The mRNA synthesis raw materials market is expected to reach USD 1.8 billion in 2026, with contracted supply extending that base to USD 3.2 billion by 2036 at a CAGR of 5.9%. Procurement begins inside quality frameworks that define grade, impurity limits, and documentation scope before any price discussion occurs. Once enzymes, nucleotides, capping reagents, and buffers enter validated processes, change control governs every substitution. Campaign schedules reserve capacity months ahead. Release timing depends on analytical queues and stability windows. Buyer attention stays fixed on audit history, lot continuity, and deviation handling rather than on unit pricing.
Economic performance inside the mRNA synthesis raw materials market tracks process yield, shelf life utilization, and testing load. Manufacturing plans coordinate fermentation, purification, and formulation steps that determine lead times. Shipment approval depends on completed batch records and closed deviations. Inventory policies position safety stock near fill finish sites to protect start dates. Supplier relationships run through audits and scorecards that influence allocation decisions. Switching costs remain high because assays and validations require alignment. Long range scale plans commit volume early. Financial outcomes follow scrap control, throughput discipline, and documentation workload rather than sales list adjustments.
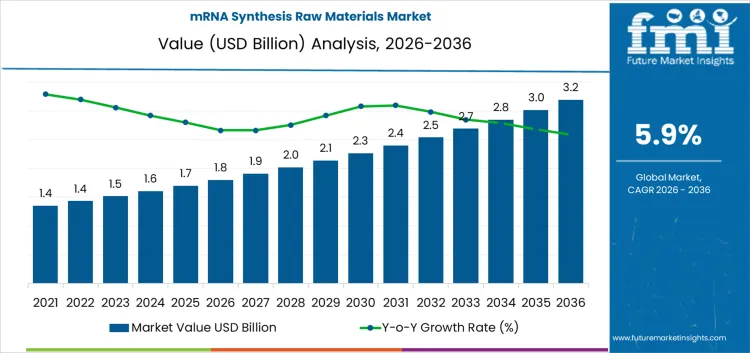
Program driven demand sets the pace for the mRNA synthesis raw materials market rather than short procurement cycles. In 2026, when the mRNA synthesis raw materials market stands near USD 1.8 billion, purchasing is tied to clinical pipelines, scale up plans, and long term manufacturing commitments. Enzymes, nucleotides, capping reagents, and lipid related inputs enter supply chains through qualification tracks that take years rather than quarters. Once a process set is fixed, material consumption follows batch scheduling and yield targets. The stepwise rise toward the low two billion range reflects additional programs reaching later development stages and more sites moving from pilot to routine production. Growth stays measured because process change carries validation cost and operational risk.
Advanced platforms that limit process variation push material selection toward standardized, pre-qualified reagent sets that align with mRNA cancer vaccine biologic lines development standards. Supply discipline and process efficiency shape the later phase of the mRNA synthesis raw materials market. As value moves toward roughly USD 3.2 billion by 2036, spending concentrates on higher purity inputs, tighter lot control, and improved consistency across production runs. Manufacturing groups place weight on reproducibility, shelf life management, and secure sourcing rather than on headline volume alone.
Nucleotide chemistries, enzymes, and capping systems used in routine clinical operations share regulatory principles with broader vaccines market deployment frameworks. Capacity planning favors redundant suppliers and longer contract coverage to protect campaign schedules. The climb from the low two billion into the three billion range reflects cumulative additions of commercial scale programs rather than a single expansion wave. Companies that combine documentation quality, batch reliability, and stable delivery performance tend to secure preferred supplier status.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 1.8 billion |
| Forecast Value (2036) | USD 3.2 billion |
| Forecast CAGR 2026 to 2036 | 5.9% |
mRNA synthesis raw materials include nucleotides, enzymes, capping reagents, buffers, and purification inputs used in in vitro transcription workflows. Before large scale clinical programs, many labs relied on research grade inputs with limited batch consistency and narrow documentation. That approach worked for small studies but created delays when processes moved toward regulated production. The shift to vaccine and therapeutic pipelines changed buying criteria toward traceability, lot stability, and predictable yields. Process developers now lock specific reagent sets early because any later substitution can trigger comparability work. Suppliers are evaluated on impurity profiles, activity ranges, and supply continuity. Demand concentrates in organizations building platform processes rather than single products, since standardization reduces validation effort across multiple candidates and manufacturing sites.
Commercial relationships in this field resemble process partnerships more than simple materials supply contracts. Earlier purchasing focused on availability and unit price, since volumes were small and timelines flexible. Current programs negotiate long term supply, reserved capacity, and change control commitments because schedule risk carries direct financial impact. Manufacturers track cost per gram of RNA output rather than per unit of reagent, which shifts attention to yield and rework rates. Enzyme performance, nucleotide purity, and capping efficiency determine throughput more than reactor size alone. Buyers also require documentation packages that support audits and filings. Over time, competition centers on consistent performance at scale and on the ability to support process transfers between sites, not on catalogue breadth. Suppliers that reduce batch to batch variation gain repeat business across expanding pipelines.
Most spending in mRNA Synthesis Raw Materials Market is decided before any production batch is scheduled. Process flows, impurity tolerances, and release testing frameworks are fixed early and remain unchanged for years. Once a formulation and transcription route is validated, material choices become part of the regulatory file, not a sourcing variable. This makes demand follow pipeline maturity and manufacturing readiness rather than short term program counts. Volume growth depends on how many processes move from development into routine manufacturing. Risk management, not price negotiation, governs vendor selection. The resulting segment structure reflects where control points sit inside the production chain rather than how materials look on a purchase order.

Nucleotides represent about 37% of demand because they scale directly with reaction volume, following patterns established in specialized nucleotide market applications. Their role makes batch consistency and impurity profiling more important than marginal unit cost. Capping agents sit at a lower volume share yet control translation efficiency and stability, which places them under tighter change control. Plasmid DNA acts as the upstream template, where sequence integrity and contamination risk determine acceptance. Enzymes influence yield and error rates, tying their procurement to activity windows consistent with drug discovery enzymes performance standards. Lipids and formulation materials bridge synthesis and delivery, extending quality management into chemical supply chains similar to lipid nanoparticle reagents kits deployment.
Cost exposure by material type is shaped by rework risk and schedule impact rather than by invoice value. Nucleotides benefit from mature synthesis routes and parallel supplier capacity, which supports buffer inventory strategies. Enzymes and capping reagents require narrower storage and handling conditions, increasing discard risk when campaigns shift. Plasmid DNA carries long lead times because fermentation and purification sit upstream of final scheduling decisions. Lipids introduce coordination with formulation and fill lines, which links their availability to downstream capacity. Once a process is locked, substitutions remain rare because each change forces comparability work and regulatory notifications, extending timelines and consuming development resources.
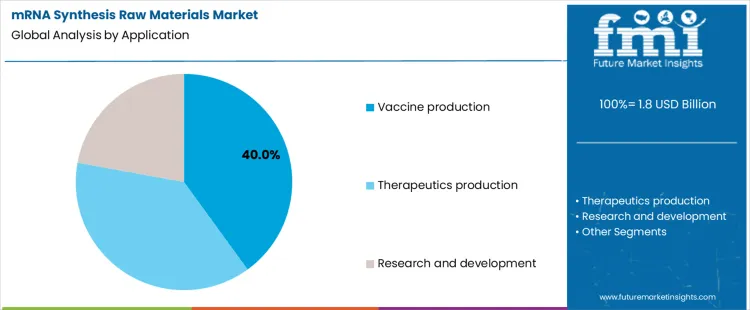
Vaccine production accounts for about 40% of demand in mRNA synthesis raw materials, mirroring the therapeutic scale and supply chain discipline seen across the vaccines market. This segment prioritizes supply continuity, inventory planning, and multi-site material approval. Therapeutics production runs smaller volumes across diverse sequences, reflecting the regulatory rigor required in advanced therapy medicinal products manufacturing. Research and development uses limited quantities across many variants, positioning the market within broader drug discovery enzymes adoption cycles. These structural differences explain why some suppliers build scale driven businesses while others specialize in low volume, high documentation materials.
Application mix also determines how quality agreements and capacity reservations are written. Vaccine programs favor long horizon contracts and dual sourcing because interruptions affect public supply commitments. Therapeutics programs accept narrower volume commitments but require close coordination on changes and deviations. Research organizations value responsiveness and access to experimental grades, even when unit costs are higher. The balance across these uses shapes how suppliers divide capacity between high throughput production lines and small batch controlled environments. Segment shares therefore move with pipeline conversion rates and manufacturing readiness, not with short term shifts in research spending or single product launches.
The mRNA synthesis raw materials market is entering a phase where manufacturing discipline defines purchasing behavior more than research flexibility. As mRNA programs move into routine clinical and commercial operations, enzymes, nucleotides, capping reagents, and purification inputs become fixed elements of audited production systems. Buyers now judge suppliers by documentation depth, change control reliability, batch traceability, and supply continuity. Process stability and regulatory defensibility dominate sourcing decisions. This concentrates demand among fewer suppliers and turns raw material selection into a long-term operational commitment rather than a tactical procurement exercise tied to short project cycles.
The mRNA synthesis raw materials market is being shaped by the rise of common synthesis and purification workflows used across multiple products. Companies design production platforms that limit process variation, which pushes material selection toward standardized, pre-qualified reagent sets. Portfolio optimization now favors stability over customization. Each raw material choice carries long-term consequences for validation scope, documentation, and scale-up planning. Suppliers are therefore assessed on specification discipline, controlled change management, and the ability to support volume expansion without reformulation. Competition increasingly centers on operational reliability and depth of integration into customer manufacturing systems.
The mRNA synthesis raw materials market is generating value through sustained therapy pipelines in oncology, rare disease, and chronic treatment programs that require repeated production campaigns. These programs depend on long-term supply agreements and closer supplier involvement in process definition. Demand is also growing for support with validation packages, comparability documentation, and scale-up continuity. Companies building internal manufacturing capacity prefer suppliers that can co-develop material specifications and support dual-source strategies. This shifts competition away from simple unit pricing toward service depth, regulatory support, and long-term operational alignment.
The mRNA synthesis raw materials market operates under conditions where every approved material becomes part of a regulatory commitment. Any change in enzyme source, nucleotide specification, or purification consumable introduces additional testing, documentation, and review exposure. Operations and quality teams therefore resist changes that could disturb validated processes. Over time, this behavior reinforces dependence on a small group of trusted suppliers. New entrants face not only technical qualification requirements but also organizational risk controls that slow adoption. This creates a structurally conservative purchasing environment focused on continuity and compliance rather than rapid supplier rotation.
| Country | CAGR |
|---|---|
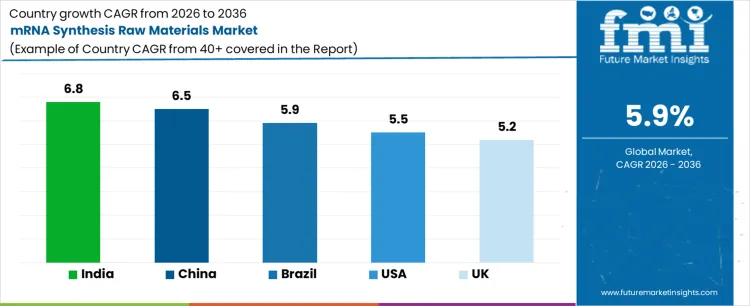
| USA | 5.5% |
| UK | 5.2% |
| China | 6.5% |
| India | 6.8% |
| Brazil | 5.9% |
Demand for mRNA synthesis raw materials is expanding as biopharmaceutical pipelines move from emergency scale programs toward broader vaccine and therapeutic development. India leads with a 6.8% CAGR, supported by pharmaceutical companies and research laboratories that develop mRNA-based vaccines and therapeutics through infrastructure matching the India biopharmaceutical sector expansion. China follows at 6.5%, driven by expanding clinical programs and state backed capacity building in advanced biologics. Brazil shows 5.9%, reflecting gradual localization of vaccine and biologics supply chains. The USA grows at 5.5%, shaped by sustained research funding and stable commercial scale production needs. The UK, at 5.2%, reflects steady demand from academic, clinical, and early stage manufacturing programs.
United States is growing at a CAGR of 5.5%, with innovation hubs leading the adoption of high-purity nucleotides, modified oligonucleotides, and enzymatic reagents. Federal funding for advanced therapeutics strengthens the dense adoption centers in Boston, San Diego, and San Francisco similar to biopharmaceutical contract manufacturing hubs. Vaccine manufacturers and gene therapy developers investing in scalable production lines align with the advanced therapy medicinal products market expansion in North America. Demand is concentrated among vaccine manufacturers, gene therapy developers, and research hospitals conducting large-scale clinical trials. Manufacturers are investing in scalable production lines, robust supply chains, and automation-ready synthesis platforms. Growth is influenced by federal funding for advanced therapeutics, partnerships between universities and biotech firms, and regulatory frameworks ensuring quality and safety. Urban biotech clusters create dense adoption centers, while industrial logistics support timely distribution. The expanding need for personalized medicine, novel vaccines, and precision therapeutics further reinforces the market’s trajectory, making the United States a critical hub for mRNA raw material consumption and innovation.
United Kingdom is witnessing a CAGR of 5.2%, driven by CDMOs and research-focused pharmaceutical companies in Cambridge, London, and Oxford. The market demand comes from developers of mRNA vaccines and novel therapeutics requiring GMP-grade reagents, transcription enzymes, and high-quality nucleotides. Investments focus on reagent stability, regulatory compliance with EMA guidelines, and training for laboratory personnel. Adoption is concentrated in metropolitan biotech corridors, where collaborations between academic institutions and private firms accelerate commercialization. Growth reflects increasing government support for advanced therapy medicinal products, integration of synthetic biology into drug development pipelines, and rising interest in outpatient and hospital-based clinical trials. Manufacturers target operational efficiency, high-throughput synthesis capabilities, and reliable distribution networks to serve this expanding demand.
China is experiencing a CAGR of 6.5% during the forecast period. Government-backed pharmaceutical and biotechnology initiatives coordinate fermentation, purification, and formulation steps consistent with China's biopharmaceutical contract manufacturing market acceleration. State backed capacity building in advanced biologics positions hospitals, vaccine manufacturers, and research institutes for nucleotide premixes integration. Investments prioritize local production, regulatory certification, and integration with automated high-volume synthesis systems. Market adoption is concentrated in coastal industrial clusters, while secondary cities are gradually increasing demand. Growth reflects national strategies to strengthen domestic biotech capabilities, expand vaccine self-sufficiency, and enhance research infrastructure. Urban industrial zones anchor most procurement, whereas regional biotech incubators contribute to pilot-scale synthesis demand. Rising middle-class healthcare access and increased focus on infectious disease prevention reinforce the market trajectory.
The demand for mRNA synthesis raw materials in India is rising a CAGR of 6.8%. Biotech clusters in Bengaluru, Hyderabad, and Pune leverage contract manufacturing services and rising domestic research activity aligned with enzymatic RNA manufacturing market growth. Hospitals and specialty clinics drive adoption by incorporating low-volume, high-quality reagents for experimental studies and pilot-scale production. Investments focus on cost-effective sourcing, compliance with national regulatory requirements, and staff training in proper handling of sensitive materials. Market concentration is highest in metropolitan hubs, while regional industrial parks expand distribution networks. Growth reflects government programs encouraging domestic biopharma manufacturing, the rise of contract manufacturing services, and increasing research collaborations. Demand is also shaped by the need for scalable solutions for clinical trials, ensuring consistent supply of critical raw materials.
Brazil is growing at a CAGR of 5.9%, with major activity centered in São Paulo, Rio de Janeiro, and Curitiba, where hospitals, research institutes, and vaccine producers require reliable raw materials for mRNA synthesis. Operational risk sensitivity drives procurement of reagents with consistent quality and stability. Investments focus on formulation reliability, adherence to ANVISA standards, and training for laboratory personnel. Market concentration is highest in urban healthcare and industrial zones, while regional demand gradually expands through emerging biotech initiatives. Growth reflects increased domestic vaccine development, public health programs promoting advanced therapeutics, and partnerships with international suppliers. Demand is further reinforced by hospitals and private laboratories seeking high-purity nucleotides and enzymes to support both clinical and pilot-scale mRNA production programs.
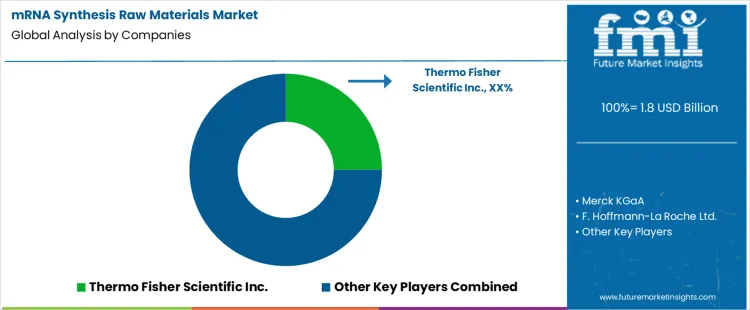
mRNA synthesis raw materials purchasing follows platform decisions rather than routine catalog buying. Process teams select nucleotide chemistries, enzymes, and capping systems while defining the manufacturing route. Those choices bind downstream quality files, validation protocols, and regulatory submissions. Thermo Fisher Scientific, Merck KGaA, Roche, Maravai LifeSciences, New England Biolabs, Jena Bioscience, Yeasen Biotechnology compete at that early design stage. Thermo Fisher Scientific and Merck KGaA benefit from portfolios that cover most process steps. Maravai LifeSciences concentrates on high value capping and nucleotide systems. New England Biolabs holds strong positions in enzymes used during transcription and cleanup. Roche aligns materials with internal platform processes. Jena Bioscience and Yeasen Biotechnology serve programs that value regional supply control and shorter procurement cycles.
Competition reflects risk management more than unit pricing. Manufacturing teams prioritize lot traceability, documentation depth, change notification discipline, and long term capacity access. Audit histories shape approved vendor lists across development programs. Scale up planning pushes buyers toward suppliers that control fermentation, nucleotide synthesis, and formulation under one quality system. Thermo Fisher Scientific uses integrated workflow coverage to secure multi reagent positions inside single accounts. Merck KGaA relies on long standing quality agreements and supply contracts. Maravai LifeSciences benefits from process dependence on specific capping chemistries. New England Biolabs defends enzyme share through performance consistency. Roche supplies materials inside its own development networks. Yeasen Biotechnology and Jena Bioscience compete where regional manufacturing, pricing control, and delivery timing drive decisions. Market shares differ by therapy class, development phase, and regulatory pathway.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD billion |
| Type | Nucleotides, Capping agents, Plasmid DNA, Enzymes, Lipids and formulation materials |
| Application | Vaccine production, Therapeutics production, Research and development |
| Region | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, United States, Canada, Mexico, Brazil, Chile, Saudi Arabia, Turkey, South Africa, and other regional markets |
| Key Companies Profiled | Thermo Fisher Scientific Inc., Merck KGaA, F. Hoffmann-La Roche Ltd., Maravai LifeSciences, New England Biolabs, Jena Bioscience GmbH, Yeasen Biotechnology (Shanghai) Co., Ltd. |
| Additional Attributes | Dollar by sales by type and application; demand driven by pipeline maturity, batch scheduling, and validation lock-in; growth led by vaccines and therapeutics scale-up; purchasing shaped by audit history, lot continuity, change control, shelf life management, and secure long-term supply rather than unit price. |
How big is the mrna synthesis raw materials market in 2026?
The global mrna synthesis raw materials market is estimated to be valued at USD 1.8 billion in 2026.
What will be the size of mrna synthesis raw materials market in 2036?
The market size for the mrna synthesis raw materials market is projected to reach USD 3.2 billion by 2036.
How much will be the mrna synthesis raw materials market growth between 2026 and 2036?
The mrna synthesis raw materials market is expected to grow at a 5.9% CAGR between 2026 and 2036.
What are the key product types in the mrna synthesis raw materials market?
The key product types in mrna synthesis raw materials market are nucleotides , capping agents, plasmid dna, enzymes and lipids & formulation materials.
Which application segment to contribute significant share in the mrna synthesis raw materials market in 2026?
In terms of application, vaccine production segment to command 40.0% share in the mrna synthesis raw materials market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.