mRNA Cancer Vaccine Biologic Lines Market
mRNA Cancer Vaccine Biologic Lines Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
mRNA Cancer Vaccine Biologic Lines Market Forecast and Outlook 2026 to 2036
The mRNA cancer vaccine biologic lines market is valued at USD 585.4 million in 2026 and is projected to reach USD 1,738.6 million by 2036, expanding at a CAGR of 11.50% over the forecast period. Capital allocation in this market is shaped by high upfront intensity in GMP suites, single-use upstream systems, sterile fill-finish capacity, and specialized analytics required for mRNA and lipid nanoparticle processes. Investment decisions are being made selectively because returns are tightly linked to clinical progression, regulatory clarity, and the ability to secure long-term manufacturing utilization rather than one-off campaign output.
Payback timelines are influenced by qualification cycles, validation burdens, and the need to maintain compliant capacity even when programs pivot or pause. Expansion is therefore being favored in modular, scalable line designs that can be repurposed across indications and adjusted for batch-size variability, especially for personalized oncology models. As more assets move into later-stage trials, capital is expected to concentrate around platforms with repeatable tech transfer, predictable yields, and contracting structures that stabilize utilization through 2036.
Quick Stats for mRNA Cancer Vaccine Biologic Lines Market
- mRNA Cancer Vaccine Biologic Lines Market Value (2026): USD 585.4 Million
- mRNA Cancer Vaccine Biologic Lines Market Forecast Value (2036): USD 1,738.6 Million
- mRNA Cancer Vaccine Biologic Lines Market Forecast CAGR 2026 to 2036: 11.50%
- Leading Development Stage in mRNA Cancer Vaccine Biologic Lines Market: Phase II
- Leading Manufacturing Mode in mRNA Cancer Vaccine Biologic Lines Market: In-House
- Key Growth Regions in mRNA Cancer Vaccine Biologic Lines Market: North America, Europe, Asia Pacific
- Top Key Players in mRNA Cancer Vaccine Biologic Lines Market: Moderna, BioNTech, Roche, Pfizer, CureVac, GSK
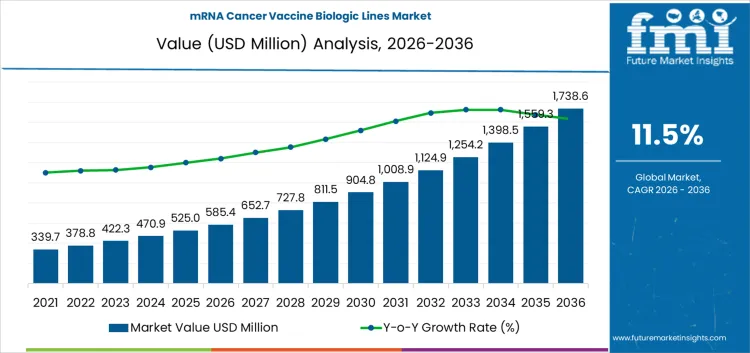
mRNA Cancer Vaccine Biologic Lines Market Key Takeaways
| Metric | Value |
|---|---|
| Industry Value (2026) | USD 585.4 Million |
| Forecast Value (2036) | USD 1,738.6 Million |
| Forecast CAGR 2026 to 2036 | 11.50% |
How Is the Global mRNA Cancer Vaccine Biologic Lines Market Advancing?
The global mRNA cancer vaccine biologic lines market is advancing at a strong pace, driven by the growing clinical focus on personalized and targeted oncology therapies. Biologic production lines dedicated to mRNA cancer vaccines are increasingly being established to support clinical trials and commercial-scale manufacturing, as pharmaceutical and biotechnology companies expand their oncology pipelines based on mRNA platforms.
A key driver supporting market growth is the rising investment in mRNA-based cancer immunotherapies and the expansion of clinical development programs across multiple cancer indications. Unlike conventional vaccine manufacturing, mRNA cancer vaccines require highly controlled biologic lines capable of supporting complex processes such as in vitro transcription, lipid nanoparticle formulation, and sterile fill-finish operations. This has increased demand for specialized production infrastructure with high flexibility and rapid scale-up capabilities.
Technological advancements in biologic manufacturing systems are further accelerating market expansion. Improvements in single-use bioprocessing equipment, automation, and modular production line designs are enabling faster facility deployment and improved batch consistency. In addition, enhanced quality control systems and digital monitoring tools are improving process reliability and regulatory compliance. As mRNA oncology programs continue to progress toward late-stage trials and commercialization, the biologic lines market supporting mRNA cancer vaccines is expected to experience sustained growth over the forecast period.
What Is the Segment-Wise Overview of the mRNA Cancer Vaccine Biologic Lines Market?
The mRNA cancer vaccine biologic lines market is shaped by development-stage concentration and manufacturing strategy choices. Phase II programs dominate with a 38% share, reflecting a strong pipeline of oncology-focused mRNA candidates advancing into efficacy-driven clinical trials. This stage requires scalable yet flexible biologic lines capable of supporting trial expansion while enabling rapid process optimization. On the manufacturing side, in-house production leads with a 58% share, as developers prioritize tight process control, intellectual property protection, and fast iteration cycles. In-house biologic lines are particularly important for personalized cancer vaccines, where customization, quality oversight, and short production timelines are critical.
Why Does Phase II Dominate the mRNA Cancer Vaccine Biologic Lines Market?
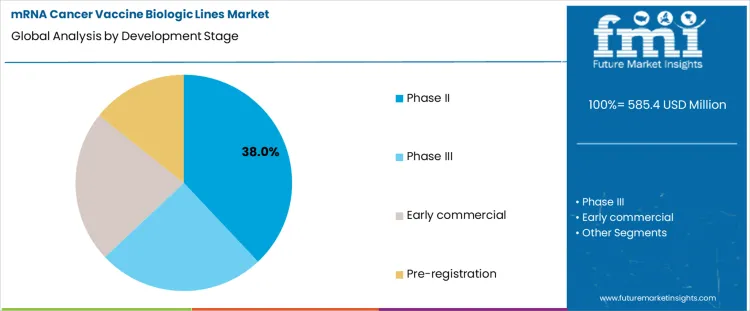
Phase II development accounts for 38% of the development stage segment in the mRNA Cancer Vaccine Biologic Lines Market, reflecting the high volume of candidates transitioning from early validation to efficacy-focused clinical evaluation. At this stage, developers scale biologic production to support multi-site trials while optimizing formulation stability, dosing consistency, and process reproducibility. mRNA cancer vaccines in Phase II often target personalized or tumor-specific antigens, requiring flexible and modular biologic line configurations. Manufacturing at this stage emphasizes process refinement rather than full commercial scale, driving demand for adaptable production systems. The concentration of assets in Phase II also highlights continued investment in oncology-focused mRNA pipelines, as developers seek to demonstrate clinical benefit before committing to large-scale commercial manufacturing infrastructure.
Why Does In-House Manufacturing Lead the mRNA Cancer Vaccine Biologic Lines Market?
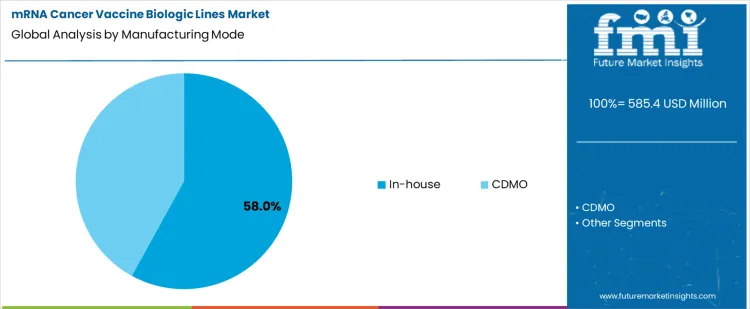
In-house manufacturing represents 58% of total manufacturing mode demand, driven by the strategic importance of process control, intellectual property protection, and rapid iteration in mRNA cancer vaccine development. Developers often prefer in-house biologic lines to tightly manage critical steps such as mRNA synthesis, lipid nanoparticle formulation, and fill-finish operations. This approach enables faster process optimization, improved quality oversight, and reduced dependency on external capacity availability. In-house production is particularly important for personalized cancer vaccines, where short turnaround times and batch customization are essential. While CDMOs play a growing role in later-stage and commercial supply, in-house manufacturing remains dominant during clinical development, supporting agility and technical control as mRNA oncology pipelines continue to expand.
What Are the Key Dynamics for the mRNA Cancer Vaccine Biologic Lines Market?
The mRNA cancer vaccine biologic lines market is driven by the rising focus on next-generation oncology therapies that harness messenger RNA (mRNA) technology to trigger targeted immune responses against cancer cells. mRNA cancer vaccines represent a promising class of biologics designed to educate the immune system to recognize and attack tumor-specific antigens. Key market dynamics include advancements in mRNA therapeutic platforms, increasing prevalence of various cancer types, and strong interest from biopharmaceutical companies and research institutions in personalized immunotherapy. The ability of mRNA vaccines to be rapidly designed and manufactured enhances their potential in both personalized and off-the-shelf oncology applications.
Why Is the mRNA Cancer Vaccine Biologic Lines Market Growing?
The market is expanding due to the success of mRNA technologies in infectious disease vaccines, which has accelerated research and investment into oncology applications. Continuous improvements in delivery systems, such as lipid nanoparticles, are enhancing the stability and cellular uptake of mRNA cancer vaccines. Growing clinical pipeline activity across multiple cancer indications, increasing incidence and mortality associated with cancer globally, and enhanced funding for cancer research are collectively supporting market growth. Strategic collaborations between biotech firms, academic centers, and large pharmaceutical companies are helping transition mRNA vaccine candidates through critical clinical stages.
How Are the Key Drivers Shaping the mRNA Cancer Vaccine Biologic Lines Market?
Key drivers shaping the market include strong technical progress in mRNA design, formulation, and delivery platforms that improve immunogenicity and safety profiles of cancer vaccines. The scalability and modular nature of mRNA manufacturing allows for rapid adaptation of vaccine constructs to target diverse tumor antigens, which is critical for personalized medicine approaches. Increasing regulatory support for innovative oncology therapies and expanded clinical trial enrollments are also boosting development activity. Rising cancer incidence rates worldwide and growing investment in precision oncology are encouraging biopharma companies to expand their mRNA vaccine pipelines.
What Is the Growth Outlook for the mRNA Cancer Vaccine Biologic Lines Market by Country?
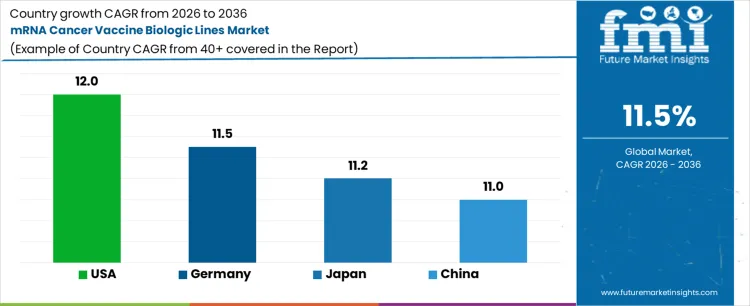
| Country | CAGR (%) |
|---|---|
| USA | 12.0% |
| Germany | 11.5% |
| Japan | 11.2% |
| China | 11.0% |
The mRNA cancer vaccine biologic lines market is expanding across major biopharmaceutical regions as oncology-focused mRNA pipelines advance toward commercialization. The USA leads with a 12.0% CAGR, supported by strong clinical activity, funding, and advanced biologics infrastructure. Germany follows at 11.5%, driven by leadership in mRNA research and precision oncology manufacturing. Japan grows at 11.2%, supported by investment in advanced therapeutics and high manufacturing standards. China expands at 11.0%, fueled by rapid biotech sector growth and government support for innovative cancer treatments. As personalized cancer vaccines gain momentum, demand for specialized mRNA biologic production lines is expected to rise across all regions.
What Is Driving the Growth of the mRNA Cancer Vaccine Biologic Lines Market in the USA?
The mRNA cancer vaccine biologic lines market in the USA is growing at a CAGR of 12.0%, driven by strong clinical pipeline activity and continued investment in mRNA-based oncology research. The USA hosts a large concentration of biotechnology firms and pharmaceutical companies developing personalized and therapeutic cancer vaccines using mRNA platforms. Expansion of clinical trials, particularly in melanoma, lung cancer, and solid tumors, is increasing demand for scalable and compliant biologic manufacturing lines. Advanced biologics infrastructure, availability of skilled bioprocessing talent, and strong collaboration between academia, biotech companies, and contract manufacturing organizations support rapid capacity expansion. In addition, regulatory familiarity with mRNA platforms following their use in infectious disease vaccines has improved development timelines. Public and private funding for cancer immunotherapy remains robust, encouraging further investment in dedicated mRNA biologic lines. As personalized oncology approaches gain traction, demand for specialized mRNA cancer vaccine production capacity is expected to grow steadily.
How Is the mRNA Cancer Vaccine Biologic Lines Market Expanding in Germany?
The mRNA cancer vaccine biologic lines market in Germany is expanding at a CAGR of 11.5%, supported by the country’s strong biotechnology ecosystem and leadership in mRNA research. Germany plays a key role in advancing mRNA-based cancer therapies, supported by well-established pharmaceutical manufacturing infrastructure and research institutions. Growing focus on precision oncology and individualized cancer treatment is driving demand for flexible biologic production lines capable of small-batch and multi-product manufacturing. Germany’s emphasis on high-quality bioprocessing standards and regulatory compliance supports adoption of advanced single-use and modular production systems. In addition, public funding initiatives and innovation programs focused on biotechnology and oncology research are strengthening domestic manufacturing capabilities. Collaboration between biotech innovators and contract development and manufacturing organizations is further expanding capacity. As clinical pipelines mature and commercialization pathways become clearer, demand for mRNA cancer vaccine biologic lines in Germany is expected to increase consistently.
How Is the mRNA Cancer Vaccine Biologic Lines Market Developing in Japan?
The mRNA cancer vaccine biologic lines market in Japan is growing at a CAGR of 11.2%, driven by increasing investment in advanced therapeutics and cancer immunotherapy research. Japan’s pharmaceutical sector is actively exploring mRNA platforms to address unmet needs in oncology, particularly for difficult-to-treat cancers. Expansion of translational research and clinical development programs is increasing the need for specialized biologic manufacturing lines that meet stringent quality and safety requirements. Japan’s strong focus on precision medicine and aging population challenges further supports interest in innovative cancer treatment approaches. Government-backed initiatives aimed at strengthening domestic biopharmaceutical manufacturing are also encouraging capacity development. In addition, Japanese manufacturers prioritize process reliability, consistency, and automation, driving adoption of advanced biologic line technologies. As mRNA cancer vaccine candidates progress through clinical stages, demand for dedicated production infrastructure is expected to rise steadily.
How Is the mRNA Cancer Vaccine Biologic Lines Market Expanding in China?
The mRNA cancer vaccine biologic lines market in China is expanding at a CAGR of 11.0%, supported by rapid growth in domestic biotechnology capabilities and increasing focus on innovative oncology treatments. China is investing heavily in mRNA technology development to strengthen its biopharmaceutical sector and reduce reliance on imported advanced therapies. Rising cancer incidence and growing clinical research activity are driving demand for scalable biologic manufacturing infrastructure. Chinese biotech firms are expanding pilot and commercial-scale biologic lines to support mRNA vaccine development and clinical trial supply. Government initiatives promoting innovation in biologics manufacturing and oncology drug development are further accelerating market growth. Improvements in regulatory frameworks and manufacturing quality standards are also supporting wider adoption of advanced production systems. As local mRNA cancer vaccine pipelines expand, demand for biologic lines is expected to increase steadily.
How Are Companies Competing in the mRNA Cancer Vaccine Biologic Lines Market?
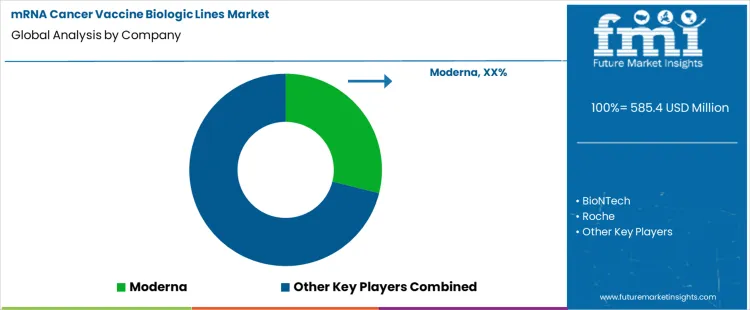
Competition in the mRNA cancer vaccine biologic lines market centers on platform scalability, manufacturing flexibility, regulatory readiness, and the ability to support personalized and tumor-specific vaccine production. Unlike conventional biologics, mRNA cancer vaccines require rapid batch changeovers, small-lot manufacturing, and tight control over lipid nanoparticle (LNP) formulation, encapsulation, and cold-chain handling. As oncology pipelines increasingly shift toward individualized and neoantigen-based vaccines, companies differentiate by how efficiently their biologic lines can move from sequence design to GMP-grade clinical material while maintaining consistency and compliance.
Moderna and BioNTech lead competition through vertically integrated mRNA manufacturing platforms purpose-built for oncology applications. Moderna emphasizes modular, digitally enabled biologic lines that support fast scale-up and rapid customization for personalized cancer vaccines. BioNTech differentiates through flexible production lines optimized for patient-specific batches, supported by in-house mRNA design, LNP expertise, and tightly coupled clinical development capabilities. Pfizer and Roche compete by leveraging large-scale biologics infrastructure and global regulatory experience, pairing mRNA vaccine development with established oncology portfolios and clinical trial networks. Their strategies focus on industrializing mRNA production while ensuring robustness, reproducibility, and global supply readiness.
CureVac and GSK compete through platform innovation and strategic partnerships. CureVac emphasizes optimized mRNA constructs and streamlined manufacturing processes aimed at improving yield and stability, supporting cost-effective biologic line operation for cancer vaccines. GSK brings deep biologics manufacturing experience, quality systems, and oncology commercialization strength, positioning its biologic lines to support late-stage development and global launch requirements. Across the market, competitive advantage is defined by speed to clinic, flexibility for personalized therapies, process automation, and the ability to meet stringent oncology-grade regulatory and quality expectations as mRNA cancer vaccines move toward broader clinical adoption.
Key Players in mRNA Cancer Vaccine Biologic Lines Market
- Moderna
- BioNTech
- Roche
- Pfizer
- CureVac
- GSK
Scope of Report
| Attributes | Description |
|---|---|
| Quantitative Unit (2026) | USD Million |
| Development Stage | Phase II, Phase III, Early Commercial, Pre-Registration |
| Manufacturing Mode | In-House, CDMO |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Rest of Asia Pacific, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, Rest of Europe, United States, Canada, Mexico, Brazil, Chile, Rest of Latin America, Kingdom of Saudi Arabia, Other GCC Countries, Turkey, South Africa, Other African Union, Rest of Middle East & Africa |
| Key Companies Profiled | Moderna, BioNTech, Roche, Pfizer, CureVac, GSK |
| Additional Attributes | Dollar sales by development stage and manufacturing mode; regional market size and forecast analysis; growth outlook across major regions; capacity expansion and technology adoption trends in mRNA cancer vaccine biologic production lines; evaluation of scale-up timelines, regulatory readiness, and outsourcing patterns across in-house and CDMO manufacturing models. |
mRNA Cancer Vaccine Biologic Lines Market by Key Segments
Development Stage
- Phase II
- Phase III
- Early commercial
- Pre-registration
Manufacturing Mode
- In-house
- CDMO
Region
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- European Medicines Agency (EMA). (2024). Guideline on Quality, Non-Clinical, and Clinical Requirements for mRNA Vaccines. European Medicines Agency.
- U.S. Food and Drug Administration (FDA). (2023). Guidance for Industry: Chemistry, Manufacturing, and Control (CMC) Information for Human Gene Therapy Investigational New Drug Applications (INDs). Center for Biologics Evaluation and Research (CBER).
- World Health Organization (WHO). (2024). WHO Technical Report Series No. 1044 — Annex 2: Guidelines for Good Manufacturing Practices for Biological Products. World Health Organization, Geneva.
- International Council for Harmonisation (ICH). (2023). ICH Q5A(R2): Viral Safety Evaluation of Biotechnology Products Derived from Cell Lines of Human or Animal Origin. International Council for Harmonisation Secretariat.
- European Commission. (2023). Commission Delegated Regulation (EU) 2023/503 supplementing Regulation (EU) 536/2014 as regards Good Manufacturing Practice for Investigational Medicinal Products. Official Journal of the European Union.
Frequently Asked Questions
How big is the mrna cancer vaccine biologic lines market in 2026?
The global mrna cancer vaccine biologic lines market is estimated to be valued at USD 585.4 million in 2026.
What will be the size of mrna cancer vaccine biologic lines market in 2036?
The market size for the mrna cancer vaccine biologic lines market is projected to reach USD 1,738.6 million by 2036.
How much will be the mrna cancer vaccine biologic lines market growth between 2026 and 2036?
The mrna cancer vaccine biologic lines market is expected to grow at a 11.5% CAGR between 2026 and 2036.
What are the key product types in the mrna cancer vaccine biologic lines market?
The key product types in mrna cancer vaccine biologic lines market are phase ii, phase iii, early commercial and pre-registration.
Which manufacturing mode segment to contribute significant share in the mrna cancer vaccine biologic lines market in 2026?
In terms of manufacturing mode, in-house segment to command 58.0% share in the mrna cancer vaccine biologic lines market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Development Stage
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Development Stage , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Development Stage , 2026 to 2036
- Phase II
- Phase III
- Early commercial
- Pre-registration
- Phase II
- Y to o to Y Growth Trend Analysis By Development Stage , 2021 to 2025
- Absolute $ Opportunity Analysis By Development Stage , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Manufacturing Mode
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Manufacturing Mode, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Manufacturing Mode, 2026 to 2036
- In-house
- CDMO
- In-house
- Y to o to Y Growth Trend Analysis By Manufacturing Mode, 2021 to 2025
- Absolute $ Opportunity Analysis By Manufacturing Mode, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Development Stage
- By Manufacturing Mode
- By Country
- Market Attractiveness Analysis
- By Country
- By Development Stage
- By Manufacturing Mode
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Development Stage
- By Manufacturing Mode
- By Country
- Market Attractiveness Analysis
- By Country
- By Development Stage
- By Manufacturing Mode
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Development Stage
- By Manufacturing Mode
- By Country
- Market Attractiveness Analysis
- By Country
- By Development Stage
- By Manufacturing Mode
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Development Stage
- By Manufacturing Mode
- By Country
- Market Attractiveness Analysis
- By Country
- By Development Stage
- By Manufacturing Mode
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Development Stage
- By Manufacturing Mode
- By Country
- Market Attractiveness Analysis
- By Country
- By Development Stage
- By Manufacturing Mode
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Development Stage
- By Manufacturing Mode
- By Country
- Market Attractiveness Analysis
- By Country
- By Development Stage
- By Manufacturing Mode
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Development Stage
- By Manufacturing Mode
- By Country
- Market Attractiveness Analysis
- By Country
- By Development Stage
- By Manufacturing Mode
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Development Stage
- By Manufacturing Mode
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Development Stage
- By Manufacturing Mode
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Development Stage
- By Manufacturing Mode
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Development Stage
- By Manufacturing Mode
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Development Stage
- By Manufacturing Mode
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Development Stage
- By Manufacturing Mode
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Development Stage
- By Manufacturing Mode
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Development Stage
- By Manufacturing Mode
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Development Stage
- By Manufacturing Mode
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Development Stage
- By Manufacturing Mode
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Development Stage
- By Manufacturing Mode
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Development Stage
- By Manufacturing Mode
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Development Stage
- By Manufacturing Mode
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Development Stage
- By Manufacturing Mode
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Development Stage
- By Manufacturing Mode
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Development Stage
- By Manufacturing Mode
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Development Stage
- By Manufacturing Mode
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Development Stage
- By Manufacturing Mode
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Development Stage
- By Manufacturing Mode
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Development Stage
- By Manufacturing Mode
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Development Stage
- By Manufacturing Mode
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Development Stage
- By Manufacturing Mode
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Development Stage
- By Manufacturing Mode
- Competition Analysis
- Competition Deep Dive
- Moderna
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- BioNTech
- Roche
- Pfizer
- CureVac
- GSK
- Moderna
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Development Stage , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Manufacturing Mode, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Development Stage , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Manufacturing Mode, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Development Stage , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Manufacturing Mode, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Development Stage , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Manufacturing Mode, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Development Stage , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Manufacturing Mode, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Development Stage , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Manufacturing Mode, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Development Stage , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Manufacturing Mode, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Development Stage , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Manufacturing Mode, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Development Stage , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Development Stage , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Development Stage
- Figure 6: Global Market Value Share and BPS Analysis by Manufacturing Mode, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Manufacturing Mode, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Manufacturing Mode
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Development Stage , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Development Stage , 2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by Development Stage
- Figure 23: North America Market Value Share and BPS Analysis by Manufacturing Mode, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Manufacturing Mode, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Manufacturing Mode
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Development Stage , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Development Stage , 2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by Development Stage
- Figure 30: Latin America Market Value Share and BPS Analysis by Manufacturing Mode, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Manufacturing Mode, 2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by Manufacturing Mode
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Development Stage , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Development Stage , 2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by Development Stage
- Figure 37: Western Europe Market Value Share and BPS Analysis by Manufacturing Mode, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Manufacturing Mode, 2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by Manufacturing Mode
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Development Stage , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Development Stage , 2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Development Stage
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Manufacturing Mode, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Manufacturing Mode, 2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Manufacturing Mode
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Development Stage , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Development Stage , 2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by Development Stage
- Figure 51: East Asia Market Value Share and BPS Analysis by Manufacturing Mode, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Manufacturing Mode, 2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by Manufacturing Mode
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Development Stage , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Development Stage , 2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Development Stage
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Manufacturing Mode, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Manufacturing Mode, 2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Manufacturing Mode
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Development Stage , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Development Stage , 2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Development Stage
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Manufacturing Mode, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Manufacturing Mode, 2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Manufacturing Mode
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

