Closed System Bioprocessing Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
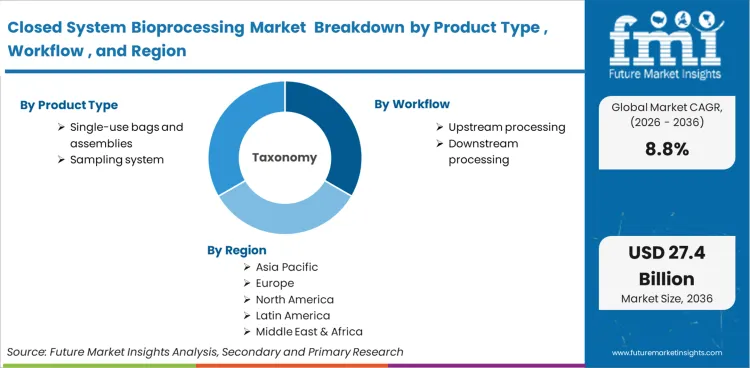
The Closed System Bioprocessing Market is segmented by Product Type (Single-use Bags and Assemblies, Aseptic Connectors and Disconnectors, Filtration Devices, Single-use Bioreactors and Mixers, Sampling Systems, Sensors and Control Accessories, and Others), Workflow (Upstream Processing, Downstream Processing, and Fill-finish and Aseptic Transfer), End User (Biopharmaceutical Manufacturers, CDMOs and CMOs, and Academic and Research Institutes), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Closed System Bioprocessing Market Size and Share Forecast Outlook By FMI
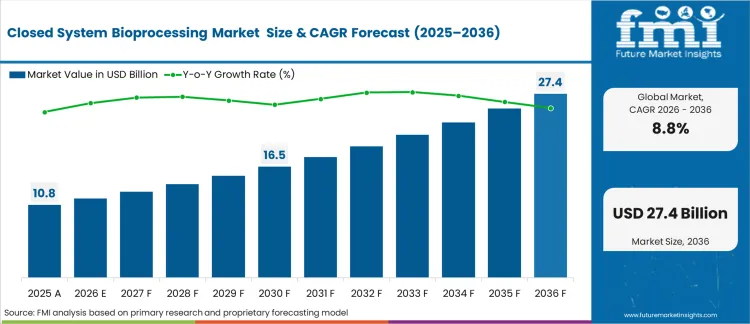
The closed system bioprocessing market was valued at USD 10.9 billion in 2025. The market is projected to reach USD 11.8 billion in 2026 and USD 27.4 billion by 2036, expanding at a CAGR of 8.8% during the forecast period. Single-use bags and assemblies are expected to lead product type with a 31.4% share in 2026. Upstream processing is projected to remain the leading workflow with a 46.7% share in 2026. Biopharmaceutical manufacturers are expected to lead end-user demand with a 57.9% share in 2026.
Summary of Closed System Bioprocessing Market
- Key Drivers
- Contamination-control pressure: EU GMP Annex 1 and FDA aseptic guidance continue to favor closed systems because they reduce intervention risk and strengthen sterile processing control.
- Biologics and advanced-therapy growth: Expansion in biologics, biosimilars, and cell and gene therapies is increasing demand for cleaner and more flexible manufacturing architectures.
- Efficiency and productivity gains: Closed single-use systems improve batch changeover, reduce cleaning requirements, and help facilities run multi-product operations more efficiently.
- Key Segments Analyzed in the Report
- Product type: Single-use bags and assemblies, aseptic connectors and disconnectors, filtration devices, single-use bioreactors and mixers, sampling systems, sensors and control accessories, others
- Workflow: Upstream processing, downstream processing, fill-finish and aseptic transfer
- End user: Biopharmaceutical manufacturers, CDMOs and CMOs, academic and research institutes
- Region: North America, Latin America, Europe, East Asia, South Asia Pacific, Middle East and Africa
- Analyst Opinion
- Sabyasachi Ghosh, Principal Consultant at FMI says, “Closed system bioprocessing looks attractive, but the market does not reward broad catalog depth alone. In practice, that means the strongest value pools sit in recurring assemblies, sterile transfer, filtration, and application-specific closed workflows rather than in generic hardware alone.”
Closed System Bioprocessing Market Definition
The closed system bioprocessing market includes equipment, consumables, and sterile-transfer components used to handle biologic materials within sealed or functionally closed process pathways. It covers single-use bags and assemblies, tubing manifolds, aseptic connectors, filtration devices, closed mixing systems, single-use bioreactors, sampling systems, and related accessories used in biopharmaceutical manufacturing.
Closed System Bioprocessing Market Inclusions
- Closed and functionally closed systems used in upstream, downstream, and aseptic transfer operations
- Revenue from consumables, process-contact hardware, and closed fluid-management components sold into biopharma, CDMO, and GMP manufacturing environments
- Demand across North America, Latin America, Europe, East Asia, South Asia & Pacific, and the Middle East & Africa
Closed System Bioprocessing Market Exclusions
- Open bioprocess handling equipment, except as a benchmark
- Finished biologic drug sales
- General lab plastics, non-GMP research disposables, and packaging products not primarily used in bioprocessing
Closed System Bioprocessing Market Research Methodology
This study evaluates the closed system bioprocessing market by product type, workflow, end user, and region, with 2025 as the base year and 2026 to 2036 as the forecast period. Market sizing is expressed in value terms. Evidence inputs include aseptic-processing regulations, biologics manufacturing signals, public company filings, process-solution revenue disclosures, filtration and single-use product launches, and technical literature on contamination control and process intensification. The final estimates are modelled through triangulation across vendor revenue anchors, modality growth, contamination-control requirements, recurring consumables intensity, and the growing share of biologics workflows that favor sealed handling over open processing.
Key Drivers, Restraints, and Trends in Closed System Bioprocessing Market
Drivers
Tighter contamination control requirements in biologics manufacturing are pushing producers toward closed processing environments. Exposure risk during upstream and downstream operations remains a central concern, particularly in high-value biologic production. Regulators continue to place strong emphasis on aseptic processing, which reinforces the shift toward sealed and functionally closed workflows within GMP facilities. Operational efficiency is another factor. Closed configurations reduce manual intervention, limit cleaning cycles, and allow faster batch changeovers between production runs. CDMOs are expanding manufacturing capacity across biologics, vaccines, and cell-based therapies, which increases demand for modular and scalable process platforms. The market also benefits from the recurring nature of consumables. Items such as bags, connectors, filters, and assemblies generate continuous demand across production cycles.
Restraints
Despite strong adoption momentum, several operational constraints remain. Supply reliability is a persistent concern because manufacturers rely on uninterrupted availability of validated consumables and components. Even short disruptions can affect production schedules. Vendor switching is also slow. Extractables and leachables assessments, documentation requirements, and qualification processes make supplier transitions complex and time-consuming. Cost pressure can emerge as well. Single-use systems introduce recurring consumable expenses that are more visible for smaller manufacturers operating with tighter budgets. Integration issues may appear when facilities use components sourced from multiple vendors, particularly when compatibility across process steps is not seamless. Many production sites still operate legacy open or semi-open systems. Converting these environments to closed architectures often requires site-specific validation work and operator retraining.
Trends
The structure of the market is gradually evolving. Buyers are moving away from isolated single-use products toward integrated closed process trains that support continuity across manufacturing steps. Suppliers are adjusting their positioning accordingly. Instead of selling individual components, many now emphasize end-to-end workflow solutions that combine equipment, consumables, and technical support. Commercial dynamics are also shifting. Recurring consumables are becoming more important to revenue stability than capital equipment sales in several workflows. Closed system design is gaining relevance in advanced therapy manufacturing, where sterility control and process flexibility are critical. Competitive advantage increasingly rests with suppliers capable of integrating sterile transfer, filtration, mixing, and process validation support within a unified offering.
Segmental Analysis
Closed System Bioprocessing Market Analysis by Product Type
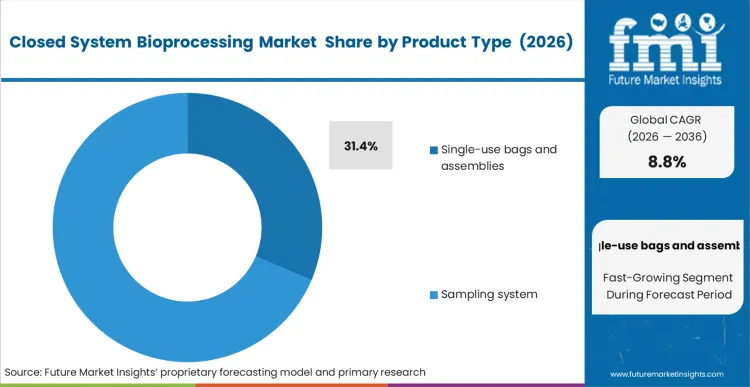
Single-use bags and assemblies sit at the center of the product mix and are likely to hold 31.4% of global market value in 2026. That lead comes from replacement frequency and process intimacy. These components are used repeatedly across media preparation, inoculation, hold, transfer, harvest, and formulation steps. The economic logic is stronger than for capital equipment. A bioreactor or mixer may anchor a line, but recurring assemblies capture spending every time a batch moves through the plant. Sartorius explicitly described consumables such as filters and single-use bags as the main driver of divisional growth in 2025.
Closed System Bioprocessing Market Analysis by Workflow
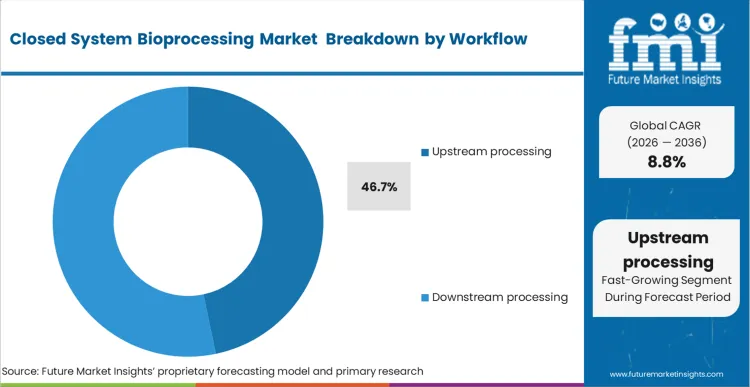
Upstream processing is set to account for 46.7% of global revenue in 2026. This reflects where closed architecture solves immediate operational problems. Cell expansion, media transfer, seed train management, and culture operations benefit from reduced contamination exposure, lower cleaning load, and faster turnaround between campaigns. Thermo Fisher’s single-use materials and sustainability papers repeatedly position upstream systems around changeover speed, lower cleaning burden, and closed system processing capabilities.
Competitive Aligners for Market Players
Breadth alone is not a durable advantage in the closed system bioprocessing market. Buyers need validated assemblies, supplier documentation, extractables support, filtration performance, reliable lead times, and application guidance that survives tech transfer. The strongest firms therefore combine recurring consumables with workflow engineering and regulatory confidence. This is one reason recurring revenue matters so much in the sector. Danaher highlights high recurring revenue in its model, and Sartorius links divisional momentum to consumables rather than equipment alone.
Competitive advantage is also moving toward end-to-end fit. Thermo Fisher strengthened its filtration footprint through the Solventum purification and filtration transaction, while Merck has expanded its viral vector and ADC-oriented process offering through Mirus Bio and the Mobius ADC Reactor. Market leadership is being built by firms that can align sterile transfer, filtration, mixing, and process support into one coherent operating system for biologics plants.
Key Players in Closed System Bioprocessing Market
- Thermo Fisher Scientific
- Danaher Corporation
- Sartorius AG
- Merck KGaA
- Repligen Corporation
- Saint-Gobain Life Sciences
- Avantor
- Eppendorf SE
- Corning Incorporated
Strategic Outlook by FMI on Closed System Bioprocessing Market
Closed system bioprocessing is moving from a contamination-control choice to a core manufacturing design principle in biologics. The market’s next phase will be shaped less by generic single-use adoption and more by how deeply suppliers can integrate sterile transfer, filtration, upstream efficiency, and downstream process continuity into one validated operating architecture. The companies most likely to strengthen their position are those that combine recurring consumables, regulatory confidence, and workflow-specific technical support rather than relying on product breadth alone.
Scope of the Report
| Metric | Value |
|---|---|
| Market Value | USD 11.8 billion in 2026 to USD 27.4 billion by 2036 |
| CAGR | 8.8% from 2026 to 2036 |
| Base Year | 2025 |
| Forecast Period | 2026 to 2036 |
| Product Type Segmentation | Single-use bags and assemblies, aseptic connectors and disconnectors, filtration devices, single-use bioreactors and mixers, sampling systems, sensors and control accessories, others |
| Workflow Segmentation | Upstream processing, downstream processing, fill-finish and aseptic transfer |
| End User Segmentation | Biopharmaceutical manufacturers, CDMOs and CMOs, academic and research institutes |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia Pacific, Middle East and Africa |
Bibliography
- European Commission. 2022. EU GMP Annex 1: Manufacture of Sterile Medicinal Products.
- U.S. Food and Drug Administration. Guidance for Industry: Sterile Drug Products Produced by Aseptic Processing.
- European Medicines Agency. 2026. Human Medicines in 2025.
- Danaher. 2026. Danaher Overview 2025.
- Sartorius. 2026. Preliminary Results 2025 and Annual Report 2025.
- Thermo Fisher Scientific. 2024. Single-use Bioprocessing Systems: Greener by Design.
- Merck KGaA. 2024. Closes Mirus Bio Acquisition, Bolstering Viral Vector Bioprocessing Offering.
Frequently Asked Questions
How large is the closed system bioprocessing market in 2025?
The global closed system bioprocessing market is estimated at USD 10.9 billion in 2025.
What will be the closed system bioprocessing market size by 2036?
The closed system bioprocessing market is projected to reach USD 27.4 billion by 2036.
What is the expected growth rate during the forecast period?
The closed system bioprocessing market is projected to expand at a CAGR of 8.8% from 2026 to 2036.
Which product type leads the closed system bioprocessing market?
Single-use bags and assemblies lead the market with a 31.4% share in 2026.
Which workflow is dominant in the closed system bioprocessing market?
Upstream processing is the leading workflow with a 46.7% share in 2026.
Which end-user segment contributes the largest share in the closed system bioprocessing market?
Biopharmaceutical manufacturers are the largest end-user segment with a 57.9% share in 2026.
Which region is the largest market for closed system bioprocessing?
North America is the largest regional market for closed system bioprocessing.
Which region grows the fastest in the closed system bioprocessing market?
East Asia records the fastest regional growth through 2036.
What is the main structural shift in the closed system bioprocessing market?
The biggest structural shift is the move from partial single-use adoption toward broader closed, validated process trains that cover upstream, downstream, and sterile transfer steps.
Why do recurring consumables matter so much in the closed system bioprocessing market?
They sit in the sterile fluid path, are replaced repeatedly, and carry a high compliance burden, which makes them commercially heavier than capital equipment in many workflows.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Single-use bags and assemblies
- Sampling system
- Single-use bags and assemblies
- Y-o-Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Workflow
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Workflow, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Workflow, 2026 to 2036
- Upstream processing
- Downstream processing
- Upstream processing
- Y-o-Y Growth Trend Analysis By Workflow, 2021 to 2025
- Absolute $ Opportunity Analysis By Workflow, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Biopharmaceutical manufacturers
- CDMOs and CMOs
- Biopharmaceutical manufacturers
- Y-o-Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Workflow
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Workflow
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Workflow
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Workflow
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Workflow
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Workflow
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Workflow
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Workflow
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Workflow
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Workflow
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Workflow
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Workflow
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Workflow
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Workflow
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Workflow
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Workflow
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Workflow
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Workflow
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Workflow
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Workflow
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Workflow
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Workflow
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Workflow
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Workflow
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Workflow
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Workflow
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Workflow
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Workflow
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Workflow
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Workflow
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Workflow
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Workflow
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Workflow
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Workflow
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Workflow
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Workflow
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Workflow
- By End User
- Competition Analysis
- Competition Deep Dive
- Thermo Fisher Scientific
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Danaher Corporation
- Sartorius AG
- Merck KGaA
- Repligen Corporation
- Saint-Gobain Life Sciences
- Avantor
- Thermo Fisher Scientific
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Workflow, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Workflow, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Workflow, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Workflow, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Workflow, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Workflow, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Workflow, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Workflow, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Workflow, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Workflow, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Workflow
- Figure 9: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by End User
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Product Type
- Figure 26: North America Market Value Share and BPS Analysis by Workflow, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Workflow, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Workflow
- Figure 29: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by End User
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Product Type
- Figure 36: Latin America Market Value Share and BPS Analysis by Workflow, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Workflow, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by Workflow
- Figure 39: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by End User
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Product Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by Workflow, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Workflow, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by Workflow
- Figure 49: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by End User
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Workflow, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Workflow, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Workflow
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End User
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Product Type
- Figure 66: East Asia Market Value Share and BPS Analysis by Workflow, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Workflow, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by Workflow
- Figure 69: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by End User
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Workflow, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Workflow, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Workflow
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Workflow, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Workflow, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Workflow
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

