regions stems directly from varying regulatory attitudes toward automated real-time monitoring in CGT bioreactors.
Segmental Analysis
Cell and Gene Therapy Bioreactor PAT and Analytics Test Platforms Market Analysis by Platform Type
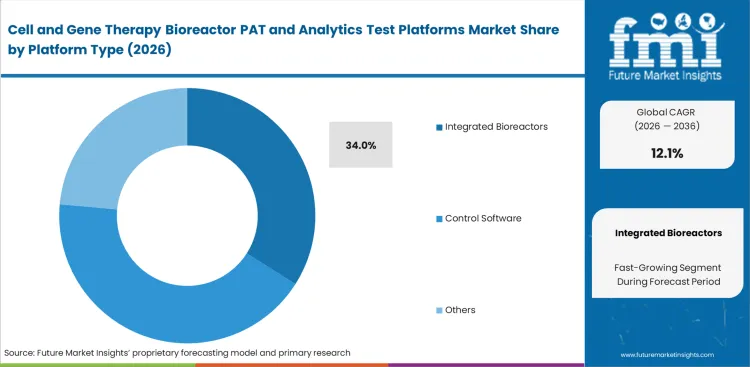
Factory floor space limits dictate how companies buy their monitoring equipment. Buying separate sensors and control towers forces facilities to validate multiple software connections, which creates heavy integration bottlenecks. Pre-packaged closed system bioprocessing architecture removes this risk completely. Driven by manufacturing directors actively choosing all-in-one units to minimize dangerous aseptic connections, the integrated bioreactors segment is expected to account for a 34.0% share in 2026. Production managers frequently find that mixing probes from different vendors causes timing errors during fast metabolic shifts. Trying to piece together a custom setup simply delays getting the facility running by several months.
- Sensor Consolidation: Pre-calibrated optical interfaces eliminate manual probe insertion steps. Manufacturing supervisors avoid daily calibration routines that risk batch sterility.
- Footprint Optimization: Combining hardware reduces cleanroom space requirements per therapy batch. Facility planners gain higher throughput within existing physical infrastructure constraints.
- Software Unification: Centralized dashboards aggregate metabolic data into single compliance reports. Quality assurance directors avoid matching timestamps across disconnected vendor databases.
Cell and Gene Therapy Bioreactor PAT and Analytics Test Platforms Market Analysis by Analytics Modality
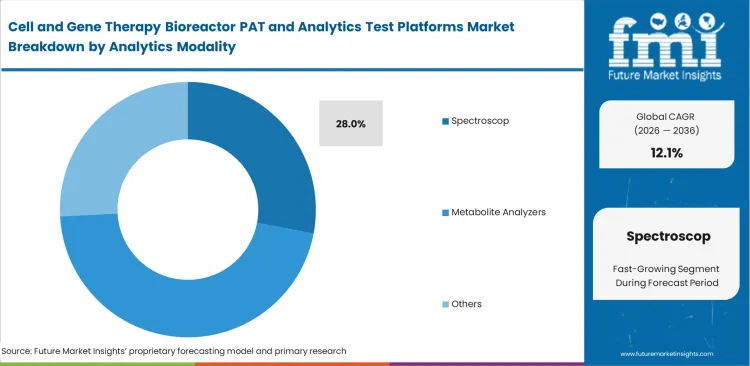
As the spectroscopy category is anticipated to capture a 28.0% share in 2026, reading critical cell health indicators without destroying the sample is changing how facilities handle sensitive cell expansion. Operators use this modality to measure multiple analytes simultaneously. Process development scientists rely on real-time bioprocess Raman analyzer tools to watch glucose and lactate trends without ever taking a physical sample out of the tank. Old-fashioned offline counting wastes expensive media and opens the door to contamination. Small-batch autologous treatments simply cannot afford the volume loss tied to daily manual extraction. Continuing to use older sampling methods guarantees a lower final yield and creates major gaps in cell therapy quality control analytics.
- Multiplexed Tracking: Optical signatures identify multiple metabolic changes simultaneously. Bioprocess engineers adjust feed strategies before cell viability drops.
- Volume Preservation: Non-invasive reads consume zero physical product volume. Clinical researchers maximize final dose quantities for rare patient materials.
- Trend Prediction: Continuous data feeds enable predictive algorithmic process control. Automation specialists prevent catastrophic pH crashes hours before they occur.
Cell and Gene Therapy Bioreactor PAT and Analytics Test Platforms Market Analysis by Workflow Stage
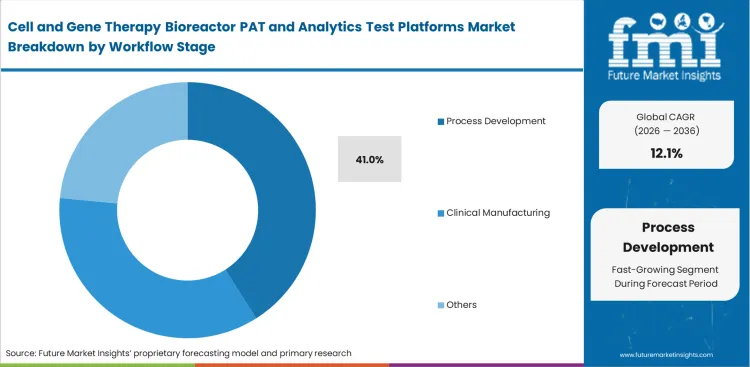
Turning laboratory science into a full-scale manufacturing line requires massive amounts of data right at the start. Scientists need to map the exact metabolic boundaries of new therapies before they ever attempt a commercial batch. Researchers use highly instrumented single-use bioreactors to establish safe operating ranges for their clinical filings. Skipping this deep analytical work early on guarantees production failures during the tech transfer phase. Fixing a broken process later in development costs far more than buying the right tracking tools up front, which explains why the process development segment is set to secure a 41.0% share in 2026 as facilities prioritize early-stage optimization.
- Parameter Mapping: Intense monitoring defines critical operational thresholds. Development scientists build robust regulatory dossiers demonstrating process understanding.
- Scale-Down Modeling: Small-format automated systems replicate massive commercial environments. R&D directors test extreme variations without wasting expensive clinical-grade media.
- Tech Transfer Validation: Standardized analytical platforms ensure seamless protocol handover. Manufacturing site leads replicate exact conditions achieved in the original laboratory.
Cell and Gene Therapy Bioreactor PAT and Analytics Test Platforms Market Analysis by Therapy Type
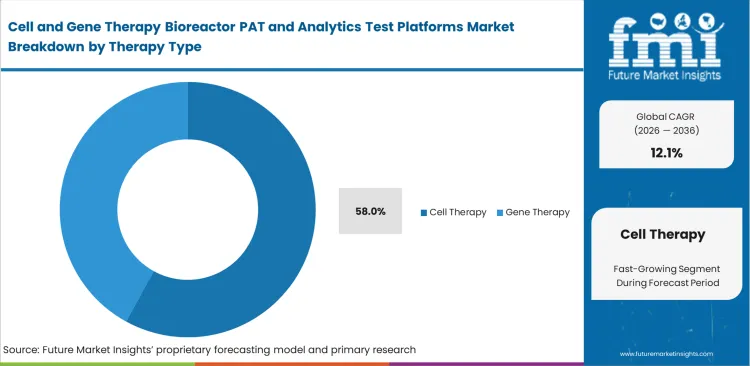
Patient-sourced starting materials vary wildly and demand incredibly responsive production controls. Production supervisors rely on automated and closed cell therapy processing systems to handle these highly individualized batches safely. Reflecting the massive wave of autologous treatments currently entering clinical trials, the cell therapy segment is poised to account for a 58.0% share in 2026. Processing multiple patients at once in a single building requires perfect segregation and tracking. Trying to manage this level of operational complexity with manual testing scales terribly. Facilities that refuse to upgrade their monitoring systems hit strict capacity ceilings almost immediately.
- Batch Segregation: Automated inline tracking prevents cross-contamination across concurrent patient runs. Quality heads ensure absolute traceability from apheresis to final infusion.
- Autologous Variability: Real-time feedback adjusts parameters dynamically based on unique patient cell health. Production managers salvage weak starting materials that fixed protocols would destroy.
- Throughput Scaling: Closed-loop analytics allow single operators to manage multiple bioreactors. Operations directors expand facility capacity without linearly increasing headcount.
Cell and Gene Therapy Bioreactor PAT and Analytics Test Platforms Market Analysis by End User
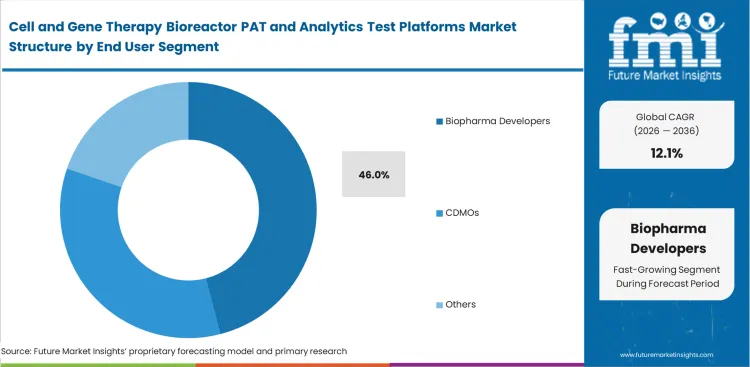
Keeping critical production capabilities in-house protects valuable manufacturing secrets from competitors. Executive boards are approving huge budgets to build their own internal testing suites, putting the biopharma developers segment on track to represent a 46.0% share in 2026. Outsourcing the entire cell therapy manufacturing process creates major intellectual property risks for drug sponsors. Internal teams want direct access to live analytical data so they can master their own manufacturing workflows natively. Handing everything over to an outside partner leaves the original developer blind to small process deviations. Running the analytics internally speeds up the creation of next-generation product iterations.
- IP Protection: Keeping advanced analytical recipes in-house secures competitive process advantages. Corporate strategy officers block competitors from reverse-engineering proprietary growth profiles.
- Iteration Speed: Direct access to raw sensor data accelerates process optimization. R&D heads modify protocols immediately without waiting for external partner reports.
- Strategic Capacity: Owning instrumented bioreactors guarantees clinical trial material availability. Supply chain directors eliminate wait times associated with crowded contract manufacturing schedules.
Cell and Gene Therapy Bioreactor PAT and Analytics Test Platforms Market Drivers, Restraints, and Opportunities
Contamination during cell therapy production ruins highly sensitive patient materials. Quality control officers are replacing manual sampling routines with fully automated sensors to solve this exact problem. Every time a technician opens a bioreactor to take a sample, they introduce a failure risk. Switching to bioprocess fermentation monitoring removes this physical step entirely. Facilities want to reduce their batch failure rates to keep costs down. Taking human handling out of the equation improves the final product and saves money that would otherwise be lost on failed batches.
Older software systems make it difficult for facilities to install new testing tools. Automation engineers often struggle to connect modern optical sensors to outdated data networks. Many factories still run on disconnected systems that simply cannot process constant streams of dense analytical data. Putting in new sensors usually means the facility also has to replace its core control software. Replacing the entire IT system at once creates a heavy validation burden for CGT analytics platforms, causing many operations managers to delay these upgrades.
- Centralized Data Storage: Cloud architects build unified systems that gather information from different factory machines. Connecting these data points helps facilities run better CGT batch failure reduction analytics.
- Predictive Algorithm Deployment: Machine learning teams train models on continuous production data. These algorithms help operators catch process deviations early before they ruin a batch.
- Miniaturized Sensor Integration: Hardware designers fit single use bioprocessing probes and sensors straight into very small development vessels. Early-stage researchers gain exact metabolic readings without needing large-scale equipment.
Regional Analysis
Based on regional analysis, cell and gene therapy bioreactor PAT and analytics test platforms market is segmented into North America, Europe, and Asia Pacific across 40 plus countries. Regulatory attitudes toward automated release testing and aggressive investments in local manufacturing capacity decide how quickly different regions adopt these essential tools.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 14.6% |
| China | 13.8% |
| South Korea | 11.7% |
| United Kingdom | 10.9% |
| United States | 10.8% |
| Germany | 10.3% |
| Japan | 9.7% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Asia Pacific Cell and Gene Therapy Bioreactor PAT and Analytics Test Platforms Market Analysis
Contract manufacturers across this territory are rushing to build out massive facilities to win global pharmaceutical clients. These regional operators buy advanced testing systems to prove they can match Western quality standards. Companies running older setups just cannot secure new deals because they lack the ability to monitor cell health continuously.
- India: India is upgrading manufacturing capacity at a pace that is lifting demand for automated bioreactor testing platforms. Demand for these systems in the country is anticipated to rise at a 14.6% CAGR through 2036. Many local contract manufacturers are bypassing manual setups and moving directly to digital monitoring architectures, since early investment in automation improves process control and strengthens their position against global facilities still working through legacy upgrades.
- China: China is expanding regenerative medicine production through state-backed funding and large-scale capacity build-outs. The market for these platforms in China is expected to grow at a 13.8% CAGR through 2036. Biopharma operators are building multi-line manufacturing campuses that require connected sensors and centralized analytics to manage high batch volumes with tighter operational control and lower execution risk.
- South Korea: South Korea places a premium on development speed, which keeps analytical reliability at the center of equipment purchasing decisions. Sales of these platforms in South Korea are projected to increase at a 11.7% CAGR by 2036. Standardized testing supports faster progression from clinical production into commercial scale and gives regional contract manufacturers a better chance of securing early development programs before rival facilities complete validation.
- Japan: Japan’s market is shaped by strict process control expectations and close regulatory attention to manufacturing consistency. Demand for these platforms across Japan is likely to rise at a 9.7% CAGR during the assessment period. Buyers continue to favor advanced optical and spectral monitoring tools because detailed batch traceability remains central to compliance, validation, and day-to-day production oversight.
FMI's report includes additional countries not in bullets. Suppliers across the region are grouping together to support these high-tech installations.
Europe Cell and Gene Therapy Bioreactor PAT and Analytics Test Platforms Market Analysis
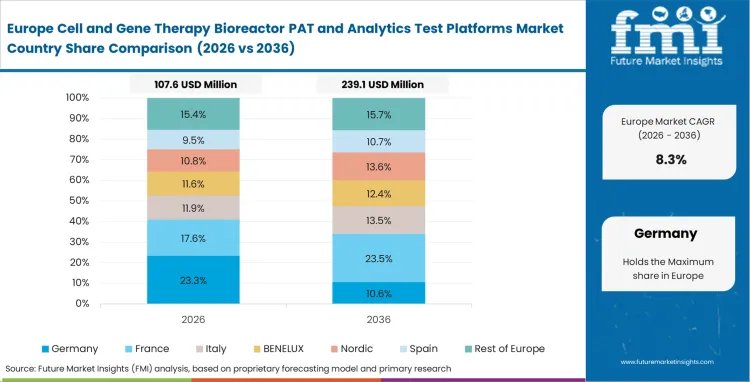
Strict European rules regarding advanced medical products force local companies to rethink how they buy equipment. Quality assurance directors actively seek continuous data logs to prove they have absolute control over their biological processes. Facilities that fall behind in automating their documentation usually face long, difficult audit cycles.
- United Kingdom: The United Kingdom benefits from close collaboration between academic research centers and private therapy developers, which accelerates movement from laboratory work into clinical manufacturing. Demand for these platforms in the country is expected to grow at a 10.9% CAGR through 2036. Developers are bringing bioprocess validation tools into the workflow early so process behavior is understood before transfer to commercial facilities, reducing scale-up risk for sensitive cell cultures.
- Germany: Germany’s high labor cost structure is pushing manufacturers to reduce dependence on manual sampling and operator-intensive monitoring routines. The market for these platforms in Germany is forecast to expand at a 10.3% CAGR during the study period. Manufacturing teams are replacing manual checks with automated optical sensing and integrated analytics, which improves labor productivity and helps control the cost of personalized therapy production across regulated sites.
FMI's report includes additional countries not in bullets. Cross-border partnerships often agree on specific equipment setups to keep their data consistent.
North America Cell and Gene Therapy Bioreactor PAT and Analytics Test Platforms Market Analysis
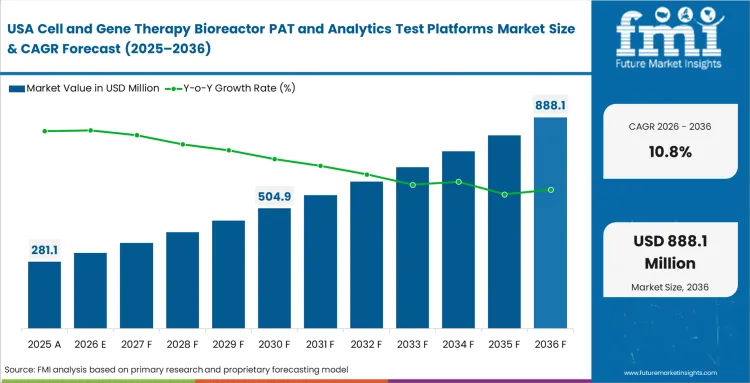
The huge volume of clinical trials happening locally creates a massive need for tools that can handle scale-up challenges. R&D directors are always looking for better ways to understand complicated viral vectors and engineered cells. Moving successfully into final clinical phases requires the deep insights that these continuous platforms provide.
- United States: The United States continues to attract strong venture funding for new cell and gene therapy manufacturing infrastructure, which supports premium spending on advanced bioreactor testing and analytics systems. Sales of these platforms in the United States are projected to rise at a 10.8% CAGR by 2036. Companies are using these investments to strengthen process control, build regulatory credibility early, and reduce dependence on scarce skilled operators through higher levels of automation.
FMI's report includes additional countries not in bullets. A lack of specialized workers pushes these companies even harder toward automation.
Competitive Aligners for Market Players
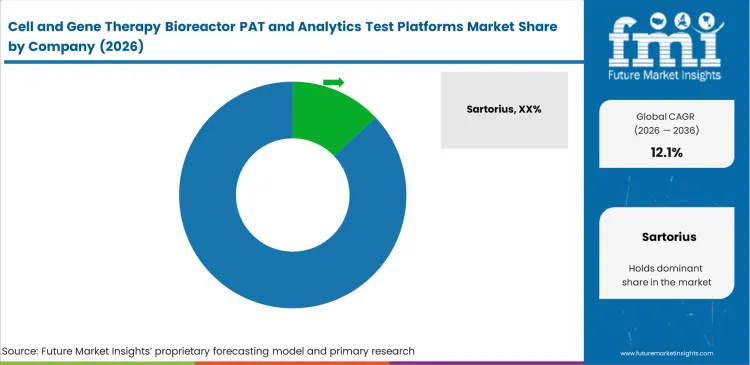
Sensor stability over extended production runs remains a decisive factor in vendor selection. Platforms that require repeated manual recalibration of optical probes during an active batch are usually screened out. Suppliers that secure contracts in the CGT analytics platform market typically show that their sensors can maintain baseline accuracy across roughly three weeks of cell growth. Greater weight is placed on that operating consistency than on headline sensitivity figures at installation.
Software compatibility with installed facility systems is another key screening factor. Established vendors strengthen their position by embedding new optical monitoring tools into control environments that operators already know how to use. Teams comparing suppliers such as Sartorius, Cytiva, and Repligen often find that implementation risk sits at the center of the decision. Smaller hardware providers lose ground when their software cannot exchange data cleanly with legacy manufacturing record systems, since that creates avoidable validation work and slows commercial deployment.
Contract manufacturers are equally cautious about sensor lock-in. Supply chain teams prefer readers that can work with single-use probes sourced from multiple CGT bioreactor PAT suppliers. Platforms tied to proprietary consumables face resistance from large buyers that need flexibility in sourcing and cost control. Vendor choice often favors analytics tools that can operate across different bioreactor brands, since multi-source compatibility helps facilities manage procurement risk and keep recurring operating costs under tighter control.
Key Players in Cell and Gene Therapy Bioreactor PAT and Analytics Test Platforms Market
- Sartorius
- Cytiva
- Thermo Fisher Scientific
- Repligen
- Getinge
- PBS Biotech
Scope of the Report
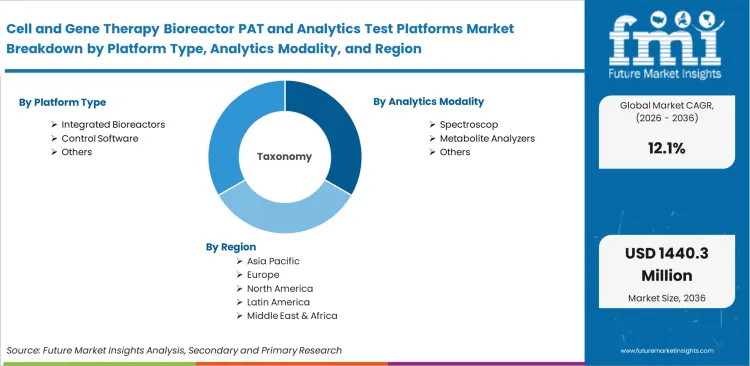
| Metric | Value |
|---|---|
| Quantitative Units | USD 410 million to USD 1,290 million, at a CAGR of 12.1% |
| Market Definition | This category includes integrated instrumentation and software systems designed to continuously monitor and control critical parameters during advanced therapy biomanufacturing. |
| Segmentation | Platform type, Analytics modality, Workflow stage, Therapy type, End user, Region |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia & Pacific, Middle East & Africa |
| Countries Covered | United States, China, India, Germany, United Kingdom, Japan, South Korea |
| Key Companies Profiled | Sartorius, Cytiva, Thermo Fisher Scientific, Repligen, Getinge, PBS Biotech |
| Forecast Period | 2026 to 2036 |
| Approach | Installed base of single-use systems in commercial-stage biopharma facilities anchors baseline metrics. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Cell and Gene Therapy Bioreactor PAT and Analytics Test Platforms Market by Segments
Platform Type
- Integrated bioreactors
- At-line analyzers
- Single-use sensors
- Control software
Analytics Modality
- Spectroscopy
- Metabolite analyzers
- Cell counters
- Imaging systems
- Molecular assays
Workflow Stage
- Process development
- Clinical manufacturing
- Commercial manufacturing
Therapy Type
- Cell therapy
- Gene therapy
End User
- Biopharma developers
- CDMOs
- Academic centers
- Research institutes
Regions:
- Asia Pacific
- India
- China
- Japan
- South Korea
- Indonesia
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- Italy
- France
- United Kingdom
- Spain
- Benelux
- Nordics
- Central & Eastern Europe
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Turkey
- Rest of Middle East & Africa
Bibliography
- European Directorate for the Quality of Medicines & HealthCare. (2024). New European Pharmacopoeia Commission approach to gene therapy.
- Food and Drug Administration. (2024). Human gene therapy products incorporating human genome editing: Guidance for industry.
- Randall, N., Gavin, C., Driscoll, S., Hartlep, M., Kuennecke, W., Neffling, M., & Piras, G. (2024). Improving cell and gene therapy manufacturing processes by automated on-line and in-line bioprocess analytical technologies. ECI Symposium Series.
- Dan, L., Ying Ying, W., Prabhu, A. V., bin Abdul Rahim, A. A., & Jia Sheng, Z. L. (2024). Device for automated aseptic sampling: Automated sampling solution for future cell and gene manufacturing. Frontiers in Bioengineering and Biotechnology, 12, 1452674.
- McColl-Carboni, A., Dollive, S., Laughlin, S., Lushi, R., MacArthur, M., Zhou, S., Gagnon, J., Smith, C. A., Burnham, B., Horton, R., Lata, D., Uga, B., Natu, K., Michel, E., Slater, C., DaSilva, E., Gally, J., Domm, J., & McGivney, J. B. (2024). Analytical characterization of full, intermediate, and empty AAV capsids. Gene Therapy, 31(5-6), 285-294.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Real-time metabolic tracking methodologies across clinical production workflows.
- Single-use sensor integration limitations within legacy cleanroom architectures.
- Optical interface requirements for continuous non-destructive cell monitoring.
- Commercial stakes of automated batch release replacing manual extraction protocols.
- Competitive positioning among contract manufacturers adopting closed-loop testing infrastructure.
- Specific algorithmic approaches bridging physical sensors with centralized data historians.
- Regional facility expansion variations driven by localized regenerative medicine investments.
- Regulatory validation hurdles impeding widespread spectroscopic probe deployments.
Frequently Asked Questions
What is PAT in cell and gene therapy manufacturing?
Process analytical technology involves integrating inline sensors and software to continuously monitor critical quality attributes without halting production.
Why is PAT important in cell therapy bioreactors?
Continuous monitoring eliminates manual sampling errors and drastically reduces the risk of contamination in fragile autologous batches.
How are critical quality attributes monitored in CGT bioreactors?
Facilities utilize integrated optical probes and Raman spectroscopy to track glucose, lactate, and cell viability directly within the culture fluid.
Which analytics are used in cell therapy manufacturing?
Production suites rely heavily on multiplexed spectroscopy, metabolite analyzers, and automated cell counters to maintain precise environmental control.
What are the leading cell therapy bioreactor platforms?
Top choices generally feature closed-system designs with pre-integrated single-use sensors to minimize aseptic connection risks.
Can you explain the baseline demand for CGT bioreactor analytics platforms?
Revenue generated sits at USD 410 million for 2026, highlighting strong initial clinical deployment phases globally.
Who are the top vendors in cell therapy bioreactor PAT?
Leading suppliers shaping this sector include Sartorius, Cytiva, Thermo Fisher Scientific, and Repligen.
How big is the CGT bioreactor analytics space by 2036?
Projections indicate the overall valuation for these specialized systems will reach USD 1,290 million by 2036.
Which countries are growing fastest in CGT process analytics?
India expands at 14.6% while China follows closely at 13.8%, reflecting aggressive regional capacity expansion.
What speed of expansion is anticipated overall?
Sales are advancing at a CAGR of 12.1% during this measurement timeframe.
Which hardware format leads adoption?
Integrated bioreactors maintain 34.0% share due to severe facility cleanroom space limitations.
Which analytical technique dominates usage?
Spectroscopy commands 28.0% share because it offers non-destructive continuous measurement capabilities.
Where does most implementation occur?
Process development captures 41.0% share as researchers define critical operational thresholds early before commercialization.
What treatment category drives equipment purchases?
Cell therapy represents 58.0% share reflecting high volumes of highly variable autologous patient materials.
Who makes the primary purchasing decisions?
Biopharma developers account for 46.0% share to protect proprietary manufacturing advantages natively.
What causes batch failures manually?
Periodic offline extraction exposes fragile cellular materials directly to catastrophic contamination events.
How do facilities solve manual risks?
Implementing integrated testing platforms completely eliminates dangerous physical sampling breaches.
Why do some optical sensors fail?
Certain probe designs compromise sterile barriers or create excessive fluid shear stress on sensitive cells.
What blocks broad software integration?
Connecting modern optical sensors to outdated legacy data historians frequently causes synchronization errors.
How does multiplexed tracking function?
Optical signatures identify multiple metabolic changes simultaneously without requiring any physical fluid extraction.
Why is scale-down modeling critical?
Testing extreme variations in small automated formats prevents wasting phenomenally expensive clinical-grade media.
What happens if companies ignore continuous monitoring?
Failing to automate parameter tracking dramatically increases per-dose production costs and delays regulatory filings.
Do contract organizations prefer proprietary probes?
Supply chain directors actually demand open-architecture readers accepting diverse single-use designs from multiple vendors.
How do European guidelines impact adoption?
Regulatory agencies strongly encourage implementing automated closed-loop models for streamlined commercial release.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Platform Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Platform Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Platform Type , 2026 to 2036
- Integrated Bioreactors
- Control Software
- Others
- Integrated Bioreactors
- Y to o to Y Growth Trend Analysis By Platform Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Platform Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Analytics Modality
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Analytics Modality, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Analytics Modality, 2026 to 2036
- Spectroscop
- Metabolite Analyzers
- Others
- Spectroscop
- Y to o to Y Growth Trend Analysis By Analytics Modality, 2021 to 2025
- Absolute $ Opportunity Analysis By Analytics Modality, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Workflow Stage
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Workflow Stage, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Workflow Stage, 2026 to 2036
- Process Development
- Clinical Manufacturing
- Others
- Process Development
- Y to o to Y Growth Trend Analysis By Workflow Stage, 2021 to 2025
- Absolute $ Opportunity Analysis By Workflow Stage, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Therapy Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Therapy Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Therapy Type, 2026 to 2036
- Cell Therapy
- Gene Therapy
- Cell Therapy
- Y to o to Y Growth Trend Analysis By Therapy Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Therapy Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Biopharma Developers
- CDMOs
- Others
- Biopharma Developers
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Platform Type
- By Analytics Modality
- By Workflow Stage
- By Therapy Type
- By End User
- Competition Analysis
- Competition Deep Dive
- Sartorius
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Cytiva
- Thermo Fisher Scientific
- Repligen
- Getinge
- PBS Biotech
- Sartorius
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Platform Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Analytics Modality, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Therapy Type, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Platform Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Analytics Modality, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Therapy Type, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Platform Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Analytics Modality, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Therapy Type, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Platform Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Analytics Modality, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Therapy Type, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Platform Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Analytics Modality, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Therapy Type, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Platform Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Analytics Modality, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Therapy Type, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Platform Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Analytics Modality, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Therapy Type, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Platform Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Analytics Modality, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Therapy Type, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Platform Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Platform Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Platform Type
- Figure 6: Global Market Value Share and BPS Analysis by Analytics Modality, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Analytics Modality, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Analytics Modality
- Figure 9: Global Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Workflow Stage
- Figure 12: Global Market Value Share and BPS Analysis by Therapy Type, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Therapy Type, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Therapy Type
- Figure 15: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by End User
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Platform Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Platform Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Platform Type
- Figure 32: North America Market Value Share and BPS Analysis by Analytics Modality, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Analytics Modality, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Analytics Modality
- Figure 35: North America Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Workflow Stage
- Figure 38: North America Market Value Share and BPS Analysis by Therapy Type, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Therapy Type, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Therapy Type
- Figure 41: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by End User
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Platform Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Platform Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Platform Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Analytics Modality, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Analytics Modality, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Analytics Modality
- Figure 51: Latin America Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Workflow Stage
- Figure 54: Latin America Market Value Share and BPS Analysis by Therapy Type, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Therapy Type, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Therapy Type
- Figure 57: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by End User
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Platform Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Platform Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Platform Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Analytics Modality, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Analytics Modality, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Analytics Modality
- Figure 67: Western Europe Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Workflow Stage
- Figure 70: Western Europe Market Value Share and BPS Analysis by Therapy Type, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Therapy Type, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Therapy Type
- Figure 73: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by End User
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Platform Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Platform Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Platform Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Analytics Modality, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Analytics Modality, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Analytics Modality
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Workflow Stage
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Therapy Type, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Therapy Type, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Therapy Type
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by End User
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Platform Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Platform Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Platform Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Analytics Modality, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Analytics Modality, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Analytics Modality
- Figure 99: East Asia Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Workflow Stage
- Figure 102: East Asia Market Value Share and BPS Analysis by Therapy Type, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Therapy Type, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by Therapy Type
- Figure 105: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by End User
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Platform Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Platform Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Platform Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Analytics Modality, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Analytics Modality, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Analytics Modality
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Workflow Stage
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Therapy Type, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Therapy Type, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Therapy Type
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Platform Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Platform Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Platform Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Analytics Modality, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Analytics Modality, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Analytics Modality
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Workflow Stage
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Therapy Type, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Therapy Type, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Therapy Type
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

