The mRNA vaccine formulation stability and stress test systems market crossed a valuation of USD 0.7 billion in 2025. Industry value is estimated to reach USD 0.8 billion in 2026 at a CAGR of 10.6% during the forecast period. Market is projected to rise USD 2.2 billion by 2036.
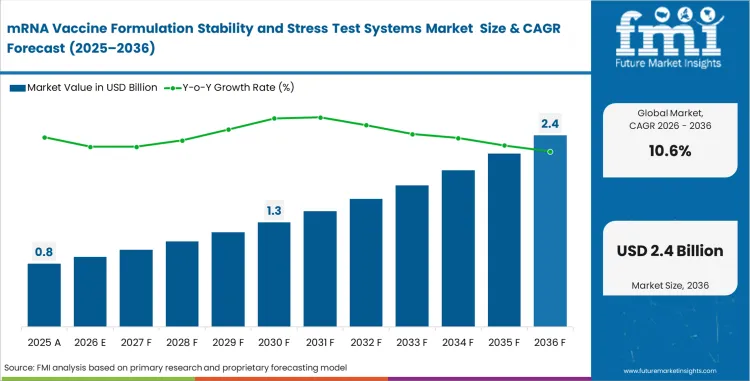
| Parameter | Details |
|---|---|
| Market value (2026) | USD 0.8 billion |
| Forecast value (2036) | USD 2.2 billion |
| CAGR (2026 to 2036) | 10.6% |
| Estimated market value (2025) | USD 0.7 billion |
| Leading formulation format | LNP dispersions |
| Formulation format share (2026) | 72% |
| Leading study type | Accelerated stability |
| Study type share (2026) | 28% |
| Leading workflow stage | Formulation screening |
| Workflow share (2026) | 30% |
| Leading readout type | Particle size |
| Readout share (2026) | 26% |
| Leading end user | Vaccine developers |
| End-user share (2026) | 35% |
| Fastest-growing country | India |
| India CAGR | 12.9% |
Source: Future Market Insights, 2026.
Greater attention now falls on whether the same formulation can tolerate handling storage repeat testing without creating variability. This is increasing demand for systems that can compare candidate formulations faster and support consistent assay results. Demand for mRNA synthesis raw materials and vaccine stabilizers is supporting this market because stability testing is shaping formulation decisions much earlier.
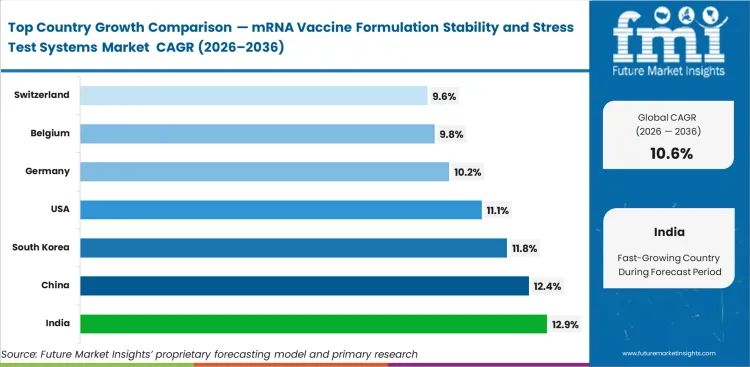
India is expected to post 12.9% CAGR in this market through 2036 as regional manufacturing capability keeps moving upward. China is likely to record 12.4% CAGR, supported by scale and local analytical depth. South Korea follows with 11.8% CAGR as platform development remains active. Industry outlook in the United States points to 11.1% CAGR from 2026 to 2036, while Germany is forecast to register 10.2% CAGR, Belgium is set to advance at a CAGR of 9.8%, and Switzerland is projected to grow at a CAGR of 9.6% during the assessment period.
The market covers the test systems that evaluate mRNA vaccine formulations during storage and stress testing. It focuses on tools that help development teams decide formulation stability and confirm whether test results are reliable for further development.
This market includes systems used to evaluate formulation condition and stability in mRNA vaccine development. It includes chromatography platforms and software that supports the testing process. It also includes systems used in accelerated stability studies and real-time storage evaluation. mRNA sequencing falls under the scope where it supports formulation interpretation. Solid lipid nanoparticles and freeze drying equipment is included where it supports formulation assessment.
This sector excluded upstream mRNA synthesis equipment and cold-chain transport assets. General laboratory tools are outside scope when they are not used for formulation stability or stress evaluation. Vaccine packaging systems and standard biologics manufacturing lines are excluded unless they directly support this testing function.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 0.8 billion |
| Industry Value (2036) | USD 2.2 billion |
| CAGR (2026-2036) | 10.6% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
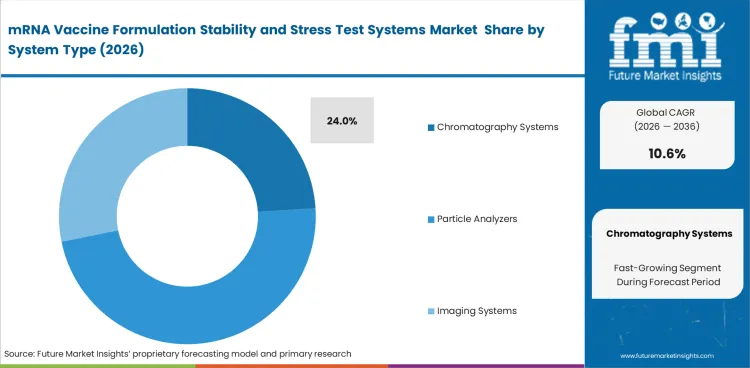
Chromatography systems are expected to account for 24% share in 2026 because they are used often in formulation assessment. Development teams need systems that can compare multiple formulation iterations without making each round harder to manage. This becomes more important when material is limited and each round of analysis must deliver clear results. Poor system fit can delay interpretation before broader formulation evaluation begins.
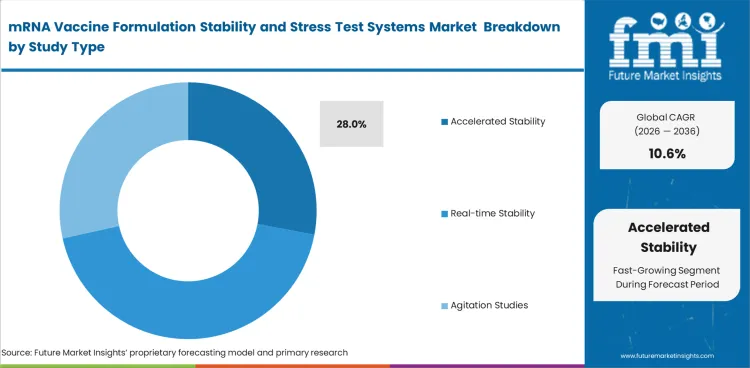
Accelerated stability leads because development teams use it to identify storage sensitivity and formulation weakness early. In 2026, accelerated stability is projected to contribute 28% of total market share. This study type helps screen out weaker formulations before broader analytical resources are used. Early stress signals often determine which formulation moves for deeper evaluation. Faster readouts support better coordination between formulation scientists and quality teams when storage concerns appear.
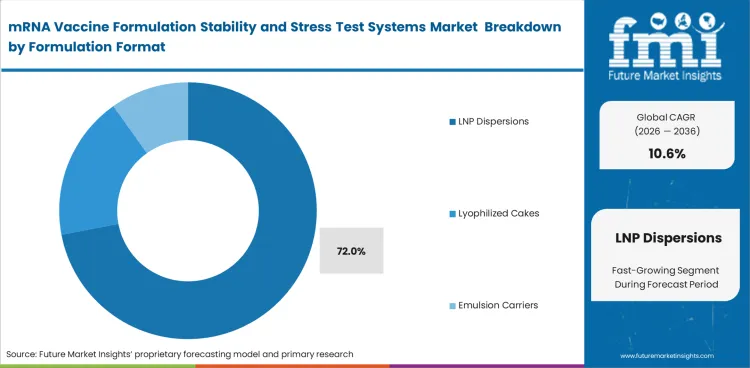
Lipid nanoparticle dispersions lead this market because system selection often starts with the need to access how LNP-based formulations respond to storage and stress. Alternative delivery approaches remain relevant, though they play a smaller role in current vaccine formulation practice. LNP dispersions are anticipated to represent 72% of the market in 2026. This reflects how often particle behavior and storage response must be assessed together.
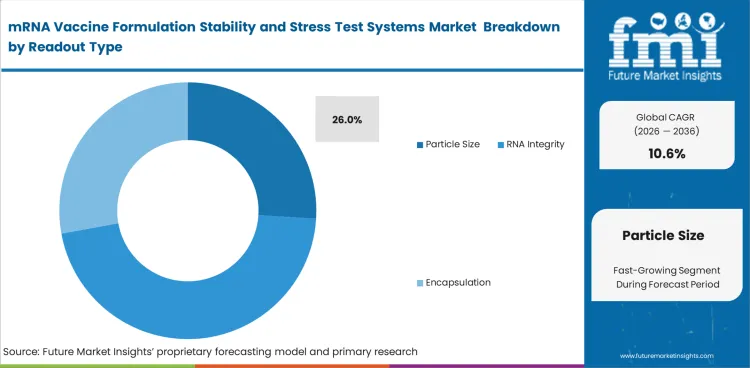
Particle size is likely to secure 26% share in 2026. It holds strong position because it provides an early signals on formulation consistency. Early readouts often shape multiple downstream decisions, especially when development groups assess many formulation options in parallel. Lead position comes from how frequently it is used as a first checkpoint before more detailed analytical work begins.
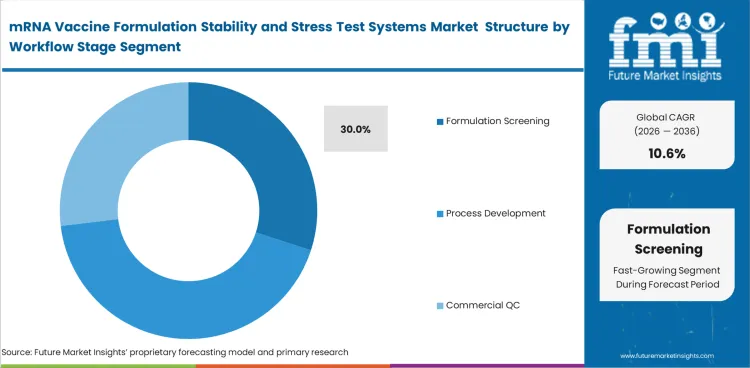
Development groups must compare excipient combinations and assay compatibility while the formulation changes are still manageable. Formulation screening is projected to account for 30% share in 2026. That reflects the amount of early decision work needed to narrow formulation options before analytic methods are fixed. Screening increases the need for throughput and interpretive speed at the same time.
Formulation stability has become a central part quality review. It affects release decisions and guides the response when temperature conditions fall outside the expected range after distribution. Current mRNA vaccine guidance ties this work closely to real-use risk because storage sensitivity and freeze-thaw exposure can affect batch acceptability. Weak stability support can lead to product loss or added clinical review. Market value comes from helping development and quality groups decide whether material can continue forward after a storage event.
EMA’s draft guideline explicitly allows platform technology and prior knowledge to support new targets. The broader 2025 clinical-trial quality guidance says platform knowledge can inform stability protocol design. Product relevance must be justified and verified with product-specific data. Commercially, stronger stress-test systems are the ones that help comparability work and product-specific justification with less delay.
Based on the regional analysis, the mRNA Vaccine Formulation Stability and Stress Test Systems Market is segmented into North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, and Middle East & Africa across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 12.9% |
| China | 12.4% |
| South Korea | 11.8% |
| United States | 11.1% |
| Germany | 10.2% |
| Belgium | 9.8% |
| Switzerland | 9.6% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America holds a strong position in this category because formulation science and commercial manufacturing operate in close alignment. Labs in this region usually look beyond single-instrument capability and give more weight to method continuity across screening and stability work.
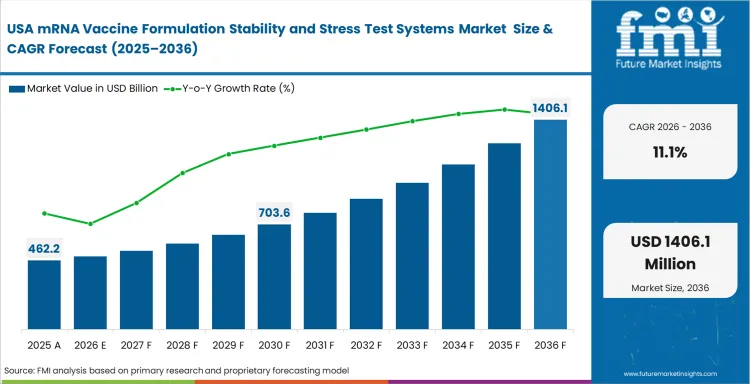
United States: Broad analytical capability and a deep mRNA development base keep the United States at the center of this category. Workflows in the country often move quickly from candidate screening to more formal evaluation, so labs place high value on systems that can keep results comparable across stages. The United States market is expected to grow at a CAGR of 11.1% through 2036. This reflects continued investment in analytical consistency, especially where RNA integrity, particle behavior, and storage response must be assessed together with clear interpretation.
FMI's report includes Canada and Mexico. Laboratory capacity in these countries adds regional support where biologics testing, quality review, and specialized analytical services strengthen wider category adoption.
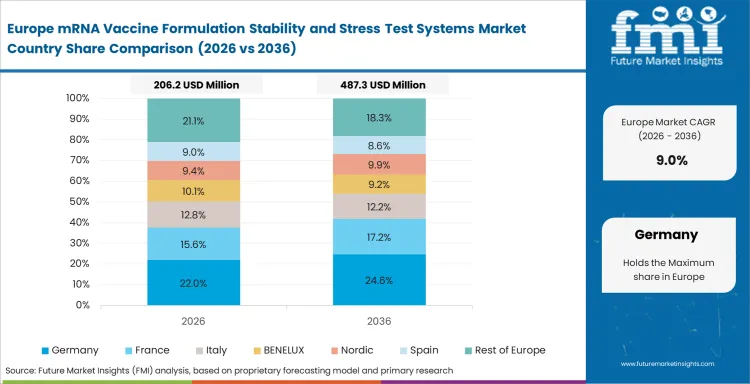
Western Europe holds a solid position because analytical discipline is deeply embedded in vaccine and biologics work. Spending across the region is selective and shaped by validation needs. Laboratories often prefer systems that improve interpretive confidence and fit established validation routines rather than adding tools without a clear analytical purpose.
FMI's report includes France, the Netherlands, Italy, Spain, Benelux, and Nordic countries. Testing centers in these areas function as key European hubs for biologics development, formulation evaluation, and analytical review, which helps maintain consistent regional demand.
Asia Pacific is expanding because local capability in mRNA development and advanced analytical work is strengthening across several countries. Laboratories in this region need systems that can support formulation comparison and assay continuity without dependence on external testing.
FMI's report includes Japan, Australia, New Zealand, ASEAN countries, and the wider South Asia & Pacific region. Analytical laboratories in these countries are adding depth to regional capability, particularly where biologics development and formulation review require more dependable stability interpretation.
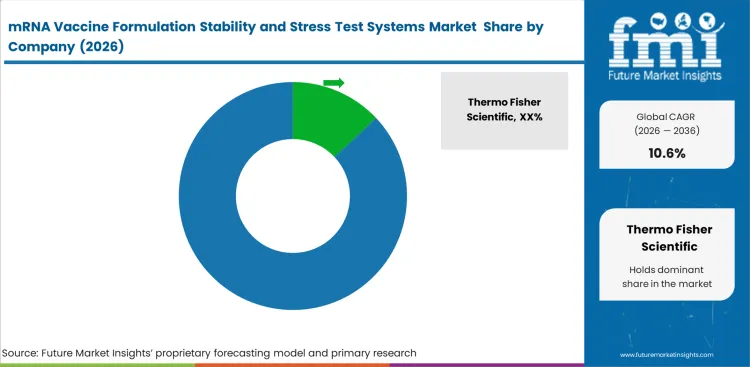
Preference in this category centers on reliability of interpretation rather than broad instrument coverage. Thermo Fisher Scientific, Agilent Technologies, Waters Corporation, Sartorius AG, Malvern Panalytical, Beckman Coulter Life Sciences, and Unchained Labs compete where formulation teams want methods that can connect early comparison work with later analytical use. Suppliers evaluation often depends on how well their systems support continuity across particle analysis and chromatography related data review. Weak continuity can create internal doubt even when individual tools perform well on their own.
Incumbents benefit from established relationships, assay familiarity, and broader technical support around connected workflows. Agilent Technologies, Waters Corporation, Thermo Fisher Scientific, and Sartorius AG are well placed where labs want linked analytical paths instead of isolated instruments. Challengers can still earn space when they solve a specific bottleneck cleanly, especially in high-throughput screening or formulation comparison work. Categories adjacent to analytical laboratory service, large molecule bioanalytical testing services, bioprocess integrity testing systems, and pharmaceutical lipids show why connected method value stays important.

| Metric | Value |
|---|---|
| Quantitative Units | USD 0.8 billion to USD 2.2 billion, at a CAGR of 10.6% |
| Market Definition | mRNA vaccine formulation stability and stress test systems include the platforms used to evaluate formulation behavior under storage and stress conditions. Coverage includes systems that assess RNA integrity, particle behavior, encapsulation consistency, and thermal response. Boundary is defined by formulation stability and stress-testing use. |
| System Type Segmentation | Chromatography systems, Particle analyzers, Thermal analyzers, Stability chambers, Freeze-thaw units, Imaging systems |
| Study Type Segmentation | Accelerated stability, Real-time stability, Freeze-thaw studies, Temperature excursion, Photostability studies, Agitation studies |
| Formulation Format Segmentation | LNP dispersions, Lyophilized cakes, Polymer particles, Emulsion carriers |
| Readout Type Segmentation | Particle size, RNA integrity, Encapsulation, Lipid purity, Thermal transitions, Subvisible particles |
| Workflow Stage Segmentation | Formulation screening, Process development, Clinical release, Commercial QC, Lifecycle monitoring |
| End User Segmentation | Vaccine developers, Biopharma CMOs, CRO labs, Academic centers, Government labs |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, Middle East & Africa |
| Countries Covered | United States, Germany, Belgium, Switzerland, China, South Korea, India, and 40 plus countries |
| Key Companies Profiled | Thermo Fisher Scientific, Agilent Technologies, Waters Corporation, Sartorius AG, Malvern Panalytical, Beckman Coulter Life Sciences, Unchained Labs |
| Forecast Period | 2026 to 2036 |
| Approach | FMI combined primary interviews with formulation scientists, quality leaders, and manufacturing teams. Desk work covered public technical material, company solution pages, and assay practice relevant to mRNA formulation testing. Output was checked against vendor exposure, workflow relevance, and regional capability buildup. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is this market in 2026?
Industry valuation is estimated at USD 0.8 billion in 2026. That number shows a specialized category with a clear role in mRNA formulation work.
What will it be worth by 2036?
Valuation is projected to reach USD 2.2 billion by 2036. That rise points to wider use of testing systems that check storage and stress behavior.
What CAGR is projected from 2026 to 2036?
FMI projects a CAGR of 10.6% for 2026 to 2036. Pace at that level suggests steady expansion rather than a short spike.
Which System Type leads?
Chromatography systems lead System Type. They are expected to account for 24% share in 2026 because they remain important for impurity review, integrity checks, and repeatable comparison work.
Which Study Type leads?
Accelerated stability leads Study Type. It is likely to represent 28% of the market in 2026 because teams want earlier signs of storage weakness.
Which Formulation Format leads?
LNP dispersions lead Formulation Format with 72% share expected in 2026. Most current mRNA vaccine work still revolves around lipid nanoparticle delivery.
Which Readout Type leads?
Particle size leads Readout Type. It is expected to contribute 26% of total market share in 2026 because it gives a quick check on formulation consistency.
Which Workflow Stage leads?
Formulation screening leads with 30% share expected in 2026. Early comparison work decides which formulation paths deserve deeper study.
Which End User leads?
Vaccine developers lead End User with 35% share expected in 2026. Early system choices often begin inside companies that own the formulation program.
Why is this market moving upward?
Labs need clearer answers on RNA condition, particle behavior, and storage response. Better testing systems help them spot weak formulations earlier.
What slows wider adoption?
Main restraint is method fragmentation. Many labs still use tool combinations that work separately but do not read well together.
Which country posts the fastest CAGR?
India leads the country outlook with 12.9% CAGR through 2036. China follows at 12.4%, so India’s faster pace comes from quicker capability buildup.
What does this market cover?
It covers systems used to test how mRNA vaccine formulations behave during storage and stress. That includes tools used to check RNA integrity, particle behavior, encapsulation consistency, and thermal response.
What is outside this market?
Upstream synthesis equipment, fill-finish machinery, cold-chain assets, and general lab tools are outside scope unless they are used directly for formulation stability or stress testing.
Why does accelerated stability matter so much?
It helps teams see early whether a formulation may weaken under storage or stress. That makes it easier to narrow candidates before spending more time and material.
Why does particle size matter so much?
Particle size is often one of the first useful checks on dispersion quality. A poor result can warn teams before deeper testing begins.
Why do vaccine developers stay ahead of CMOs and CRO labs?
They usually make the first decisions on formulation direction and test logic. Later partners often follow the same path.
Why are Asia Pacific countries posting stronger CAGRs than some Western European countries?
Several Asia Pacific countries are building capability from a lower base. Western Europe remains important, though current installed capacity is more mature.
What are labs really looking for when they choose a supplier?
They want test systems that make results easier to connect across assays. Clear method continuity matters more than a long feature list.
Does a higher CAGR mean every country is at the same stage?
No. A faster CAGR can come from a smaller base, rising local capability, or newer program activity. It does not always mean a country already has the deepest installed base.
Why does formulation screening lead the workflow outlook?
Most key choices are made early. Teams need to compare candidates before later methods become fixed.
What should readers take from the 10.6% CAGR?
It points to a category gaining a firmer place in mRNA vaccine work. Testing for storage and stress behavior is becoming harder to treat as an optional step.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.