The B-cell depletion therapy market is projected at USD 26.7 billion in 2026, expanding to USD 62.6 billion by 2036 at a CAGR of 8.9%. Anti-CD20 monoclonal antibodies hold a 70% share of the product segment, primarily used for treating autoimmune diseases at 60%. Hospital infusion centers account for 55% of the end-use segment, with specialty pharmacies and other care facilities accounting for the remainder. Key players include Roche, Novartis, Biogen, GSK, and Johnson & Johnson.
Market growth is supported by the rising prevalence of autoimmune and hematologic conditions, alongside the broader adoption of next-generation B-cell therapies. Increasing hospital infusion capacities and specialty pharmacy networks enhance patient access. Research advancements and pipeline therapies are expected to contribute to sustained expansion. Demand for personalized dosing regimens, combination therapies, and safer immunomodulatory options also drives the market forward, making it a high-growth segment in immunotherapy.
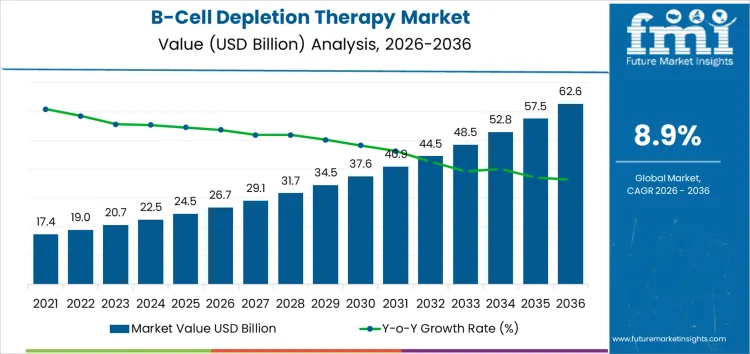
From 2026 to 2031, the B-cell depletion therapy market grows from USD 26.7 billion to approximately USD 37.7 billion, representing the early adoption phase. Annual increments rise from USD 2.2 billion in 2026 to USD 3.1 billion in 2031. Growth is driven by adoption in autoimmune diseases, oncology, and hematology indications where B-cell targeted therapies improve patient outcomes. North America and Europe account for roughly 65% of early adoption, supported by clinical trial validation, regulatory approvals, and integration into hospital and specialty clinic protocols. Emerging adoption in Asia Pacific is concentrated in high-volume urban hospitals and specialty care centers.
Between 2031 and 2036, the market expands from roughly USD 37.7 billion to USD 62.6 billion, illustrating the late adoption phase characterized by broad commercialization and structural penetration. Annual increments increase from USD 3.1 billion to USD 4.6 billion, driven by repeat treatment cycles, multi-region adoption, and portfolio-wide integration of B-cell depletion therapies. Growth is reinforced by increasing prevalence of autoimmune and hematologic conditions, expanded indications, and normalized adoption in hospitals and specialty clinics. Early-stage growth relied on selective high-value institutions and trial-based deployments, while later expansion reflects scale deployment and global standard-of-care adoption.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 26.7 billion |
| Forecast Value (2036) | USD 62.6 billion |
| Forecast CAGR (2026 to 2036) | 8.9% |
Demand for B-cell depletion therapies initially emerged from clinical limitations of earlier treatment approaches for autoimmune diseases and certain B-cell driven malignancies. Standard immunosuppressive drugs modulated immune activity broadly but often lacked specificity, resulting in incomplete disease control or adverse systemic effects. Researchers identified B lymphocytes as central drivers in conditions such as rheumatoid arthritis, systemic lupus erythematosus, and certain non-Hodgkin lymphomas. Targeted depletion of B cells offered a way to reduce pathogenic antibody production and aberrant immune signalling while sparing non-pathogenic immune functions. Early use was concentrated in patient’s refractory to conventional therapies, where the risk-benefit profile justified adoption in specialised clinical settings rather than general practice.
Future demand for B-cell depletion therapies is being shaped by evolving clinical evidence, expanded indication approvals, and integration into treatment guidelines rather than by isolated use in refractory cases alone. Clinical trials demonstrating sustained remission, improved quality of life, and manageable safety profiles are supporting broader application in earlier disease stages and additional autoimmune and hematologic conditions. Regulatory agencies are recognising differentiated mechanisms within the B-cell depletion class, leading to indication expansions for newer agents with distinct binding targets or depletion kinetics. As guideline bodies incorporate these agents into standard of care recommendations, demand will expand beyond specialist centres to mainstream clinical practice.
The B cell depletion therapy market is defined by mature biologics, chronic disease demand, and conservative prescribing frameworks. Established antibodies dominate due to evidence depth and reimbursement familiarity. Autoimmune indications drive volume through long term management requirements. Innovation continues, but adoption remains measured, favoring validated therapies over rapid shifts. Market structure reflects stability, retreatment frequency, and guideline embedded use rather than episodic oncology intensity within regulated healthcare systems prioritizing predictability, safety, and long term population outcomes globally across regions today.
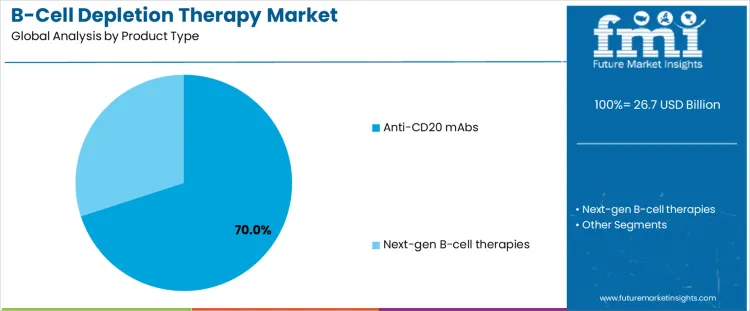
Anti CD20 monoclonal antibodies account for about 70% of demand because they established clinical efficacy across multiple B cell mediated conditions over more than two decades. These therapies provide predictable depletion kinetics, well understood safety profiles, and standardized dosing protocols, which supports broad physician confidence. Payers and treatment guidelines favor them due to extensive outcome data and comparability across indications. Manufacturing scale, biosimilar availability, and mature supply chains further reinforce dominance.
Next generation B cell therapies attract interest through improved specificity, alternative targets, or enhanced durability, but adoption remains selective due to limited long term evidence and higher development risk. Their use is often confined to refractory patients or clinical trials. Product type leadership therefore reflects risk tolerance, accumulated evidence, and reimbursement familiarity rather than innovation pace. As treatment algorithms emphasize reliability and population scale deployment, established anti CD20 antibodies continue to anchor prescribing behavior while newer modalities progress gradually through validation and access pathways. This balance persists as clinicians prioritize consistent response rates, manageable monitoring requirements, and predictable retreatment schedules, factors that favor incumbent antibodies in routine practice while limiting rapid displacement by emerging approaches lacking equivalent longitudinal performance assurance across healthcare systems with constrained budgets and conservative oversight.
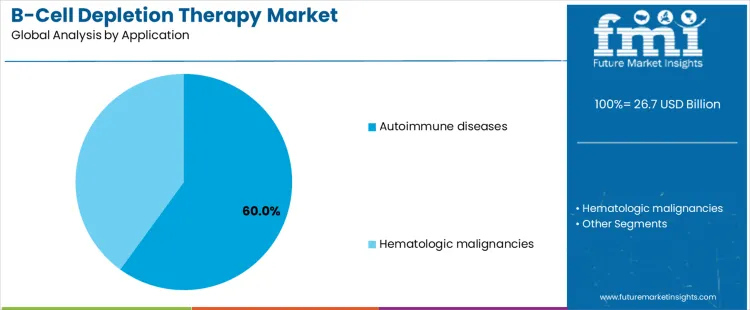
Autoimmune diseases represent about 60% of demand because B cell depletion has demonstrated durable benefit in conditions driven by aberrant antibody production and immune activation. Clinical protocols for diseases such as rheumatoid arthritis and multiple sclerosis incorporate B cell therapies as established treatment options, supporting repeat utilization. Chronic disease prevalence ensures sustained therapy cycles, reinforcing volume demand. Hematologic malignancies also rely on B cell targeting, but treatment courses are episodic and often combined with chemotherapy or cellular therapies, limiting relative share. Oncology adoption is influenced by rapidly evolving standards and competitive modalities.
Application leadership therefore reflects chronicity, retreatment frequency, and guideline stability rather than severity alone. Autoimmune indications favor long term maintenance strategies, predictable monitoring, and outpatient administration, aligning with broad system capacity. Malignancy use remains essential but more variable, shaped by disease stage, combination regimens, and emerging alternatives. This dynamic concentrates demand in autoimmune care pathways where consistency and longitudinal management outweigh episodic intensive interventions. Health system planning, reimbursement continuity, and patient adherence further reinforce autoimmune dominance, while oncology pathways face periodic disruption from innovation cycles, trial readouts, and shifting standards that redistribute utilization across competing therapeutic classes over time within regional formularies and specialty care networks globally today.
Use appears in hospitals, specialty clinics, and infusion centers where modulation of B-cell activity is critical for treating diseases such as multiple sclerosis, rheumatoid arthritis, and certain hematologic malignancies. Monoclonal antibodies targeting CD20 or related B-cell markers are administered to reduce aberrant immune responses or tumor proliferation. Physicians adopt therapy protocols to balance efficacy with patient safety, often in combination with other immunomodulatory treatments. These applications reflect clinical and operational priorities rather than aesthetic objectives, with adoption driven by disease severity, therapeutic effectiveness, and patient-specific considerations.
Selection aligns with treatment centers capable of providing intravenous administration, patient monitoring, and adherence to immunotherapy protocols. Pharmaceutical-grade monoclonal antibodies require cold chain storage, handling, and preparation to maintain potency. Clinical teams monitor immunosuppression, infection risk, and infusion reactions, adjusting dosing schedules as needed. Regulatory approvals, guideline compliance, and hospital formulary inclusion influence therapy deployment. These conditions emerge from operational safety, therapeutic precision, and regulatory adherence priorities in structured healthcare workflows.
High drug cost and administration expenses can restrict access in low-resource settings. Risk of immunosuppression and infection necessitates careful patient screening and monitoring. Long-term efficacy and adverse effect profiles vary by indication, affecting therapy choice. Cold chain and specialized infusion infrastructure are required for safe handling. These factors lead to selective adoption where clinical efficacy, patient safety, and healthcare infrastructure justify incremental cost and operational management.
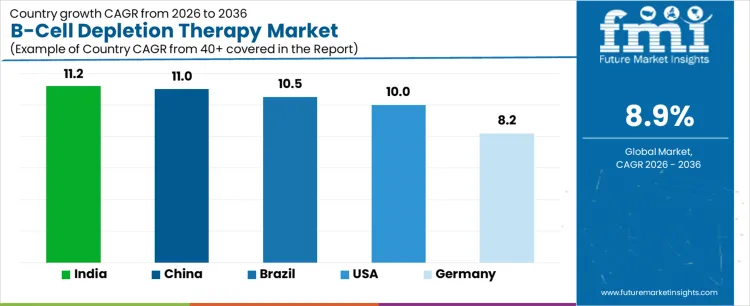
| Country | CAGR (%) |
|---|---|
| USA | 10.0% |
| Brazil | 10.5% |
| China | 11.0% |
| India | 11.2% |
| Germany | 8.2% |
The demand for B cell depletion therapy varies across countries, driven by rising autoimmune disease prevalence, expanding healthcare infrastructure, and adoption of advanced immunotherapies. India leads with an 11.2% CAGR, supported by increasing cases of autoimmune disorders, growing awareness, and enhanced access to targeted therapies. China follows at 11.0%, driven by hospital modernization, rising healthcare expenditure, and adoption of monoclonal antibody therapies. Brazil records 10.5%, shaped by expanding healthcare services and patient access programs. The USA grows at 10.0%, reflecting established healthcare infrastructure and high adoption of biologic therapies. Germany posts 8.2%, supported by mature healthcare systems and steady use of B cell targeted treatments.
In the United States, revenue from the B-Cell Depletion Therapy Market is expanding at a CAGR of 10% through 2036, driven by increasing adoption of monoclonal antibodies and biologic therapies for autoimmune disorders such as rheumatoid arthritis, multiple sclerosis, and lupus. Hospitals, specialty clinics, and research centers are integrating B-cell depletion therapies to target pathogenic B cells, improve patient outcomes, and reduce disease progression. Demand is concentrated in academic hospitals, specialty treatment centers, and outpatient clinics. Domestic manufacturers are supplying high-performance, clinically validated therapeutics compatible with standard infusion and administration protocols. Rising autoimmune disease prevalence, regulatory approvals, and recurring treatment cycles are sustaining predictable adoption nationwide.
In Brazil, the market is growing at a CAGR of 10.5% through 2036, supported by increasing awareness of autoimmune conditions and expansion of specialty care programs. Hospitals and clinics are adopting B-cell depletion therapies to manage chronic autoimmune diseases and enhance patient quality of life. Demand is concentrated in urban hospitals, specialty clinics, and research institutions. Local manufacturers and distributors provide validated biologics compatible with infusion protocols and clinical standards. Growth in autoimmune patient populations, government healthcare initiatives, and recurring treatment schedules are sustaining adoption nationwide.
China is witnessing growth at a CAGR of 11% through 2036, driven by rapid expansion of hospitals, specialty clinics, and autoimmune treatment programs. Manufacturers are integrating B-cell depletion therapies to provide targeted treatment for chronic autoimmune diseases, improving clinical outcomes and reducing disease progression. Demand is concentrated in tertiary hospitals, urban specialty centers, and research institutions. Domestic suppliers are scaling production of high-performance monoclonal antibodies compatible with infusion and administration systems. Expanding healthcare infrastructure, patient awareness, and recurring therapy cycles are sustaining rapid adoption nationwide.
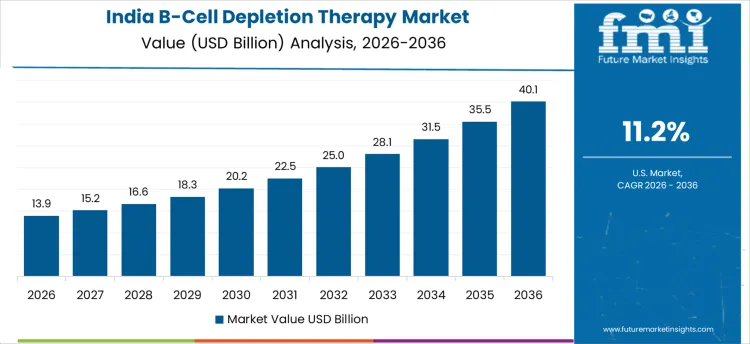
In India, revenue from the B-Cell Depletion Therapy Market is expanding at a CAGR of 11.2% through 2036, supported by increasing autoimmune disease prevalence and adoption of biologic therapies. Hospitals and specialty clinics are using B-cell depletion treatments to manage chronic conditions, improve patient outcomes, and support long-term care strategies. Demand is concentrated in urban hospitals, specialty centers, and outpatient care facilities. Domestic suppliers provide high-quality, clinically validated biologics compatible with standard infusion protocols. Rising patient awareness, healthcare infrastructure expansion, and recurring therapy cycles are sustaining robust adoption nationwide.
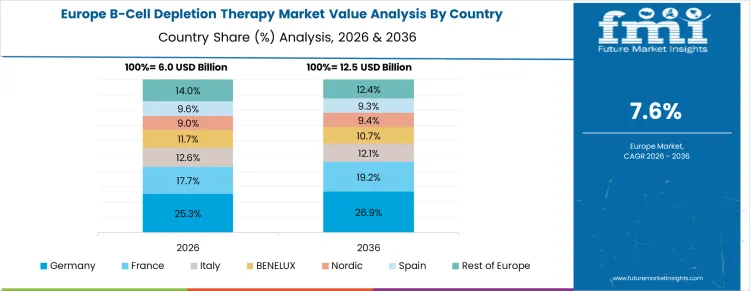
Germany is witnessing growth at a CAGR of 8.2% through 2036, driven by adoption of B-cell depletion therapies in hospitals and specialty clinics for autoimmune disorder management. Manufacturers are integrating monoclonal antibodies to target B cells, improve patient outcomes, and support long-term disease management. Demand is concentrated in academic hospitals, specialty centers, and research institutions. Domestic suppliers provide high-performance, clinically validated biologics compatible with infusion and administration systems. Regulatory standards, autoimmune treatment protocols, and recurring therapy cycles are sustaining consistent procurement nationwide.
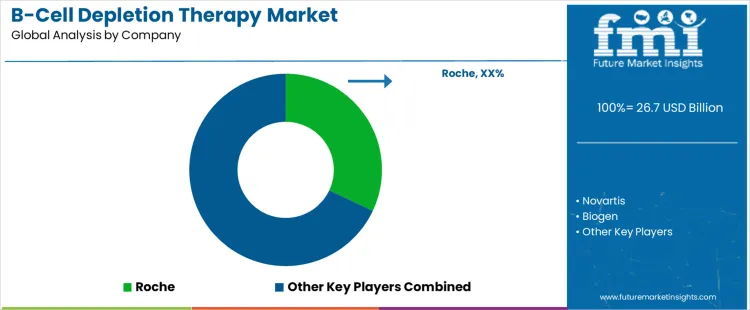
Key players in the B cell depletion therapy market compete by shaping how their therapies are embedded within long term treatment strategies rather than by pursuing rapid product turnover. Roche relies on lifecycle extension strategies, including formulation improvements, dosing flexibility, and broad label maintenance, to defend entrenched clinical positions. Novartis and Biogen focus on selective indication targeting, advancing therapies in disease areas where relapse management, durability of response, or neurological involvement create room for differentiation. Clinical trial design is used strategically to demonstrate advantages in specific patient subgroups rather than across broad populations. Companies prioritize alignment with treatment guidelines and specialist prescribing behavior, recognizing that early positioning within care pathways limits future displacement.
Another core strategy centers on healthcare system alignment and risk management. GSK and Johnson and Johnson emphasize administration models that reduce burden on infusion centers, support outpatient delivery, or lower long term monitoring requirements. Investment in real world evidence, safety registries, and post marketing studies is used to reinforce payer and clinician confidence, particularly as biosimilar competition increases. Across the market, companies pursue incremental innovation that preserves regulatory continuity while expanding use duration and patient eligibility. Partnerships, co-development, and licensing are favored over aggressive head to head competition. Strategic success depends on reimbursement resilience, physician trust, and sustained outcomes across chronic treatment horizons rather than short term clinical differentiation.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD billion |
| Product Type | Anti-CD20 monoclonal antibodies, Next-gen B-cell therapies |
| Application | Autoimmune diseases, Hematologic malignancies, Others |
| End-Use Industry | Hospital infusion centers, Specialty pharmacies |
| Region | Asia Pacific (China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Rest of Asia Pacific), Europe (Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, Rest of Europe), North America (United States, Canada, Mexico), Latin America (Brazil, Chile, Rest of Latin America), Middle East & Africa (KSA, Other GCC Countries, Turkey, South Africa, Other African Union, Rest of Middle East & Africa) |
| Key Companies Profiled | Roche, Novartis, Biogen, GSK, Johnson & Johnson, Others |
| Additional Attributes | Dollar by sales by product type, Dollar by sales by application, Dollar by sales by end-user, Dollar by sales by region, Annual treatment cycles, Average infusion duration, Dosing precision and regimen frequency, Biologic storage and cold chain requirements, Infusion center capacity, Safety and monitoring protocols, Real-world efficacy and patient adherence, Repeat treatment scheduling, Regulatory approvals and guideline inclusion, Treatment protocol integration, Combination therapy compatibility, Expansion into emerging regions, Hospital formulary and payer coverage, Operational efficiency of infusion workflows, Patient population access and awareness |
How big is the b-cell depletion therapy market in 2026?
The global b-cell depletion therapy market is estimated to be valued at USD 26.7 billion in 2026.
What will be the size of b-cell depletion therapy market in 2036?
The market size for the b-cell depletion therapy market is projected to reach USD 62.6 billion by 2036.
How much will be the b-cell depletion therapy market growth between 2026 and 2036?
The b-cell depletion therapy market is expected to grow at a 8.9% CAGR between 2026 and 2036.
What are the key product types in the b-cell depletion therapy market?
The key product types in b-cell depletion therapy market are anti-cd20 mabs and next-gen b-cell therapies.
Which application segment to contribute significant share in the b-cell depletion therapy market in 2026?
In terms of application, autoimmune diseases segment to command 60.0% share in the b-cell depletion therapy market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.